��Ŀ����
��Ȼ���У�����������������������������������ã�������ͭ��Cu2S����ͭ����CuS����
��֪��Cu2S��CuS�����ֲ�����ˮ�ĺ�ɫ���壬��һ�������¶�����ϡHNO3��Ӧ��
��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O
��3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O
�ֽ��ķ�������ͬ��ijCu2S��CuS�������Ʒ�ֱ���100mL 5mol/L��ϡ�����ַ�Ӧ����Ʒ����������������������ۺϳɱ�״�������±���ʾ��
�Իش��������⣨����������Ļӷ��Լ���Һ����仯����
��1������������Ϊ0.63���ܶ�Ϊ1.42g/cm3��Ũ��������100mL��5mol/L��ϡ���ᣬ��Ũ��������Ϊ ��������1λС����
��2����ʵ���������Һ��c��NO3-��= ��
��3���������Ʒ��Cu2S��CuS�����ʵ���֮���Ƕ��٣���д��������̣�
��4����ʵ���������Һ�������ӵ�Ũ���Ƕ��٣���д��������̣�
��5�������ʵ���в�������������V������д��������̣�
��֪��Cu2S��CuS�����ֲ�����ˮ�ĺ�ɫ���壬��һ�������¶�����ϡHNO3��Ӧ��
��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O
��3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O
�ֽ��ķ�������ͬ��ijCu2S��CuS�������Ʒ�ֱ���100mL 5mol/L��ϡ�����ַ�Ӧ����Ʒ����������������������ۺϳɱ�״�������±���ʾ��
| ʵ���� | �� | �� | �� |
| ��Ʒ������g�� | 9.6 | 12.8 | 64.0 |
| ���������L�� | 5.04 | 6.72 | V |
��1������������Ϊ0.63���ܶ�Ϊ1.42g/cm3��Ũ��������100mL��5mol/L��ϡ���ᣬ��Ũ��������Ϊ
��2����ʵ���������Һ��c��NO3-��=
��3���������Ʒ��Cu2S��CuS�����ʵ���֮���Ƕ��٣���д��������̣�
��4����ʵ���������Һ�������ӵ�Ũ���Ƕ��٣���д��������̣�
��5�������ʵ���в�������������V������д��������̣�
���㣺�йػ���ﷴӦ�ļ���
ר�⣺
��������1������c=
�������Ũ�����Ũ�ȣ�Ȼ�����ϡ���������ʵ����ʵ�������������ҪŨ����������
��2�����ݷ�Ӧ��֪��������������ʵ�����������������ӵ����ʵ�����ȣ��ݴ˼������Ӧ����Һ�����������Ũ�ȣ�
��3�����ݼ��������ж����л����ǡ����ȫ��Ӧ��Ȼ������������Cu2S��CuS�����ʵ������ֱ������������������������ʵ�����ʽ���㣻
��4����ϣ�3���������Ӧ�����������ӵ����ʵ������ٸ���c=
���������������Ũ�ȣ��������ʣ��������Ũ�ȣ�
��5�����ݣ�3�������64.0g������к��е�CuS��Cu2S�����ʵ�����Ȼ�����ü�ֵ���ֱ���������NO�����ʵ�����������Ӷ��ó�V�ķ�Χ��
| 1000�Ѧ� |
| M |
��2�����ݷ�Ӧ��֪��������������ʵ�����������������ӵ����ʵ�����ȣ��ݴ˼������Ӧ����Һ�����������Ũ�ȣ�
��3�����ݼ��������ж����л����ǡ����ȫ��Ӧ��Ȼ������������Cu2S��CuS�����ʵ������ֱ������������������������ʵ�����ʽ���㣻
��4����ϣ�3���������Ӧ�����������ӵ����ʵ������ٸ���c=
| n |
| V |
��5�����ݣ�3�������64.0g������к��е�CuS��Cu2S�����ʵ�����Ȼ�����ü�ֵ���ֱ���������NO�����ʵ�����������Ӷ��ó�V�ķ�Χ��
���
�⣺��1����������Ϊ0.63���ܶ�Ϊ1.42g/cm3��Ũ�����Ũ��Ϊ��
=14.2mol/L������100mL��5mol/L��ϡ���ᣬ���ƹ�������������ʵ������䣬����Ҫ��Ũ��������Ϊ��
��0.0352L=35.2mL��
�ʴ�Ϊ��35.2mL��
��2�����ݷ�Ӧ��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O����3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O��֪����Ӧ����NO��������ʵ�����������������ӵ����ʵ�����ȣ���ʵ��������NO�����ʵ���Ϊ��
=0.225mol�����������Ũ��Ϊ��
=2.25mol/L����Ӧ����ҺŨ��Ϊ��5mol/L-2.25mol/L=2.75mol/L��
�ʴ�Ϊ��2.75mol/L��
��3��9.6g���������5.04LNO���壬12.8g������ܹ�����һ�����������Ϊ��
��5.04L=6.72L��˵�����л������ȫ��Ӧ��
��12.8g����Ʒ��CuS�����ʵ���Ϊx��Cu2S�����ʵ���Ϊy��
���������ɵã���96x+160y=12.8��
������������ɵã���
x+
y=
=0.3��
���ݢ٢ڽ�ã�x=y=0.05mol��
���Ի������Ʒ��Cu2S��CuS�����ʵ���֮��Ϊ��1��1��
�𣺻������Ʒ��Cu2S��CuS�����ʵ���֮��1��1��
��4�����ݷ���ʽ�١��ڿ�֪���������ӵ����ʵ���Ϊ��n��H+��=0.05mol��
+0.05mol��
=0.4mol��
�����������ӵ�Ũ��Ϊ��c��H+��=
=4mol/L��
����ʣ��������Ũ��Ϊ��c��H+��=5mol/L-4mol/L=1mol/L��
����ʵ���������Һ�������ӵ�Ũ��Ϊ1mol/L��
��5��100mL 5mol/L��ϡ�����к�����������ʵ���Ϊ��5mol/L��0.1L=0.5mol��
���ݣ�3����֪��64g��Ʒ��CuS��Cu2S�����ʵ���Ϊ��n��Cu2S��=n��CuS��=0.05mol��
=0.25mol��
��HNO3ֻ��CuS��Ӧ��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O��0.25molCuS��ȫ��Ӧ�������0.25mol��
��0.5mol������㣬����ݷ�Ӧ��֪����NO�����ʵ���Ϊ��0.5mol�������NO���Ϊ��V��NO��=0.5mol��22.4L/mol=11.2L��
��HNO3ֻ��Cu2S��Ӧ��3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O��0.25molCu2S�������0.25mol��
��0.5mol������㣬���ݷ�Ӧ��֪����NO�����ʵ���Ϊ��0.5mol��
=
mol�������NO�����22.4L/mol��
mol=7L��
���Ա��в������������V��Ϊ��7L��V��11.2L��
�𣺱�ʵ���в�����������Ϊ��7L��V��11.2L��
| 1000��1.42��0.63 |
| 63 |
| 5mol/L��0.1L |
| 14.2mol/L |
�ʴ�Ϊ��35.2mL��
��2�����ݷ�Ӧ��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O����3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O��֪����Ӧ����NO��������ʵ�����������������ӵ����ʵ�����ȣ���ʵ��������NO�����ʵ���Ϊ��
| 5.04L |
| 22.4L/mol |
| 0.225mol |
| 0.1L |
�ʴ�Ϊ��2.75mol/L��
��3��9.6g���������5.04LNO���壬12.8g������ܹ�����һ�����������Ϊ��
| 12.8g |
| 9.6g |
��12.8g����Ʒ��CuS�����ʵ���Ϊx��Cu2S�����ʵ���Ϊy��
���������ɵã���96x+160y=12.8��
������������ɵã���
| 8 |
| 3 |
| 10 |
| 2 |
| 6.72 |
| 22.4 |
���ݢ٢ڽ�ã�x=y=0.05mol��
���Ի������Ʒ��Cu2S��CuS�����ʵ���֮��Ϊ��1��1��
�𣺻������Ʒ��Cu2S��CuS�����ʵ���֮��1��1��
��4�����ݷ���ʽ�١��ڿ�֪���������ӵ����ʵ���Ϊ��n��H+��=0.05mol��
| 8 |
| 3 |
| 16 |
| 3 |
�����������ӵ�Ũ��Ϊ��c��H+��=
| 0.4mol |
| 0.1L |
����ʣ��������Ũ��Ϊ��c��H+��=5mol/L-4mol/L=1mol/L��
����ʵ���������Һ�������ӵ�Ũ��Ϊ1mol/L��
��5��100mL 5mol/L��ϡ�����к�����������ʵ���Ϊ��5mol/L��0.1L=0.5mol��
���ݣ�3����֪��64g��Ʒ��CuS��Cu2S�����ʵ���Ϊ��n��Cu2S��=n��CuS��=0.05mol��
| 64g |
| 12.8g |
��HNO3ֻ��CuS��Ӧ��3CuS+8H++8NO3-��3Cu2++3SO42-+8NO��+4H2O��0.25molCuS��ȫ��Ӧ�������0.25mol��
| 8 |
| 3 |
��HNO3ֻ��Cu2S��Ӧ��3Cu2S+16H++10NO3-��6Cu2++3SO42-+10NO��+8H2O��0.25molCu2S�������0.25mol��
| 16 |
| 3 |
| 10 |
| 16 |
| 5 |
| 16 |
| 5 |
| 16 |
���Ա��в������������V��Ϊ��7L��V��11.2L��
�𣺱�ʵ���в�����������Ϊ��7L��V��11.2L��
���������⿼���˻���ﷴӦ�ļ��㣬��Ŀ�Ѷ��еȣ�����֪ʶ��϶ࡢ�������ϴ�ֿ�����ѧ���ķ�����������������ѧ����������ע�⼫ֵ���ڻ�ѧ�����е�Ӧ�ã�

��ϰ��ϵ�д�
�����Ŀ
 25��ʱ����ʢ��50mL pH=2��HA��Һ�ľ��������м���pH=13��NaOH��Һ������NaOH��Һ�������V�������û����Һ���¶ȣ�T���Ĺ�ϵ��ͼ��ʾ������������ȷ���ǣ�������
25��ʱ����ʢ��50mL pH=2��HA��Һ�ľ��������м���pH=13��NaOH��Һ������NaOH��Һ�������V�������û����Һ���¶ȣ�T���Ĺ�ϵ��ͼ��ʾ������������ȷ���ǣ�������| A��HA��Һ�����ʵ���Ũ��Ϊ0 0l mol��L-1 |
| B��b��c�Ĺ����У��¶Ƚ��͵�ԭ������Һ�з��������ȷ�Ӧ |
| C��a��b�Ĺ����У������Һ�п��ܴ��ڣ�c��A-��=c��Na+�� |
| D��25��ʱ��HA�ĵ���ƽ�ⳣ��KԼΪ1.43��10-2 |
��֪NaHSO3��Һ�����ԣ���Һ�д�������ƽ�⣺
��HSO3-+H2O?H2SO3+OH-
��HSO3-?H++SO32-
��0.1mol/L��NaHSO3��Һ�зֱ�����������ʣ������й�˵����ȷ���ǣ�������
��HSO3-+H2O?H2SO3+OH-
��HSO3-?H++SO32-
��0.1mol/L��NaHSO3��Һ�зֱ�����������ʣ������й�˵����ȷ���ǣ�������
| A��������������Na��ƽ������ƣ�ƽ������ƣ���Һ��c��HSO3-������ | ||||||||
B����������Na2SO3���壬��c��H+��+c��Na+��=c��HSO3-��+c��OH-��+
| ||||||||
C����������NaOH��Һ��
| ||||||||
| D�����백ˮ�����ԣ���c��Na+��=c��SO32-��+c��HSO3-��+c��H2SO3�� |
20��ʱ������KNO3��Һ���ܶ�Ϊ��g/cm3���ѣ�1�������ʵ���Ũ��Ϊa mol/L������˵���в���ȷ�ģ�������
| A������Һ��KNO3����������Ϊ10.1 a/��% |
| B��20��ʱ���ܶ�С�ڦ�g/cm3����ҺΪ��������Һ |
| C������Һ��������ˮ���ٻָ���20�棬��Һ�ܶ�һ�����ڦ�g/cm3 |
| D��25��ʱ������KNO3��Һ�����ʵ���Ũ�ȴ���a mol/L |
ʵ������ȡ�����������Ȼ��̣��ܴﵽ��ʵ��Ŀ��װ���ǣ�������
A�� ��װ����ȡ���� ��װ����ȡ���� |
B�� ��װ�÷���������̺��Ȼ�����Һ ��װ�÷���������̺��Ȼ�����Һ |
C�� ��װ�÷�����ȡ���ˮ�е� ��װ�÷�����ȡ���ˮ�е� |
D��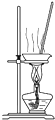 ��װ�������Ȼ�����Һ�� MnCl2?4H2O ��װ�������Ȼ�����Һ�� MnCl2?4H2O |
 ������ķ��㰷����¶�ڿ����б�Ϊ��ɫ���ñ仯���п����뱽������
������ķ��㰷����¶�ڿ����б�Ϊ��ɫ���ñ仯���п����뱽������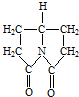 д�����л����������ˮ�����Ľṹ��ʽ
д�����л����������ˮ�����Ľṹ��ʽ