��Ŀ����
5���� Cu��N��B��Ni ��Ԫ����ɵ����Ͳ������Ź㷺��;����1����ѧ��ͨ�� X�����Ʋ���мȺ�����λ�����ֺ����������ṹʾ��ͼ�ɼ�ʾ��ͼ��������λ����������������߱�ʾ��
��д����̬Cuԭ�ӵĺ�������Ų�ʽ1s22s22p63s23p63d104s1��[Ar]3d104s1��
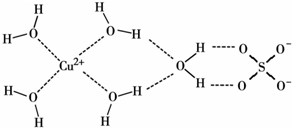
��д������������ˮ��ͭ���ӵĽṹ��ʽ�����뽫��λ����ʾ������
 ��
����ˮ���Ӽ��������������о�������ʵ˵�������ˮ�����ʵ�Ӱ��ˮ���ۡ��е�ϸߣ����ʱ�ܶȼ�С��
��2����ѧ�������Ѻϳ�һЩ�����ɵ���ɵ�������N5+��N3-�ȣ���N5+������ÿ����ԭ�Ӿ�����8���ӽṹ�������й�N5+�Ʋ���ȷ����c��
a��N5+������24�����ӡ�����������
b��N5+�����д�������δ�ɼ��ĵ��Ӷ�
c��N5+���������������������
��3��������A��H3BNH3����һ��DZ�ڵĴ�����ϣ���������Ԫ��״�����HB=NH��3ͨ��3CH4+2��HB=NH��3+6H2O��3CO2+6H3BNH3�Ƶã�
����������ѧ����ʽ�йص���������ȷ����a������ţ���
a����Ӧǰ��̼ԭ�ӵĹ���ӻ����Ͳ���
b��CH4��H2O��CO2���ӿռ乹�ͷֱ��ǣ����������Ρ�V�Ρ�ֱ����
c����һ�����ܣ�N��O��C��B
��1����HB=NH��3��������12���Ҽ���
��4�����������У�����������״����״�ȶ��ֽṹ��ʽ��ͼ��a����һ����״�ṹ�Ķ�������������������ӷ���Ϊ[BO2]nn-����BO2-����
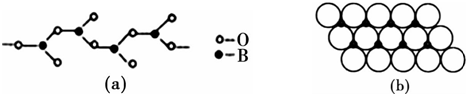
��5����һ���¶��£�NiO��������Է��ط�ɢ���γɡ������Ӳ㡱����ͼ��b����������Ϊ�����������ܵ������У�������������У���ʽ������ÿƽ��������Ϸ�ɢ�ĸþ��������Ϊ1.83��10-3g�������ӵİ뾶Ϊ1.40��10-10 m��$\sqrt{3}$��1.732����
���� ��1����ͭ��29��Ԫ�أ���ԭ�Ӻ�����29�����ӣ����ݹ���ԭ����д���̬ԭ�Ӻ�������Ų�ʽ��
��CuSO4•5H2O��ͭ���Ӻ��пչ����ˮ���Ӻ��й¶Ե��Ӷԣ�ͭ������ˮ����֮���γ���λ����ͭ����������Ϊ4����5������ͼƬ֪��ÿ����������ռ�����=1.40��10-10m��1.40��10-10m��sin60�㣬ÿ��������������=$\frac{74.7}{{N}_{A}}$g��ÿ������������������ÿƽ�����е���������������ÿƽ�����е��������������������һ����Ӽ�������ǿ��Ӱ�����ʵ��������ʣ�
��2��a��1����ԭ���к���7�����ӣ���1��N5�����к���35�����ӣ�N5+����N5����ʧȥ1�����ӵõ��ģ���1��N5+��������34�����ӣ���a����
b��N5+������ÿ����ԭ�Ӿ�����8���ӽṹ��ÿ��Nԭ���γ�3�����õ��Ӷԣ���ʣһ��δ�ɼ����ӣ�����N5+�����д������δ�ɼ��ĵ��Ӷԣ���b����
c��N5+���ӵĽṹΪ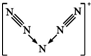 ����N5+�������д�������������������cȷ��
����N5+�������д�������������������cȷ��
��3�����ɷ���ʽ��֪��̼Ԫ����CH4��ΪCO2��̼ԭ���ӻ�������sp3ת��Ϊsp��
�ڸ��ݣ�HB=NH��3�ṹ�뱽���ƽ��з�����
��4����ͼ�ɿ�����ÿ��BO32-��Ԫ������һ��B����һ��O��ȫ���������Ԫ��ʣ���2��O�ֱ�Ϊ2��BO32-��Ԫ���ã�
��5������ͼƬ��������ÿ����������ռ�����=1.40��10-10m��1.40��10-10m��sin60�㣬ÿ��������������=$\frac{74.7}{{N}_{A}}$g��ÿ������������������ÿƽ�����е���������������ÿƽ�����е�������������
��� �⣺��1����ͭ��29��Ԫ�أ���ԭ�Ӻ�����29�����ӣ����ݹ���ԭ��֪������������ԭ����д�����Ų�ʽ�����̬ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
��CuSO4•5H2O��ͭ���Ӻ��пչ����ˮ���Ӻ��й¶Ե��Ӷԣ�ͭ������ˮ����֮���γ���λ����ͭ����������Ϊ4��ˮ��ͭ���ӵĽṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
�� Cu��N��B��Ni ��Ԫ����ɵ����Ͳ������Ź㷺��;��
�������һ����Ӽ�������ǿ��Ӱ�����ʵ��������ʣ������һ����Ӽ�������ǿ������ˮ���ۡ��е�ϸߣ�����������з����ԣ����ʱ��������࣬��������ܶȼ�С���ʴ�Ϊ��ˮ���ۡ��е�ϸߣ����ʱ�ܶȼ�С��
��2��a��1����ԭ���к���7�����ӣ���1��N5�����к���35�����ӣ�N5+����N5����ʧȥ1�����ӵõ��ģ���1��N5+��������34�����ӣ���a����
b��N5+������ÿ����ԭ�Ӿ�����8���ӽṹ��ÿ��Nԭ���γ�3�����õ��Ӷԣ���ʣһ��δ�ɼ����ӣ�����N5+�����д������δ�ɼ��ĵ��Ӷԣ���b��
c��N5+���ӵĽṹΪ ����N5+�������д�������������������cȷ���ʴ�ѡa��
����N5+�������д�������������������cȷ���ʴ�ѡa��
��3����3CH4+2��HB=NH��3+6H2O��3CO2+6H3BNH3��
a��CH4��ΪCO2��̼ԭ���ӻ�������sp3ת��Ϊsp����Ӧǰ��̼ԭ�ӵĹ���ӻ������Ѿ��ı䣬��a��
b��H4�����м۲���Ӷ�=�� �����Ӷ�+����ԭ���ϵŵ��Ӷ�=4+$\frac{1}{2}$����4-4��1��=4���Ҳ����µ��Ӷԣ�������ռ乹�����������壬H2O�м۲���ӶԸ���=2+$\frac{1}{2}$����6-2��1��=4���Һ���2���µ��Ӷԣ�����H2O��VSEPRģ��Ϊ�����壬���ӿռ乹��ΪV�ͣ�CO2�����м۲���Ӷ�=�� �����Ӷ�+����ԭ���ϵŵ��Ӷ�=2+$\frac{1}{2}$����4-2��2��=2�����Զ�����̼��ֱ���ͽṹ����bȷ��
c��һ����Ԫ���У�Ԫ�صĵ�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�صĵ�һ�����ܴ�������Ԫ�أ��⼸��Ԫ�ض��ǵڶ�����Ԫ�أ����ǵ��������ֱ��ǣ���IIIA�塢��IVA�塢��VA�塢��VIA�壬�������ǵĵ�һ�����ܴ�С˳����I1��N����I1��O����I1��C����I1��B������cȷ���ʴ�Ϊ��c��
��1����HB=NH��3��������ԭ���뵪ԭ�Ӽ��ԦҼ���ϣ���ʣ���p����γ�һ�������м���B-H����3����N-H����3����B-N�ЦҼ���6������һ��12�����ʴ�Ϊ��12��
��4��ͼ��a����һ����״�ṹ�Ķ����������ͼ�ɿ�����ÿ��BO32-��Ԫ������һ��B����һ��O��ȫ���������Ԫ��ʣ���2��O�ֱ�Ϊ2��BO32-��Ԫ���ã�����B��O=1����1+2��$\frac{1}{2}$��=1��2���ʴ�ΪΪ��[BO2]nn-����BO2-����
��5������ͼƬ֪��ÿ����������ռ�����=1.40��10-10m��1.40��10-10m��sin60�㣬1ƽ�����е�����������=$\frac{1}{1.40��10-10m��1.40��10-10m��sin60��}$��ÿ��������������=$\frac{74.7}{{N}_{A}}$g������ÿƽ�����е�����������=$\frac{74.7}{{N}_{A}}$��$\frac{1}{1.40��10-10m��1.40��10-10m��sin60��}$=1.83��10-3g���ʴ�Ϊ��1.83��10-3��
���� ���⿼����ԭ�Ӻ�������Ų�ʽ����д����λ���ı�ʾ������ԭ���ӻ���ʽ���жϵ�֪ʶ�㣬���ݹ���ԭ������λ���ĸ���۲���ӶԻ���������������ɣ��Ѷ��еȣ�

 Happy holiday���ּ��������ҵ�㶫���������ϵ�д�
Happy holiday���ּ��������ҵ�㶫���������ϵ�д� ���������������Բ��������ϵ�д�
���������������Բ��������ϵ�д�| A�� | ������֮��1��1 | B�� | �ܶ�֮��4��11 | C�� | �ܶ�֮��11��16 | D�� | ���֮��11��16 |
| A�� | Na2O2����ǿ�������ԣ������������� | |
| B�� | Na2CO3��Һ�ʼ��ԣ���������;߱�������� | |
| C�� | ������۵�ߡ�Ӳ�ȴ����������뵼����� | |
| D�� | ͭ�Ľ��������Ա����IJ���ں��������װ����ͭ���Լ����丯ʴ |
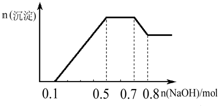
| A�� | ��Һ�е�������ֻ��H+��Mg2+��Al3+ | |
| B�� | ��Һ��һ������CO32-��һ������SO42-��NO3- | |
| C�� | ��Һ��n��NH4+��=0.2 mol | |
| D�� | �������ӵ����ʵ���֮��n��H+����n��Al3+����n��Mg2+��=1��1��1 |
| A�� | 2-�ȱ�ϩ | B�� | 1��2-������ϩ | C�� | 2-��-2-��ϩ | D�� | 1-��ϩ |
| A�� | ��ʽ�ε���Һһ���Լ��� | |
| B�� | ֻҪ���������ʵ���Ũ�Ⱥ�����ֱ���ȣ����Ƿ�Ӧ�����Һ�����Ե� | |
| C�� | ��ˮ������ʱ��Ϊˮ��c��H+��=c��OH���� | |
| D�� | ̼����Һ�������ӵ����ʵ���Ũ����̼����������ʵ���Ũ�ȵ�2�� |
 ��2��ˮ
��2��ˮ ��3������
��3������ ��4���Ȼ���
��4���Ȼ���
 ��6������H��H��7���Ȼ�þ
��6������H��H��7���Ȼ�þ ��8��������̼
��8��������̼ ��
��