��Ŀ����
�о��л���һ�㾭�����¼����������裺���롢�ᴿ��ȷ��ʵ��ʽ��ȷ������ʽ��ȷ���ṹʽ�����������о��л���ķ�������ȷ���ǣ�������
| A���ؽᾧ��һ�������ڱ��ᴿ���л������ܼ��е��ܽ�����¶�Ӱ��ϴ�Ļ����ķ��� |
| B�����л�����Ӻ������ͼ���о�������ȷ���л�����Է������� |
| C��ȼ�շ���ȷ���л�����ӹ����ŵ���Ч���� |
| D������ͨ�����ڷ���ȷ���л�����ӵ�Ԫ����� |
���㣺��ѧ�о���������������
ר�⣺
������A���¶�Ӱ���ܽ�ȣ������ᴿ����ܽ�����¶ȱ仯�������ؽᾧ�����룻
B���������ͼ��ȷ���л�������еĻ��ţ�
C��ȼ�շ���ȷ���л����е����ʽ��
D���������ܼ�¼�������ӡ���Ƭ���ӵ����������
B���������ͼ��ȷ���л�������еĻ��ţ�
C��ȼ�շ���ȷ���л����е����ʽ��
D���������ܼ�¼�������ӡ���Ƭ���ӵ����������
���
�⣺A���¶�Ӱ���ܽ�ȣ������ᴿ����ܽ�����¶ȱ仯�������ؽᾧ�����룬��A��ȷ��
B����ͬ�Ļ�ѧ�������������Ƶ�ʲ�ͬ���ں������ͼ�ϴ��ڲ�ͬ��λ�ã����Ժ������ͼ��ȷ���л�������еĻ�ѧ��������ţ���B����
C������ȼ�շ����ܽ��л���ֽ�Ϊ��������������ⶨ��ͨ������������������ɸ��л���Ԫ��ԭ�ӵ�����������Ȼ���������л����������Ԫ��ԭ����������ȣ���ȷ��ʵ��ʽ����C����
D���������ܼ�¼�������ӡ���Ƭ���ӵ��������������ͼ����ֵ���ļ��Ǹ÷��ӵ���Է�����������D����ѡA��
B����ͬ�Ļ�ѧ�������������Ƶ�ʲ�ͬ���ں������ͼ�ϴ��ڲ�ͬ��λ�ã����Ժ������ͼ��ȷ���л�������еĻ�ѧ��������ţ���B����
C������ȼ�շ����ܽ��л���ֽ�Ϊ��������������ⶨ��ͨ������������������ɸ��л���Ԫ��ԭ�ӵ�����������Ȼ���������л����������Ԫ��ԭ����������ȣ���ȷ��ʵ��ʽ����C����
D���������ܼ�¼�������ӡ���Ƭ���ӵ��������������ͼ����ֵ���ļ��Ǹ÷��ӵ���Է�����������D����ѡA��
���������⿼�����о��л���ķ������ѶȲ�����ȷ�⼸�������������ǽⱾ��Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
������ʵ����Ӧ�ķ���ʽ����ȷ���ǣ�������
| A����Na2S��ȥ��ˮ�е�Hg2+��Hg2++S2-�THgS�� |
| B��������Һ����ϴ���ۣ�CO32-+2H2O?H2CO3+2OH- |
| C���������Ȼ���ʱ�������̣�NH3+HCl=NH4Cl |
| D�����ҽ���Һ�м������ᡢ˫��ˮ��2I-+2H++H2O2�TI2+2H2O |
ClO2��һ������ɱ��Ч�ʸߡ�������ȾС��ˮ��������ʵ���ҿ�ͨ�����·�Ӧ�Ƶ�ClO2��2KClO3+H2C2O4+H2SO4
2ClO2��+K2SO4+2CO2��+2H2O������˵����ȷ���ǣ�������
| ||
| A��1mol KClO3�μӷ�Ӧ��������������Ϊ44.8L |
| B��H2C2O4�ڷ�Ӧ�б���ԭ |
| C��ClO2�������������������ˮ���ھ�ˮʱ�������� |
| D������1mol ClO2����Ӧ��ת�Ƶĵ�����1 mol |
��NAΪ����٤��������ֵ������˵����ȷ���ǣ�������
| A����50g��������Ϊ46%���Ҵ�ˮ��Һ�У�������ԭ����Ϊ3NA |
| B��Na2S��Na2O2 ��������15.6g�У����е���������Ϊ0.3NA |
| C��104g����ϩ�к���8NA��̼�����4NA��̼̼˫�� |
| D��1.6g�������ͳ�����ɵĻ�����к�����ԭ�ӵ���ĿΪ0.1NA |
����������Ԫ��X��Y��Z��M��W��Ԫ�����ڱ��е����λ�����������Z�ĺ˵������Y ������������˵����ȷ���ǣ�������
| X | Y | |
| W | M | Z |
| A��Mλ��Ԫ�����ڱ��е������ڢ�A�� |
| B��ԭ�Ӱ뾶��Z��M��X |
| C����̬�⻯������ȶ��ԣ�Y��X��W |
| D��W�������������NaOH��Ӧ��Ҳ����HF��Ӧ���������������� |
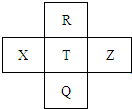 Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������ֻ��X�Ƕ����ڵĽ���Ԫ�أ������ж���ȷ���ǣ�������
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ�����±���ʾ������ֻ��X�Ƕ����ڵĽ���Ԫ�أ������ж���ȷ���ǣ�������| A��X�����������������ǿ�� |
| B���ǽ����ԣ�T��R |
| C����̬�⻯���ȶ��ԣ�T��Z |
| D��R��Q��ԭ�Ӻ�����������16 |
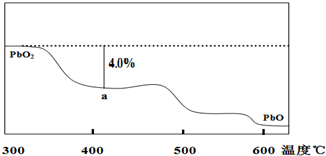 Ǧ���仯������������ء������豸��X���߷������ϣ���֪Pb�Ļ��ϼ�ֻ��0��+2��+4�ش��������⣺
Ǧ���仯������������ء������豸��X���߷������ϣ���֪Pb�Ļ��ϼ�ֻ��0��+2��+4�ش��������⣺ ������COCl2����������N2H4����H2O2������Ҫ�Ĺ�ҵ��ֵ��
������COCl2����������N2H4����H2O2������Ҫ�Ĺ�ҵ��ֵ��