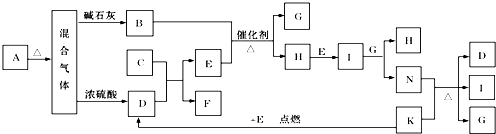
��Ŀ����
��������ؽᾧʵ������������£�
�ٽ��ֱ�����1g�ӵ�100mL���ձ��У��ټ���50mL����ˮ����ʯ�����ϱ߽�����ȣ�ʹ�ֱ������ܽ⣬��ȫ�ܺ��ټ�����������ˮ�����裮��Ȼ��ʹ�ö̾�����©�����Ƚ���Һ���˵���һ100mL�ձ��У�����Һ���ã�ʹ�仺����ȴ�ᾧ�����˳����壮�ش��������⣺
��1�������IJ��������������õ��������IJ�����
��2������ڵ�Ŀ���� ��
��3���۲���Һ��ȴʱ��ʵ������ ��
��4���ؽᾧ�����״̬�� ��
�ٽ��ֱ�����1g�ӵ�100mL���ձ��У��ټ���50mL����ˮ����ʯ�����ϱ߽�����ȣ�ʹ�ֱ������ܽ⣬��ȫ�ܺ��ټ�����������ˮ�����裮��Ȼ��ʹ�ö̾�����©�����Ƚ���Һ���˵���һ100mL�ձ��У�����Һ���ã�ʹ�仺����ȴ�ᾧ�����˳����壮�ش��������⣺
��1�������IJ��������������õ��������IJ�����
��2������ڵ�Ŀ����
��3���۲���Һ��ȴʱ��ʵ������
��4���ؽᾧ�����״̬��
���㣺���ʷ��롢�ᴿ��ʵ�鷽�����
ר�⣺ʵ�������
��������1����ʵ�鲽���֪�����ܽ⡢�ڽ��衢�۹��ˡ���ת�ƾ��壬����Ҫ��������
��2���ټ�����������ˮ��ˮ�ڼ��ȹ����л�������������������ˮ��
��3����ȴʱ����������
��3���ᾧ����Ϊ���壮
��2���ټ�����������ˮ��ˮ�ڼ��ȹ����л�������������������ˮ��
��3����ȴʱ����������
��3���ᾧ����Ϊ���壮
���
�⣺��1����ʵ�鲽���֪�����ܽ⡢�ڽ��衢�۹��ˡ���ת�ƾ��壬����Ҫ����������٢ڢܾۢ�ʹ�ã��ʴ�Ϊ���٢ڢۢܣ�
��2������ڵ�Ŀ����ʹ���ܽ⣬���ٹ���ʱ���������ʧ����ֹ��������ǰ�����ᾧ���ʴ�Ϊ�����ٹ���ʱ���������ʧ����ֹ��������ǰ�����ᾧ��
��3����Һ��ȴʱ��ʵ������Ϊ������������Խ��Խ�࣬�ʴ�Ϊ��������������Խ��Խ�ࣻ
��3���ᾧ����Ϊ���壬���й���ļ������Σ��ʴ�Ϊ�����壮
��2������ڵ�Ŀ����ʹ���ܽ⣬���ٹ���ʱ���������ʧ����ֹ��������ǰ�����ᾧ���ʴ�Ϊ�����ٹ���ʱ���������ʧ����ֹ��������ǰ�����ᾧ��
��3����Һ��ȴʱ��ʵ������Ϊ������������Խ��Խ�࣬�ʴ�Ϊ��������������Խ��Խ�ࣻ
��3���ᾧ����Ϊ���壬���й���ļ������Σ��ʴ�Ϊ�����壮
���������⿼�����ʵķ����ᴿʵ�飬Ϊ��Ƶ���㣬���ձ���������ʼ��ؽᾧΪ���Ĺؼ���ע�ⱥ����Һ�ᾧ�������壬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
�й�ҩ���������ȷ���ǣ�������
| A���˹��ϳ�ҩ��ֻ�а�˾ƥ�� |
| B��1853�꣬�¹��ݶ�ҩ���Ļ�ѧ�һ������ϳ�������ˮ���� |
| C����ù�����˹��ϳ�ҩ���е�����һ�� |
| D������ҩ���˺���̼�����ơ��������������������⣬�����е�ζ�����ϼ� |
 ij���������ᷴӦʱ������H2�����������Ľ������ʵع�ϵ��ͼ��ʾ���˽��������ǣ�������
ij���������ᷴӦʱ������H2�����������Ľ������ʵع�ϵ��ͼ��ʾ���˽��������ǣ�������| A����п | B������ |
| C��þп�Ͻ� | D��ͭ���Ͻ� |
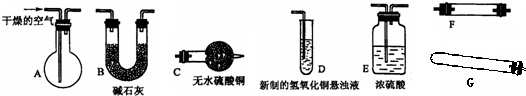
 ��ѧʵ���еĺܶ�����������������ȡ�ģ���͵�������Щ��ȡ����������������HCl���ʣ�Ҫ��ȥHCl���ʶ��õ�������Ŀ�����壬����ͼ��ʾװ�ã�������ƿ��ʢװ���DZ���NaOH��Һ�����������������������ij���װ���ǣ���ʾCl2+H2O=HCl+HClO����������
��ѧʵ���еĺܶ�����������������ȡ�ģ���͵�������Щ��ȡ����������������HCl���ʣ�Ҫ��ȥHCl���ʶ��õ�������Ŀ�����壬����ͼ��ʾװ�ã�������ƿ��ʢװ���DZ���NaOH��Һ�����������������������ij���װ���ǣ���ʾCl2+H2O=HCl+HClO����������| A��H2 |
| B��Cl2 |
| C��SO2 |
| D��CO2 |
ˮ�ĵ������ΪH2O?H++OH-���ڲ�ͬ�¶��������ӻ�Ϊ��Kw��25�棩=1��10-14��Kw��35�棩=2.1��10-14��������������ȷ���ǣ�������
| A��c��H+�������¶ȵ����߶����� |
| B����35��ʱ����ˮ�� c��H+����c��OH-�� |
| C��ˮ�ĵ��볣��K��25�棩��K��35�棩 |
| D��ˮ�ĵ�����һ�����ȹ��� |
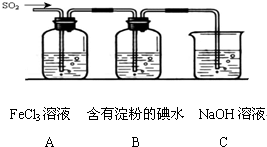 ij��ȤС��̽��SO2���廹ԭFe3+��I2������ʹ�õ�ҩƷ��װ����ͼ��ʾ��
ij��ȤС��̽��SO2���廹ԭFe3+��I2������ʹ�õ�ҩƷ��װ����ͼ��ʾ��