��Ŀ����
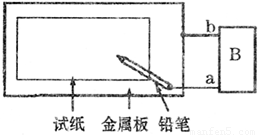
ȡһ���ñ��͵�NaCl��Һ��ʪ��ʯ����ֽ��ƽ����һ��������ϣ�����ͼ��ʾ�ķ������ӵ�·����1����������Ϊһ��Ƭ��B��Ϊһֱ����Դ����ͨ��Դ��
������ֽΪ��ɫʯ����ֽ����Ǧ������ֽ��д�֣�������ɫ�ּ�����a�缫Ϊ��Դ��______�������������������Ǧ��о����ֽ�Ӵ��ĵ缫��ӦΪ______��
������ֽΪ��ɫʯ����ֽ����Ǧ��о��������ֽ��һ��ʱ��Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ����a�缫Ϊ��Դ��______�������������������Ǧ��о����ֽ�Ӵ����ĵ缫��ӦΪ______��
��2������ֽΪ��ɫʯ�� ��ֽ��������ΪһпƬ��B��Ϊһ���������ƣ���ͨ��·����Ǧ��о��������ֽ��һ��ʱ�䣬���ֵ�����ָ�뷢��ƫת����Ӵ�����______�����ס���������������ɫ������Ǧ��о����ֽ�Ӵ����ĵ缫��ӦΪ______��
���𰸡���������1��������ֽΪ��ɫʯ����ֽ����Ǧ������ֽ��д�֣�������ɫ�ּ���˵���������������ɣ���̼�缫�������������������ӷŵ�ͬʱ�����������ƣ�����������ԭ��ص缫�Ǹ��������������������ӵõ��ӷ�����ԭ��Ӧ��
������ֽΪ��ɫʯ����ֽ����Ǧ��о��������ֽ��һ��ʱ��Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��˵��ʯī�ǵ��������������������ӷŵ�����������ԭ����������ӵ���������
��2������ֽΪ��ɫʯ����ֽ��������ΪһпƬ��B��Ϊһ���������ƣ���ͨ��·�����װ�ù���ԭ��أ�п��������ʯī�������������������õ��Ӻ�ˮ��Ӧ�������������ӣ�
����⣺��1��������ֽΪ��ɫʯ����ֽ����Ǧ������ֽ��д�֣�������ɫ�ּ���˵���������������ɣ���ʯī�缫����������a�ǵ�Դ���������������������ӵõ��ӷ�����ԭ��Ӧ���缫��ӦʽΪ��2H++2e-=H2������2H2O+2 e-=H2��+2OH-����
�ʴ�Ϊ��������2H++2e-=H2������2H2O+2 e-=H2��+2OH-����
������ֽΪ��ɫʯ����ֽ����Ǧ��о��������ֽ��һ��ʱ��Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��˵��ʯī�ǵ���������ԭ����������ӵ�������������a�缫�ǵ�Դ�����������������ӷŵ������������缫��ӦʽΪ��2Cl-2e-=Cl2�����ʴ�Ϊ��������2Cl-2e-=Cl2����
��2������ֽΪ��ɫʯ����ֽ��������ΪһпƬ��B��Ϊһ���������ƣ���ͨ��·�����װ�ù���ԭ��أ�п��������ʯī�������������������õ��Ӻ�ˮ��Ӧ�������������ӣ��缫��ӦʽΪ��O2+2H2O+4e-=4OH-��ʪ��ĺ�ɫʯ����ֽ�������ɫ��
�ʴ�Ϊ��������O2+2H2O+4e-=4OH-��
���������⿼��ԭ��غ͵���ԭ������ȷ��ֽ��ɫ�仯����Һ������ǹ�ϵ�ǽⱾ��ؼ����ѵ��ǵ缫��Ӧʽ����д���Ѷ��еȣ�
������ֽΪ��ɫʯ����ֽ����Ǧ��о��������ֽ��һ��ʱ��Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��˵��ʯī�ǵ��������������������ӷŵ�����������ԭ����������ӵ���������
��2������ֽΪ��ɫʯ����ֽ��������ΪһпƬ��B��Ϊһ���������ƣ���ͨ��·�����װ�ù���ԭ��أ�п��������ʯī�������������������õ��Ӻ�ˮ��Ӧ�������������ӣ�
����⣺��1��������ֽΪ��ɫʯ����ֽ����Ǧ������ֽ��д�֣�������ɫ�ּ���˵���������������ɣ���ʯī�缫����������a�ǵ�Դ���������������������ӵõ��ӷ�����ԭ��Ӧ���缫��ӦʽΪ��2H++2e-=H2������2H2O+2 e-=H2��+2OH-����
�ʴ�Ϊ��������2H++2e-=H2������2H2O+2 e-=H2��+2OH-����
������ֽΪ��ɫʯ����ֽ����Ǧ��о��������ֽ��һ��ʱ��Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��˵��ʯī�ǵ���������ԭ����������ӵ�������������a�缫�ǵ�Դ�����������������ӷŵ������������缫��ӦʽΪ��2Cl-2e-=Cl2�����ʴ�Ϊ��������2Cl-2e-=Cl2����
��2������ֽΪ��ɫʯ����ֽ��������ΪһпƬ��B��Ϊһ���������ƣ���ͨ��·�����װ�ù���ԭ��أ�п��������ʯī�������������������õ��Ӻ�ˮ��Ӧ�������������ӣ��缫��ӦʽΪ��O2+2H2O+4e-=4OH-��ʪ��ĺ�ɫʯ����ֽ�������ɫ��
�ʴ�Ϊ��������O2+2H2O+4e-=4OH-��
���������⿼��ԭ��غ͵���ԭ������ȷ��ֽ��ɫ�仯����Һ������ǹ�ϵ�ǽⱾ��ؼ����ѵ��ǵ缫��Ӧʽ����д���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
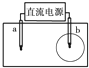 ��ͼ��ʾ��ȡһ���ñ��͵�NaCl��Һ��ʪ��pH��ֽ������Ǧ��о���缫����ֱͨ����Դ��һ��ʱ�����a�缫����ֽ�Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��������˵��������ǣ�������
��ͼ��ʾ��ȡһ���ñ��͵�NaCl��Һ��ʪ��pH��ֽ������Ǧ��о���缫����ֱͨ����Դ��һ��ʱ�����a�缫����ֽ�Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��������˵��������ǣ�������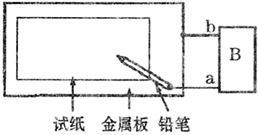 ȡһ���ñ��͵�NaCl��Һ��ʪ��ʯ����ֽ��ƽ����һ��������ϣ�����ͼ��ʾ�ķ������ӵ�·��
ȡһ���ñ��͵�NaCl��Һ��ʪ��ʯ����ֽ��ƽ����һ��������ϣ�����ͼ��ʾ�ķ������ӵ�·�� ��08���Ϻ�����ȡһ���ñ��͵�NaCl��Һ��ʪ��pH��ֽ������Ǧ��о���缫����ֱͨ����Դ��һ��ʱ�����a�缫����ֽ�Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��������˵��������� �� ��
��08���Ϻ�����ȡһ���ñ��͵�NaCl��Һ��ʪ��pH��ֽ������Ǧ��о���缫����ֱͨ����Դ��һ��ʱ�����a�缫����ֽ�Ӵ�������һ��˫ɫͬ��Բ����ȦΪ��ɫ����Ȧ��dz��ɫ��������˵��������� �� �� 