��Ŀ����
9�������йص�������ȷ���ǣ�������| A�� | ��֪2H2��g��+O2��g���T2H2O��l������H=-483.6kJ/mol����������ȼ����Ϊ241.8kJ/mol | |
| B�� | C��s��ʯī��=C��s�����ʯ������H=+1.9kJ•mol-1��˵�����ʯ��ʯī�ȶ� | |
| C�� | CaCO3��s���TCaO��s��+CO2��g����H��0����S��0���÷�Ӧ�κ��¶��¶����Է����� | |
| D�� | ��֪��C��s��+$\frac{1}{2}$O2��g���TCO��g����H1��C��s��+O2��g���TCO2��g����H2�����H1����H2 |
���� A��ȼ������ָ��ȼ����ȫȼ�������ȶ�������ʱ�ų���������H����Һ̬ˮ��
B�����ʾ��е�����Խ��Խ�ȶ���
C��̼��Ʒֽ��������ȷ�Ӧ����H��0��CaCO3��s���TCaO��s��+CO2��g��������ֽ�õ����壬���Ҷ����ӣ���S��0�����ݡ�G=��H-T•��S��0�Է��жϣ�
D����ȫȼ�շų��������ڲ���ȫȼ�շų����������ų�����Խ�෴Ӧ��ԽС��
��� �⣺A��ȼ������ָ��ȼ����ȫȼ�������ȶ�������ʱ�ų���������Hȼ������Һ̬ˮ����Ϊ2H2��g��+O2��g���T2H2O��l������H=-483.6kJ/mol������������ȼ����Ϊ241.8kJ/mol����A��ȷ��
B��ʯīת��Ϊ���ʯ���ȣ�˵��ʯī�������ϵͣ�����Խ��Խ�ȶ������ʯī�Ƚ��ʯ�ȶ�����B����
C��̼��Ʒֽ��������ȷ�Ӧ����H��0��CaCO3��s���TCaO��s��+CO2��g��������ֽ�õ����壬���Ҷ����ӣ���S��0���ڸ��������¡�G=��H-T•��S��0��Ӧ�Է����У�
����ʱ���Է�����C����
D����ȫȼ�շų��������ڲ���ȫȼ�շų����������ų�����Խ�෴Ӧ��ԽС�����ԡ�H1����H2����D����
��ѡA��
���� ���⿼���Ϊ�ۺϣ��漰��ѧ��Ӧ���еķ������ʵ��ȶ��Ե�֪ʶ���ѶȲ���ע����ݡ�G=��H-T•��S��0��Ӧ�Է����з�����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | �Ҵ��ķ���ʽ��C2H5OH | B�� | F-�Ľṹʾ��ͼ�� | ||
| C�� | ������Ϊ20����ԭ�ӣ�${\;}_{17}^{20}$Cl | D�� | +4�۵���Ԫ�أ�S4+ |
| A�� | 4�� | B�� | 5�� | C�� | 6�� | D�� | 7�� |
| A�� | ����������ȴ�ᾧ���˵ķ������Ȼ��ƺ�����صĻ����Һ���������� | |
| B�� | �÷�Һ�ķ����������������ı���̼������Һ | |
| C�� | �ü��ȵķ����ᴿ��������̼�����Ƶ�̼���� | |
| D�� | �ü�ˮ�ܽ⡢���˵ķ����Ậ�������Ƶ�̼��� |
| A�� | Na2CO3 | B�� | NaOH | C�� | HCl | D�� | BaCl2 |
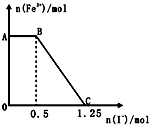 ��֪���Ը��������Һ���Խ�FeSO4��������ѧ��Ӧ�ķ���ʽΪ��2KMnO4+10FeSO4+8H2SO4�TK2SO4+2MnSO4+5Fe2��SO4��3+8H2O���ֽ�һ�����������ữ�ĸ��������Һ������������Һ��ϣ���ַ�Ӧ������������Һ�м���KI��Һ�������Һ�������ӵ����ʵ���������KI�� ���ʵ����ı仯��ϵ��ͼ��ʾ���������й�˵������ȷ���ǣ�������
��֪���Ը��������Һ���Խ�FeSO4��������ѧ��Ӧ�ķ���ʽΪ��2KMnO4+10FeSO4+8H2SO4�TK2SO4+2MnSO4+5Fe2��SO4��3+8H2O���ֽ�һ�����������ữ�ĸ��������Һ������������Һ��ϣ���ַ�Ӧ������������Һ�м���KI��Һ�������Һ�������ӵ����ʵ���������KI�� ���ʵ����ı仯��ϵ��ͼ��ʾ���������й�˵������ȷ���ǣ�������| A�� | ͼ��AB����Ҫ�Ǹ�����غ͵⻯����Һ��Ӧ | |
| B�� | ͼ��BC�η����ķ�ӦΪ2Fe3++2I-�T2Fe2++I2 | |
| C�� | ����OC�ε����ݿ�֪��ʼ����ĸ�����ص����ʵ���Ϊ0.25mol | |
| D�� | ��C���Ժ����Һ�м�������KSCN��Һ����Һ���ɫ |
| A�� | �ֱ����������Ʒ��û�в���������NaHCO3 | |
| B�� | �ֱ������Һ���ټ������ʯ��ˮ���ް�ɫ�������ɵ���NaHCO3 | |
| C�� | �ֱ�����ͬ���ʵ���Ũ�ȵ����ᷴӦ������CO2������Na2CO3��NaHCO3 | |
| D�� | ����ʱ��ˮ�е��ܽ�ȣ�Na2CO3��NaHCO3 |
| A�� | ���⻯ѧ�������������������ꡱ���γɶ��뵪���������й� | |
| B�� | �����ʺ硱��������¥������һ����Ȼ��������һ�ֹ�ѧ���� | |
| C�� | PM2.5��������ֱ��ԼΪ2.5��10-6m����ɢ�ڿ������γ����ܽ� | |
| D�� | ���⺵��Ĵ�ʩ֮һ�ǿ����øɱ���⻯�������˹����� |
 �ֻ�������һ���������õIJ����������Կ˷������ʴ������ȱ�㣮���ӽ������Dz������иֻ���һ����Ҫ����������ͨ���ƸƲ�����̼��ص�ǿ���Σ�475�棩�н���3.5Сʱ���Ƶã���ͼ�����ӽ�������ʾ��ͼ��
�ֻ�������һ���������õIJ����������Կ˷������ʴ������ȱ�㣮���ӽ������Dz������иֻ���һ����Ҫ����������ͨ���ƸƲ�����̼��ص�ǿ���Σ�475�棩�н���3.5Сʱ���Ƶã���ͼ�����ӽ�������ʾ��ͼ��