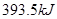��Ŀ����
��5�֣���֪��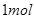 C��
C��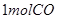 �ֱ���ʽ��Ӧ��ȼ�գ���
�ֱ���ʽ��Ӧ��ȼ�գ���
��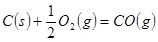 ����
����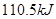
��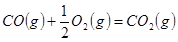 ����
����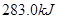
��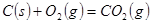 ����
����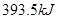
��ش�
��1������������Ӧ�����ݣ�ú̿���ȼ�վ��е��ŵ���__________��
a. �ų��������Լ��Դ b. ���ٶԻ�������Ⱦ
��2�����ݷ�Ӧ�ڣ��Ͽ�1 mol CO��g����0.5 mol O2��g���еĻ�ѧ�������յ����������γ�1 mol CO2��g���еĻ�ѧ�����ų�������__________����ࡱ���١�����
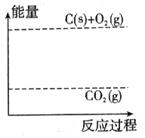
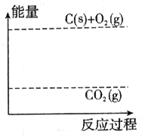
��3������ͼ�л�����Ӧ�������淴Ӧ���̵ı仯���ߡ�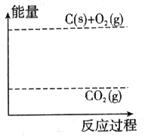
��4������̼ͨ������������Ӧ����CO��g��������������Ӧ����CO2��g�������ų�������֮������ͬ������̼��������ȫ��Ӧ����CO2��g�����ų��������Ĺ�ϵ��__________�����ȡ����ȡ�����
��1��a b��2�֣����1����1�֣� ��2���٣�1�֣�
��3��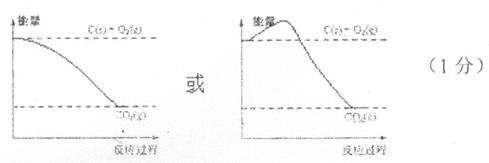
��4����ȣ�1�֣�
����

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
��֪��1mol�����顢1mol�춡��ֱ���ʽ��Ӧ��ȼ�գ���
CH3CH2CH2CH3��g��+6.5O2��g����4CO2��g��+5H2O��l������2878kJ
��CH3��2CHCH3��g��+6.5O2��g����4CO2��g��+5H2O��l������2869kJ
����˵����ȷ���ǣ�������
CH3CH2CH2CH3��g��+6.5O2��g����4CO2��g��+5H2O��l������2878kJ
��CH3��2CHCH3��g��+6.5O2��g����4CO2��g��+5H2O��l������2869kJ
����˵����ȷ���ǣ�������
| A���춡��ת��Ϊ������Ĺ�����һ�����ȹ��� | B����������ȶ��Դ����춡�� | C����������Ӵ�������������춡����� | D���춡������е�̼�����������Ķ� |
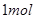 C��
C��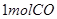 �ֱ���ʽ��Ӧ��ȼ�գ���
�ֱ���ʽ��Ӧ��ȼ�գ���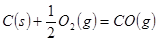 ����
����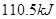
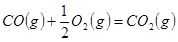 ����
����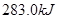
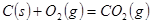 ����
����