��Ŀ����
17�����ᣨ��ѧʽ��HOCN����һ���лӷ��Ժ�ʴ�Ե�Һ�壬��ˮ�����̷������·�Ӧ�γ���X������̼泥���HOCN+2H2O��X����1����������Ӧ���漰�ĸ�Ԫ���У��뾶����Ԫ��ԭ�Ӻ�����ӹ�ռ��4�������
��2����˵����Ԫ�صķǽ����Ա�̼Ԫ��ǿ����ʵ��ac��ѡ���ţ���
a�����õ��Ӷ�ƫ��H-N��H-C b�������ԣ�NO2��CO2
c�����ԣ�HNO3��H2CO3 d���е㣺NH3��CH4
��3���ݲⶨ�����������ֽṹ��һ�ַ����ں�����������Ϊ�����ᣬ��һ�ַ����ڲ�����������Ϊ�����ᣬ�����ֽṹ������ԭ���������Ѵﵽ�ȶ��ṹ��������Ҳ������״�ṹ����ֱ�д���������������Ľṹʽ��H-O-C��N��H-N=C=O��
��4�������ǹ�ҵ��ұ��Crʱ���漰���ķ�Ӧ��
4 CrO42-+6 S+7H2O��4Cr��OH��3��+3 S2O32-+2OH-
��1���뽫����ʽ������������ƽ��
��2��������Ӧ�У���ת����3mol���ӣ��õ��Ļ�ԭ������1mol��
��5����������Ni��λ�����ڱ���ͬһ����ͬһ�壬�������ڵ������ڵ�VIII�壮
��6��������CO�е��¼��ȣ�������ɫ�ӷ���Һ̬Ni��CO��4���������幹�ͣ�150��ʱ��Ni��CO��4�ֽ�ΪNi��CO��Ni��CO��4�Ƿ��Ӿ��壬Ni��CO��4����������bc������ţ���
a��ˮ b�����Ȼ�̼ c���� d����������Һ��
���� ��1�����ݹ���ԭ��д�����������Ų�ʽ����ȷ��ռ�еĹ����
��2���ǽ�����ǿ�����жϷ����У����������ϵ����׳̶ȡ����⻯����ȶ��ԡ�����ۺ���������ԡ��������������ȣ�
��3��̼Ϊ����λ����Ϊ����λ����Ϊ����λ�������֪����ȷ����ṹ��ʽ��
��4���ٸ÷�Ӧ��SԪ�ػ��ϼ���0�۱�Ϊ+2�ۡ�CrԪ�ػ��ϼ���+6�۱�Ϊ+3�ۣ����ݵ�ʧ���������ƽ����ʽ��
�ڸ÷�Ӧ�л�ԭ������Cr��OH��3�����ݻ�ԭ�����ת�Ƶ���֮��Ĺ�ϵʽ���㣻
��5���������ڵ������ڵ�VIII�壻
��6�����Ӿ�����۷е�ϵͣ����ݸ����ʵ��۷е�ȷ���������ͣ�������������ԭ��ȷ�����ܽ��ԣ�
��� �⣺��1��̼ԭ�ӵĺ�������Ų�ʽΪ1s22s22p2����ԭ�Ӻ�����ӹ�ռ��һ��1s��һ��2s������2p��4�������
�ʴ�Ϊ��4��
��2���ǽ�����ǿ�����жϷ����У����������ϵ����׳̶ȡ����⻯����ȶ��ԡ�����ۺ���������ԡ��������������ȣ�
�ʴ�Ϊ��ac��
��3��̼Ϊ����λ����Ϊ����λ����Ϊ����λ������������ں�������������������ڲ��������������ֽṹ������ԭ���������Ѵﵽ�ȶ��ṹ��������Ҳ������״�ṹ�������������������Ľṹ��ʽ�ֱ�Ϊ��H-O-C��N��H-N=C=O��
�ʴ�Ϊ��H-O-C��N��H-N=C=O��
��4���ٸ÷�Ӧ��SԪ�ػ��ϼ���0�۱�Ϊ+2�ۡ�CrԪ�ػ��ϼ���+6�۱�Ϊ+3�ۣ�����Ԫ���غ�֪���������������� OH-�����ݵ�ʧ���������ƽ����ʽΪ4CrO42-+6 S+7 H2O=4 Cr��OH��3��+3S2O32-+2 OH-��
�ʴ�Ϊ��4��6��7��4��3��2 OH-��
�ڸ÷�Ӧ�л�ԭ������Cr��OH��3�����ݻ�ԭ�����ת�Ƶ���֮��Ĺ�ϵʽ�ã���ת����3mol���ӣ��õ��Ļ�ԭ��������ʵ���=$\frac{3mol}{3}$=1mol��
�ʴ�Ϊ��1��
��5���������ڵ������ڵ�VIII�壻
�ʴ�Ϊ�����ģ��ڢ���
��6�����ݸ����ʵ��۷е�֪�����������ڷ��Ӿ��壬�����ʵĽṹΪ��������ṹ��������������غϣ�Ϊ�Ǽ��Է��ӣ�������������ԭ��֪���Ǽ��Է��ӵ����������ڷǼ��Է��ӵ��ܼ����������Ȼ�̼���ǷǼ��Է��ӣ����Ը����������ڱ������Ȼ�̼��
�ʴ�Ϊ�����Ӿ��壻bc��
���� ���⿼����ۺϣ��漰�����Ų����ǽ�����ǿ���жϡ���������ԭ����Ԫ�����ڱ��ṹ��������ԭ��Ӧ��ԭ�ӽṹ��֪ʶ�㣬��Щ֪ʶ�㶼�ǿ����ȵ㣬���ݻ���ԭ�������ʻ�ԭ�ӽṹ���Ԫ�������������������Ŀ�Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�������ͬ��Ԫ�ص����ʣ���101.3kPaʱ�ⶨ���ǵķе�(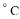 )���±���ʾ��
)���±���ʾ��
�� | He-268.8 | (a)-249.5 | Ar-185.8 | Kr151.7 |
�� | F2-187.0 | Cl2-33.6 | (b)58.7 | I2184.0 |
�� | (c)19.4 | HCl-84.0 | HBr-67.0 | HI-35.3 |
�� | H2O100.0 | H2S-60.0 | (d)-42.0 | H2Te-1.8 |
�� | CH4-161.0 | SiH4-112.0 | GeH4-90.0 | (e)-52.0 |
��Ӧ�������ݣ����и�������ȷ����
A��a��b��c�Ļ�ѧʽ�ֱ�ΪNe2��Br2��HF
B���ڢ����и���������ȶ���˳��Ϊ��H2O��H2S��H2Se��H2Te
C���ڢ������ʾ��������ԣ��ڢ������ʶ�Ӧˮ��Һ����ǿ��
D���ϱ���ͬ�����ʶ�Ӧ���⻯��ķе㣬������Է������������������
| A�� | CO2 | B�� | N2 | C�� | SO2 | D�� | CO |
| A�� | H2��g��+Cl2��g��=2HCl��g������H=-a $\frac{1}{2}$H2��g��+$\frac{1}{2}$Cl2��g��=HCl��g������H=-b | |
| B�� | C��s��+$\frac{1}{2}$O2��g��=CO ��g������H=-a C��s��+O2��g��=CO2 ��g������H=-b | |
| C�� | 2H2��g��+O2��g��=2H2O��l������H=-a 2H2��g��+O2��g��=2H2O��g������H=-b | |
| D�� | S��g��+O2��g��=SO2 ��g������H=-a S��s��+O2��g��=SO2 ��g������H=-b |
| A�� | �����ʵ�����Ϊ 2��2��3-����-3-��ϩ | |
| B�� | ��������� 4 ��̼ԭ�ӹ�ƽ�� | |
| C�� | ������ת��Ϊ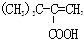 �Ĺ����������ξ����ӳɡ�ȡ������������ȥ��Ӧ �Ĺ����������ξ����ӳɡ�ȡ������������ȥ��Ӧ | |
| D�� | �������������ӳɺ�IJ��������������ԭ�ӵ���������ͬ |
 ��ͼ����ˮ����������˹ƥ�֣�Aspirin���������ȡ���ʹ���ã������������˷硢��ð��ͷʹ����ʹ����ʪ�Թؽ�ʹ�������������ڷ�����������Ѫ�ܲ���������������������˲��˷��ã��Դ˻��� �ﲻ��ȷ��˵���ǣ�������
��ͼ����ˮ����������˹ƥ�֣�Aspirin���������ȡ���ʹ���ã������������˷硢��ð��ͷʹ����ʹ����ʪ�Թؽ�ʹ�������������ڷ�����������Ѫ�ܲ���������������������˲��˷��ã��Դ˻��� �ﲻ��ȷ��˵���ǣ�������| A�� | ��˹ƥ�ֵĻ�ѧʽΪC9H8O4 | |
| B�� | �û�����������ԣ����� NaOH �� NaHCO3��Ӧ | |
| C�� | 1 mol �û������������� NaOH��Һ��Ӧ������ NaOH�������ʵ���Ϊ2 mol | |
| D�� | ������ Aspirin ������ FeCl3��Һ��Ӧ��������ˮ�������ʹFeCl3��Һ��ɫ |
| A�� | ��������ֱ�ӽӴ�ҩƷ | B�� | ���þƾ��ƶ��ձ�ֱ�Ӽ��� | ||
| C�� | �ñǿ״յ�������ȥ���������ζ | D�� | ��һ��ʵ������ƾ���ζ�� |
| A�� | ������Ϊ53��������Ϊ78�ĵ�ԭ�ӣ�${\;}_{53}^{131}$I | |
| B�� | N2�ĵ���ʽ�� | |
| C�� | S2-�Ľṹʾ��ͼ��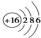 | |
| D�� | ��ȩ�Ľṹ��ʽ��CH3CH2COH |
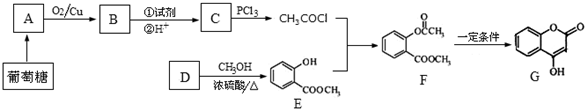
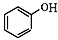 +NaOH��
+NaOH��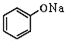 +H2O
+H2O ��
��