��Ŀ����
20�������Ԫ�����ڱ���һ���֣��ش������й����⣺| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ����A | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� | �� | |
| 4 | �� | ⑪ | ⑫ |
��2������ԭ�ӵĽṹʾ��ͼ����
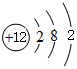 ����
���� ��
����3������ЩԪ���У�����õĽ���Ԫ����K������õķǽ���Ԫ����F������õ�Ԫ����Ar��������Ԫ�ط��ű�ʾ��
��4������ЩԪ�ص�����������Ӧˮ�����У�������ǿ����HClO4��������ǿ����KOH�������Ե�����������Al��OH��3������д�����ʵĻ�ѧʽ��
���� ��ͼ��֪�١�⑪�ֱ�ΪC��O��Na��Mg��Al��Si��S��Cl��Ar��K��Ca��
��1��������������֪Ԫ�صķ��ϣ�
��2��Mg��������Ϊ12��Al��������Ϊ13��
��3��ͬ���ڴ������ҽ����Լ������ǽ�������ǿ��ͬ������ϵ��·ǽ����Լ�������������ǿ��ϡ��������������ȶ���
��4���������������ǿ��KOH�ļ�����ǿ�����������������ԣ�
��� �⣺��ͼ��֪�١�⑪�ֱ�ΪC��O��Na��Mg��Al��Si��S��Cl��Ar��K��Ca��
��1���٢�Ԫ�ط��ŷֱ�ΪN��Si���ʴ�Ϊ��N��Si��
��2���ܢݵ�ԭ�ӵĽṹʾ��ͼ�ֱ�Ϊ ��
�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
�� ��
��
��3������ЩԪ���У�����õĽ���Ԫ����K������õķǽ���Ԫ����F������õ�Ԫ����Ar���ʴ�Ϊ��K��F��Ar��
��4������ЩԪ�ص�����������Ӧˮ�����У�������ǿ����HClO4��������ǿ����KOH�������Ե�����������Al��OH��3���ʴ�Ϊ��HClO4��KOH��Al��OH��3��
���� ���⿼��λ�á��ṹ�����ʣ�Ϊ��Ƶ���㣬����Ԫ�ص�λ�á����ʡ�Ԫ�ػ�����֪ʶΪ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע�⻯ѧ�����ʹ�ã���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
11�����б��﷽ʽ������ǣ�������
| A�� | CO2�ı���ģ�ͣ� | |
| B�� | ����ĵ���ʽ 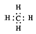 | |
| C�� | �����ӵĺ�������Ų�ʽ 1s22s22p63s23p4 | |
| D�� | ̼-12ԭ�ӹ���${\;}_6^{12}$C |
8���������ӷ���ʽ����д��ȷ���ǣ�������
| A�� | ̼��ƺ����ᷴӦ CO32-+2H+=H2O+CO2�� | |
| B�� | ������������Һ�м�������Һ Ba2++SO42-=BaSO4�� | |
| C�� | ̼�������Һ��������Һ��Ӧ HCO3-+H+=H2O+CO2�� | |
| D�� | ��ϡ������Һ�м����� 3Fe+6H+=2Fe3++3H2�� |
12������ʵ����е��ǣ�������
| A�� | �ñ���Na2CO3��Һ��ȥCO2�л��е�HCl���� | |
| B�� | ����Ͳ��ȡ20.83mL12mol/L��Ũ��������250mL1mol/Lϡ���� | |
| C�� | ��Ũ�������CO2��SO2��HI���������� | |
| D�� | ��ϡHNO3�ܼ���Mg��Na2CO3��NaAlO2��Na2SiO3���ֹ��� |
10������Ԫ�����ڱ��������ɣ������ƶ���ȷ���ǣ�������
| A�� | HCl��HBr��HI�����ȶ���������ǿ | B�� | KOH�ļ��Ա�NaOH�ļ���ǿ | ||
| C�� | HBrO4���Ա�HClO4ǿ | D�� | Na�Ľ����Ա�Al���� |
