��Ŀ����
3���������ʳס�ж��벻����ѧ����ѧ������������أ���1������Ҫ��ѡ��ǡ����ѡ������ĸ������գ�
�١��������ࡱ�еķ�Ӧ����ΪA��
A��Ԫ�� B������ C������
�����ࡢ��֬�������ʡ����Ρ�ˮ��ά�����������ȱ����Ӫ�����ʣ�����ά�����У������ڷ��λ�Ѫ������C��
A������A B������B1 C������C
�ۿ�����ҩ����ֹ����ϸ�������Ĺ��ܣ�����ҩ�����ڿ�����ҩ����B��
A����˾ƥ�� B����ù�� C������ҩ������������
�����д��������ķ����У�����ȷ����C��
A���������÷�ֽ B���������������� C������Ͼɵ��
��2����ȫ��ÿ�걻��ʴ�ĸ����У��绯ѧ��ʴ�Ȼ�ѧ��ʴ��ռ�����ߣ���ߡ��͡�����
���� ��1��������������漰���ĺ�����˵�������ǹ�ע���ʵ����Ԫ�أ�
��ά����C�ܷ��λ�Ѫ����
�ۿ�����������������ģ�������ϸ���Լ������������
�ܸ��ݴ�������Ӧ���ȷ�����գ�����ȼ�����Դ���˷ѣ����������ķ�������ʲ�ȡ��ѧ�ķ����������������ⶪ�����������գ�
��2������Ϊ�Ͻ𣬸������γ�ԭ��أ������绯ѧ��ʴ��
��� �⣺��1���٣����������ࡱ����Ʒ����Щ��Ʒ����ǿ���ķ�������˵����Щ���ʵĴ��ڣ�������ע��ʲô��ʽ���ڣ����߱���˵����δ��ڣ����Ծ��ú�۵�Ԫ��������˵�����������ΪԪ�أ�
��ѡ��A��
��ά����C�ܷ��λ�Ѫ����
�ʴ�Ϊ��C��
�۰�˾ƥ���ǽ�����ʹҩ����ù���ǿ����أ����������ǿ���ҩ��
�ʴ�Ϊ��B��
��A���������÷�ֽ���Խ�Լ��Դ�����ٻ�����Ⱦ����A��ȷ��
B���������������ޣ����Խ�Լ��Դ����B��ȷ��
C������Ͼɵ�أ��ܹ�����������ˮ���ؽ�����Ⱦ�������ڻ�����������C����
��ѡ��C��
��2������ƽʱʹ�õĸ�����Ʒ������ǺϽ𣬹ʷ����绯ѧ��ʴ�ļ���ҪԶ���ڻ�ѧ��ʴ�����绯ѧ��ʴ����ɸ�����ʴ����Ҫԭ��
�ʴ�Ϊ���ߣ�
���� ����Ϊ�ۺ��⣬�����������г�����ҩ�PҩЧ��������Ⱦ�������������ĸ�ʴ���������Ϥ���֪ʶ�ǽ���ؼ���ע�ⳣ�������������Լ���ȷ�Ĵ���������ע������֪ʶ�Ļ��ۣ�

 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д���ͨ����������������ᷴӦ���й�ʵ�飬�о���Ӧ���ʵ�Ӱ�����أ�ʵ��������ݼ�¼���£�
ʵ����� | ��Ӧ�¶�/�� | �μӷ�Ӧ������ | ||||
| Na2S2O3 | H2SO4 | H2O | ||||
| V/ml | c/��mol•L-1�� | V/ml | c/��mol•L-1�� | V/ml | ||
| A | 40 | 5 | 0.1 | 10 | 0.1 | 5 |
| B | 20 | 5 | 0.1 | 10 | 0.1 | 5 |
| C | 20 | 5 | 0.1 | 5 | 0.1 | 10 |
��2��A��B����ϱȽϣ����о����������¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죮
��3��������ʵ���У���Ӧ�����������ǣ���ʵ����ţ�C��
���Na2S2O3�������������ʽ���������Ԥ���Լ�ʵ��̽������Ӧ������Һ�н��У���
| Ԥ�� | ʵ����� | ʵ������ | ?������� | |
| ̽���� | Na2S2O3��Һ�ʼ��� | ��ҺpH=8 | S2O32-+H2O HS2O3-+OH- | |
| ̽���� | Na2S2O3�л�ԭ�� | ��������ˮ�еμ�Na2S2O3��Һ | ��ˮ��ɫ��dz |
��5������̽�����е������ԭ���ǣ������ӷ���ʽ��ʾ��S2O32-+4Cl2+5H2O�T2SO42-+8Cl-+10H+��
��������Ϸ��֣�����Ƭ��ӡ�����У�Ҫ���ع��ĵ�Ƭ�����ڹ���Na2S2O3��Һ�У�Na2S2O3���Ժ�Ϳ�ڽ�Ƭ�ϵ�Ag+��Ӧ�γɿ����Ե���ɫ����[Ag��S2O3��2]3-���Ӷ���Ӱ�����ã�
����ͬѧ����֤��һ���̣�����Aͬѧ��Na2S2O3��Һ����AgNO3��Һ�У������������ɰ�ɫ������������ֹͣ��ʵ�飮һ��ʱ����ֳ�����Ϊ��ɫ�����ˣ�����Ϊ��ɫ���ʿ�����Ag2O��Ag2S��
Bͬѧ��Aͬѧ������ʵ��Ļ����Ϸ�������������Һ��������Һ�г���δ��Ӧ���AgNO3�⣬ֻ��������Na+��SO42-��
��6��Bͬѧ��Ϊ�������Ϸ����������жϺ�ɫ������Ag2S�������Bͬѧ���������жϵ���������Һ����SO42-��˵������Ԫ�����ۣ�������ϵ�б�����Ԫ�ؽ��ۣ�������Ag2O����ϵ����Ԫ�ؽ��ۣ�����ֻ������һ������Ԫ�ؽ�Ϊ-2�ۣ�����Ag2S����
��7��Aͬѧδ����֤��Ӱ���̵�ԭ������Ƕ�Ӱ����Ҫ��Ag+�����Na2S2O3��Һ��Ӧ���ɿ����Ե���ɫ����[Ag��S2O3��2]3-��Na2S2O3��������Aͬѧ��Na2S2O3��Һ����AgNO3��Һ�У�AgNO3�����������ڷ�Ӧ��ı�����ϵ��ͬ����˷�Ӧ����ͬ��
| A�� | �ϳ���ά���ά���������л��ǽ������� | |
| B�� | ���ۡ���ά�غ���֬��������Ȼ�߷��ӻ����� | |
| C�� | �������������������������ܳɸ֡������ľ�Ϊ��ѧ�仯 | |
| D�� | �������������е����ʵ�����ǽ���Ԫ�ص���ɫ��Ӧ |
| ѡ�� | �������ʵ | ���� |
| A | �ý��ݹ����������Һ�Ĺ���������ˮ�� | ��ϩ���������ط���������ԭ��Ӧ |
| B | ��Na2S��ȥ��ˮ�е�Cu2+��Hg2+ | Na2S����ǿ��ԭ�� |
| C | ��������������ɫͿ�� | �����������ᷴӦ |
| D | ��������Һ���ͭ�������ͭ�� | ������Һ��Al3+����ͭ�ⷴӦ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | Ԫ��X��Y��Z��ԭ�Ӱ뾶��С��Y��Z��X | |
| B�� | Ԫ��W��Z���Ȼ����У���ѧ��������ͬ���Ҹ�ԭ�Ӿ�����8���� | |
| C�� | Ԫ��X��Ԫ��Y�γɵĻ�������һ�ֽϺõ��ͻ���� | |
| D�� | Ԫ��X����Ԫ��Z�����3�ֶ��������� |
| A�� | ʯī | B�� | HCl | C�� | CO2 | D�� | BaCO3 |
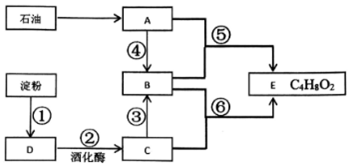
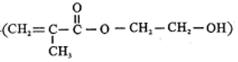 ���������з����ϳɣ�
���������з����ϳɣ�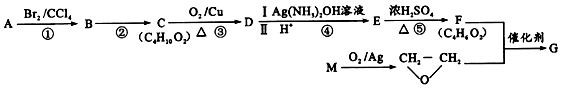
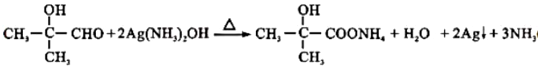 ��B��C��Ӧ����ʽΪ
��B��C��Ӧ����ʽΪ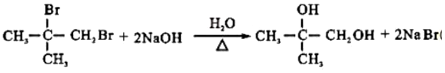 ��
��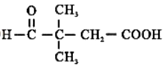 ��
�� ��
��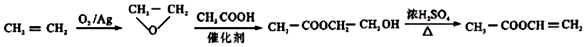 ��
��