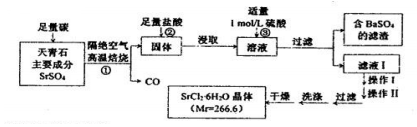
��Ŀ����
2��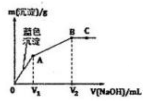 ��MgCl2��CuCl2����Һ����μ���0.1 mol•L-1��NaOH��Һ��������������m�������NaOH��Һ�����V���Ĺ�ϵ��ͼ��ʾ����֪V2=3V1������˵����ȷ���ǣ�������
��MgCl2��CuCl2����Һ����μ���0.1 mol•L-1��NaOH��Һ��������������m�������NaOH��Һ�����V���Ĺ�ϵ��ͼ��ʾ����֪V2=3V1������˵����ȷ���ǣ�������| A�� | ˮ�ĵ���̶ȣ�A��B��C | |
| B�� | ��ʵ�鲻��֤��Cu��OH��2��Ksp��Mg��OH��2��KspС | |
| C�� | ԭ��Һ��MgCl2��CuCl2�����ʵ���Ũ����� | |
| D�� | ����Mg��OH��2����Һ�м���CuCl2��Һ��һ������Cu��OH��2���� |
���� ��ͼ���֪��0��V1ʱ������ɫ����ΪCu��OH��2��V1��V2���ɵij���ΪMg��OH��2��V2=3V1��˵��n��Mg��OH��2��=2n��Cu��OH��2����A��ʱ����ΪNaCl��MgCl2��B��ʱΪNaCl��C��ΪNaCl��NaOH���ݴ˽��
��� �⣺A��A��ʱ����ΪNaCl��MgCl2��B��ʱΪNaCl��C��ΪNaCl��NaOH��þ����ˮ��ٽ�ˮ�ĵ��룬NaOH����ˮ�ĵ��룬NaCl��ˮ�ĵ�����Ӱ�죬��A��ȷ��
B��CuCl2�����ʵ���Ũ��С�����ȳ���ͭ����˵��Cu��OH��2��Ksp��Mg��OH��2��KspС����B����
C��V2=3V1��˵��n��Mg��OH��2��=2n��Cu��OH��2����ԭ��Һ��MgCl2��CuCl2�����ʵ���Ũ�Ȳ���ȣ���C����
D��Ҫ�����ɳ�������������Qc��Ksp��ͭ���Ӵ�Сδ֪�����ж��Ƿ����ɳ�������D����
��ѡA��
���� ���⿼�����ܵ���ʵ��ܽ�ƽ�⼰����ת���ı��ʣ��漰�������ܶȻ�������ˮ���Լ�����жϣ���Ŀ�ѶȲ���ͼ���жϳ���������˳���ǽ���Ĺؼ���

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ
12��ϡ��ȼ�շ��������и�Ч�����á��������ŵ㣬����ͨ�����������Ա������
��1�����ݱ������ݿ�֪��ϡ�����ĺ�����A
A��������������е����ͺ����� B��������������еĿ���������
C��������������������ĺ����� D��������������е����ĺ�����
��2��Ϊ��ϡ��ȼ�շ�����������β����CO��������٣�
��3����֪����β���еĵ��������NOX��ʾ���ǵ����������ڸ����·�Ӧ�����ģ�
�Խ���Ϊ��ϡ��ȼ�շ�����������β����NOX����Ҳ����٣�
��4��ij�ּ���β����CO��NOX�ķ�����ʹ�ô�ת������ת�������з�����Ӧ�Ļ�ѧ����ʽΪ��CO+NOX��CO2+N2��δ��ƽ������x=1.5����ѧ����ʽ��CO2��N2��ϵ��֮��Ϊ3��1��
| ������������ | ������������������������������ | ��������ת���¶� | β����CO�ĺ��� | β����NO3�ĺ��� |
| ��ͨ������ | 1��15 | �ϸ� | �ϸ� | �ϸ� |
| ϡ��ȼ�շ����� | 1��40 | �ϵ� | �ϵ� | �ϵ� |
A��������������е����ͺ����� B��������������еĿ���������
C��������������������ĺ����� D��������������е����ĺ�����
��2��Ϊ��ϡ��ȼ�շ�����������β����CO��������٣�
��3����֪����β���еĵ��������NOX��ʾ���ǵ����������ڸ����·�Ӧ�����ģ�
�Խ���Ϊ��ϡ��ȼ�շ�����������β����NOX����Ҳ����٣�
��4��ij�ּ���β����CO��NOX�ķ�����ʹ�ô�ת������ת�������з�����Ӧ�Ļ�ѧ����ʽΪ��CO+NOX��CO2+N2��δ��ƽ������x=1.5����ѧ����ʽ��CO2��N2��ϵ��֮��Ϊ3��1��
13���������ӷ���ʽ��д����ȷ���ǣ�������
| A�� | NaOH��Һ��SO2��Ӧ����n��NaOH����n��SO2��=4��3ʱ��4OH-+3SO2=SO32-+2HSO3-+H2O | |
| B�� | ��1mol��NaOH��Һ��ͨ��1mol��H2S���壺H2S+2OH-=S2-+2H2O | |
| C�� | Ca��HCO3��2��Һ������NaOH��Һ��Ӧ��HCO3-+Ca2++OH-=CaCO3��+H2O | |
| D�� | 1mol/L��NaAlO2��Һ��2.5mol/L��HCl�����������Ȼ�ϣ�2AlO2-+5H+=Al��OH��3��+Al3++H2O |
10������˵����ȷ���ǣ�������
| A�� | ij��Ӧ�����淴Ӧ�Ļ�ֱܷ�ΪEakJ/mol��EbkJ/mol�����H=-��Ea-Eb��kJ/mol | |
| B�� | H2����ֵΪ142.9kJ/g������2H2��g��+O2��g���T2H2O ��1����H=-285.8kJ/mol | |
| C�� | ��Ũ��Ϊ0.1mol/LHF��Һ��ˮ����ϡ�����У�$\frac{c��{F}^{-}��}{c��{H}^{+}��}$ʼ�ձ��ּ�С | |
| D�� | ���ǰ$\frac{c��C{H}_{3}COOH��}{c��NaOH��}$=2��������Ļ�Ϻ���Һ�У�$\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=$\frac{c��C{H}_{3}COOH��}{c��C{H}_{3}CO{O}^{-}��}$ |
17��һ�������ļ���ȼ�պ�IJ���ΪCO��CO2��ˮ�������˻����������Ϊ99.2g�����仺��������ˮCaCl2ʱ��CaCl2����50.4g��ԭ���������CO2������Ϊ��������
| A�� | 13.2 g | B�� | 22.4g | C�� | 24.4g | D�� | 26.4g |
1����ʱ��������ԭ��Ӧ����ʽ��д���������뷴Ӧ����������һ�����뷴Ӧ��ʽ��MnO4-+____H++____e---____Mn2++____H2O
�÷�Ӧ����ʽ����ƽ��ѧ�������ǣ�������
�÷�Ӧ����ʽ����ƽ��ѧ�������ǣ�������
| A�� | 1��8��4��1��4 | B�� | 2��16��5��2��8 | C�� | 1��6��5��1��3 | D�� | 1��8��5��1��4 |
18������˵����ȷ���ǣ�������
| A�� | 1molOH-��������17g | B�� | 1mol������ռ���������22.4L | ||
| C�� | ������Ħ��������2g | D�� | Ħ�������ʵ�������λ |