��Ŀ����
��FeCl3��AlCl3�Ļ����Һ100ml�������м��������NaOH��Һ����ַ�Ӧ���ˣ���������ɳ���
10.7g���ڼ���NaOH��Һ�Ĺ����У����� ���������������NaOH��Һ���������ͼ��ʾ��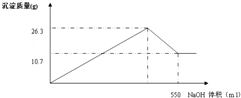
��1��д�������Һ�м������NaOH��Һ�����ӷ���ʽ �� ��
��2����10.7g�����������գ����õ����� �ˣ�
��3��FeCl3�����ʵ�����Ũ���� mol/L��
��4������AlCl3�����ʵ�����Ũ�� mol/L
��5����NaOH��Һ�����ʵ���Ũ�ȣ�����ʾ����д���ڣ�5��С��Ľ�����̣�
10.7g���ڼ���NaOH��Һ�Ĺ����У����� ���������������NaOH��Һ���������ͼ��ʾ��

��1��д�������Һ�м������NaOH��Һ�����ӷ���ʽ
��2����10.7g�����������գ����õ�����
��3��FeCl3�����ʵ�����Ũ����
��4������AlCl3�����ʵ�����Ũ��
��5����NaOH��Һ�����ʵ���Ũ�ȣ�����ʾ����д���ڣ�5��С��Ľ�����̣�
���㣺�йػ���ﷴӦ�ļ���
ר�⣺������
��������1���Ȼ������������Ʒ�Ӧ�������������������Ȼ��ƣ��Ȼ��������������������Һ��Ӧ����ƫ��������ˮ��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3������n=
���������������ʵ���������FeԪ���غ����n��Fe2O3�����ٸ���m=nM����m��Fe2O3����
��3������FeԪ���غ����n��FeCl3��=n[Fe��OH��3]���ٸ���c=
����c��FeCl3����
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ�������������������������n=
�����������������ʵ���������AlԪ���غ����n��AlCl3�����ٸ���c=
����c��AlCl3����
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��������AlԪ���غ�n��NaAlO2��=n��AlCl3���������������غ�n��NaOH��=n��NaCl��+n��NaAlO2�����ٸ���c=
����c��NaOH����
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3������n=
| m |
| M |
��3������FeԪ���غ����n��FeCl3��=n[Fe��OH��3]���ٸ���c=
| n |
| V |
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ�������������������������n=
| m |
| M |
| n |
| V |
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��������AlԪ���غ�n��NaAlO2��=n��AlCl3���������������غ�n��NaOH��=n��NaCl��+n��NaAlO2�����ٸ���c=
| n |
| V |
���
�⣺��1���Ȼ������������Ʒ�Ӧ�������������������Ȼ��ƣ���Ӧ���ӷ���ʽΪ��Fe3++3OH-=Fe��OH��3�����Ȼ��������������������Һ��Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪAl3++4OH-=AlO2-+2H2O��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3��n[Fe��OH��3]=
=0.1mol������FeԪ���غ�n��Fe2O3��=
=0.05mol����m��Fe2O3��=0.05mol��160g/mol=8g���ʴ�Ϊ��8��
��3������FeԪ���غ�n��FeCl3��=n[Fe��OH��3]=0.1mol����c��FeCl3��=
=1mol/L���ʴ�Ϊ��1��
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ���������������Ϊ26.3g-10.7g=15.6g���������������ʵ���Ϊ
=0.2mol������AlԪ���غ�n��AlCl3��=0.2mol��c��AlCl3��=
=2mol/L���ʴ�Ϊ��2��
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��=3��0.1mol+3��0.2mol=0.9mol������AlԪ���غ�n��NaAlO2��=n��AlCl3��=0.2mol�������������غ�n��NaOH��=n��NaCl��+n��NaAlO2��=0.9mol+0.2mol=1.1mol����c��NaOH��=
=2mol/L��
������������Һ�����ʵ���Ũ��Ϊ2mol/L��
�ʴ�Ϊ��Fe3++3OH-=Fe��OH��3����Al3++4OH-=AlO2-+2H2O��
��2�����ճ���10.7gΪFe��OH��3�����ȷֽ�����Fe2O3��n[Fe��OH��3]=
| 10.7g |
| 107g/mol |
| 0.1mol |
| 2 |
��3������FeԪ���غ�n��FeCl3��=n[Fe��OH��3]=0.1mol����c��FeCl3��=
| 0.1mol |
| 0.1L |
��4���������ʱΪ26.3g��Ϊ������������������������֮�ͣ���������������Ϊ26.3g-10.7g=15.6g���������������ʵ���Ϊ
| 15.6g |
| 78g/mol |
| 0.2mol |
| 0.1L |
��5������550mL NaOH��Һ��ʱ����Һ������ΪNaCl��NaAlO2�������������غ�n��NaCl��=3n��FeCl3��+3n��AlCl3��=3��0.1mol+3��0.2mol=0.9mol������AlԪ���غ�n��NaAlO2��=n��AlCl3��=0.2mol�������������غ�n��NaOH��=n��NaCl��+n��NaAlO2��=0.9mol+0.2mol=1.1mol����c��NaOH��=
| 1.1mol |
| 0.55L |
������������Һ�����ʵ���Ũ��Ϊ2mol/L��
�����������Ի�ѧ��Ӧͼ����ʽ������������㣬��ȷ���η����ķ�Ӧ�ǹؼ���ע�������غ㷨���н���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
�������ӷ���ʽ����ȷ���ǣ�������
| A����һС�������Ʒ���ˮ�У�2Na+2H2O=2Na++2OH-+H2�� |
| B��������ͨ������������Һ�У�Cl2+2OH-=C1-+C1O-+H2O |
| C����CuSO4��Һ�м�������Ba��OH��2��Һ��Ba2++SO42-=BaSO4�� |
| D������ͨ��ˮ�У�Cl2+H2O?H++HClO+Cl- |
ʵ������Ҫ����2mol/L��NaCl��Һ480mL������ʱӦѡ�õ�����ƿ�Ĺ���Ӧ��ȡ��NaCl�ֱ��ǣ�������
| A��480 mL��117g |
| B��500mL��58.5g |
| C��480 mL��58.5 g |
| D��500mL��117g |
�����£���һԪ��HA����Һ��KOH��Һ�������ϣ���������仯����ʵ���������±��������жϲ���ȷ���ǣ�������
| ʵ���� | ��ʼŨ��/��mol?L-1�� | ��Ӧ����Һ��pH | |
| c��HA�� | c��KOH�� | ||
| �� | 0.1 | 0.1 | 9 |
| �� | x | 0.2 | 7 |
| A��ʵ��ٷ�Ӧ�����Һ�У�c��K+����c��A-����c��OH-����c��H+�� |
| B��ʵ��ٷ�Ӧ�����Һ�У�c��OH-��=c��H+��+c��HA�� |
| C��ʵ��ڷ�Ӧ�����Һ�У�c��A-��+c��HA����0.2 mol/L |
| D��ʵ��ڷ�Ӧ�����Һ�У�c��K+��=c��A-����c��OH-��=c��H+�� |
��������ʵ���е�����˵��������ǣ�������
| A��Ϊ�˸��õؿ��������¶ȣ�Ӧ���¶ȼ����²���ӽ�Һ�洦 |
| B��Ϊ�˷�ֹ���У��ڶ�Һ����������������ʱӦ�ü����ʯ |
| C��Ϊ�˱�֤����Ч��������ˮӦ�������ܵ��¿������Ͽ����� |
| D��ţ�ǹܵ�������ʹ�������Һ�������䷽��������ƿ |