��Ŀ����
5��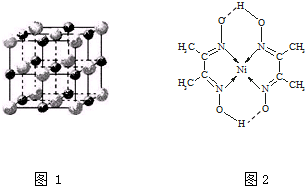 ��1����֪A��BΪ��������Ԫ�أ���ԭ�ӵĵ�һ�����ĵ��������1��ʾ��
��1����֪A��BΪ��������Ԫ�أ���ԭ�ӵĵ�һ�����ĵ��������1��ʾ����1
| ������/kJ•mol-1 | I1 | I2 | I3 | I4 |
| A | 578 | 1817 | 2745 | 11578 |
| B | 738 | 1451 | 7733 | 10540 |
��2�������Ĺ��������е�����ԼΪ399kJ•mol-1�����ݱ�2�йص����ʷ�������Ҫ��ѧ������Ϣ��˵�����峤ʱ������������Ƥ�������˺���ԭ���������е������ȵ����ʷ����еĻ�ѧ��C-C��C-N��C-S�ļ��ܴ���������������ʹ��Щ�����ѣ��Ӷ��ƻ������ʷ��ӣ�
��2
| ���ۼ� | C-C | C-N | C-S |
| ����/kJ•mol-1 | 347 | 305 | 259 |
��3��ʵ��֤����KCl��MgO��CaO��TiN��4�־���Ľṹ��NaCl����ṹ���ƣ���ͼ1��ʾ��������3�����Ӿ���ľ������������3��
��3
| ���Ӿ��� | NaCl | KCl | CaO |
| �����ܣ�kJ•mol-1�� | 786 | 715 | 3401 |
��4�����������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�ã�������������V2O5��CrO2�У��ʺ���¼�����ŷ�
ԭ�ϵ���CrO2��
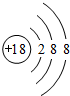
��5��ij�����ķ��ӽṹ��ͼ2��ʾ��������ڲ�����AC������ţ���
A�����Ӽ� B�����Լ�C�������� D����λ��E����� F���Ǽ��Լ�
��6������ЧӦ��Ҫ��CO2���𣬿�ѧ����Ʒ�Ӧ��CO2+4H2��CH4+2H2O�Լ�С������CO2������1mol CH4���ɣ�����6mol�Ҽ���2mol�м����ѣ�
���� ��1��A��BΪ��������Ԫ�أ�AԪ�ص��ĵ����ܾ�����˵��Aԭ���������3�����ӣ���AΪAl��BԪ�ص��������ܾ�����˵��Bԭ���������2�����ӣ���BΪMg��
��2�������Ĺ��������е��������ڵ����ʷ�����һЩ��Ҫ��ѧ�����ܣ�ʹ��ѧ����ʽ���ѣ�
��ɵ����ʵ���İ�����ΪH2N-CH2-COOH��-CH2-�е�̼ԭ���ӻ������ĿΪ4��-COOH��̼ԭ���ӻ������ĿΪ3��
��3�����Ӿ���ľ�����Խ���۵�Խ�ߣ����Ӿ�������Ӱ뾶ԽС���������Խ�࣬������Խ��ϱ��������жϣ�
MgO��NaCl�ṹ���ƣ�����ɫ����Mg2+�����ɫ��ΪO2-��������Mg2+Ϊ�о�������֮���ڽ��ҵȾ����Mg2+���ھ��������м䣻
��4�����������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�������ӵ������������ж�δ�ɶԵ��ӣ�
��5����ͬ�ǽ���ԭ��֮���γɼ��Լ���ͬ�ַǽ���Ԫ��ԭ��֮���γɷǼ��Լ���Oԭ����Hԭ��֮����������Nԭ����Ni֮���γ���λ����
��6��CO2���Ӻ���2���Ҽ���2���м���H2���Ӻ���1���Ҽ�����ϲμӷ�Ӧ��CO2��H2���ʵ������㣮
��� �⣺��1��A��BΪ��������Ԫ�أ�AԪ�ص��ĵ����ܾ�����˵��Aԭ���������3�����ӣ���AΪAl��BԪ�ص��������ܾ�����˵��Bԭ���������2�����ӣ���BΪMg��Al�������ϼ�Ϊ+3�ۣ�ͬ����������ҵ縺�����ʵ縺��Al��Mg��
�ʴ�Ϊ��+3������
��2���������е������ȵ����ʷ����еĻ�ѧ��C-C��C-N��C-S�ļ��ܴ���������������ʹ��Щ�����ѣ��Ӷ��ƻ������ʷ���
��ɵ����ʵ���İ�����ΪH2N-CH2-COOH��-COOH��̼ԭ���ӻ������ĿΪ3��-CH2-�е�̼ԭ���ӻ������ĿΪ4��������̼ԭ���ӻ�������sp2��sp3��
�ʴ�Ϊ���������е������ȵ����ʷ����еĻ�ѧ��C-C��C-N��C-S�ļ��ܴ���������������ʹ��Щ�����ѣ��Ӷ��ƻ������ʷ��ӣ�sp2��sp3��
��3�����Ӿ�������Ӱ뾶ԽС���������Խ�࣬������Խ��������۷е�Խ�ߣ�����TiN��MgO��MgO��CaO���ɱ������ݿ�֪CaO��KCl����TiN��MgO��CaO��KCl��
MgO��NaCl�ṹ���ƣ�����ɫ����Mg2+�����ɫ��ΪO2-��������Mg2+Ϊ�о�������֮���ڽ��ҵȾ����Mg2+���ھ��������м䣬����һ��Mg2+��Χ�������ڽ��ҵȾ����Mg2+����Ϊ12��
�ʴ�Ϊ��TiN��MgO��CaO��KCl��12��
��4��V2O5��V����������ȫ��ʧȥ��ɼ���CrO2��Crʧȥ4�����ӣ����ӵ���������Ϊ2��δ�ɶԣ����Ӻ�δ�ɶԵ���Խ�࣬�����Խ�����ʺ���¼�����ŷ�ԭ�ϵ���CrO2��
�ʴ�Ϊ��CrO2��
��5��Cԭ����Hԭ�ӡ�Nԭ��֮���γɼ��Լ���̼ԭ��֮���γɷǼ��Լ���Oԭ����Hԭ��֮����������Nԭ����Ni֮���γ���λ����û�����Ӽ�����������
��ѡ��AC��
��6������1mol CH4���ɣ�����1molCO2��4molH2�μӷ�Ӧ����CO2���Ӻ���2���Ҽ���2���м���H2���Ӻ���1���Ҽ�������ѦҼ�Ϊ2mol+4mol=6mol�����Ѧм�Ϊ2mol��
�ʴ�Ϊ��6��2��
���� �����Ƕ����ʽṹ�����ʵĿ��飬��Ŀ�����Ϊ�ۺϣ��漰��������֪ʶ��ע����վ����ܵıȽϣ�ע�������������������������ϵ��

 ����ѧ����ϵ�д�
����ѧ����ϵ�д�| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
 ��
����2��д���ݺ͢�����Ԫ�ص�����������Ӧ��ˮ�������Ӧ�Ļ�ѧ����ʽAl��OH��3+KOH=KAlO2+2H2O��
��3���õ���ʽ��ʾ��Ԫ�����Ԫ���γɻ�����Ĺ���
 ��
����4���١��ڡ��ޡ�������Ԫ�ص�����������ˮ������������ǿ����HClO4���ѧʽ����
��5���١�����Ԫ���γɵĻ��������ʽ��
 ���û���������ں��м��Լ�������ԡ��Ǽ��ԡ�����
���û���������ں��м��Լ�������ԡ��Ǽ��ԡ�������6������֤���ߵĵ��ʱȢ�ĵ���������ǿ�����ӷ�Ӧ����ʽΪ2Br-+Cl2=Br2+2Cl-��
| A�� | S+O2�T2SO2����H=-269kJ/mol����Ӧ�ȣ� | |
| B�� | 2NO2��g���TO2��g��+2NO��g������H=+116.2kJ/mol����Ӧ�ȣ� | |
| C�� | C2H5OH��l��+3O2��g���T2CO2��g��+3H2O��g������H=-1367.0kJ/mol��ȼ���ȣ� | |
| D�� | NaOH��aq��+HCl��aq���TNaCl��aq��+H2O��l������H=+57.3kJ/mol���к��ȣ� |
A���ֱ����ʽ�ζ��ܡ���ʽ�ζ���ע��NaOH����Һ�ʹ��������0�̶�����2��3cm��
B�����ڵζ���Һ����0��0�̶����£����¶�����
C����NaOH����Һ�ζ����յ㣬���µζ���Һ��Ŀ̶ȣ�
D���ñ���Һ��ϴ��ʽ�ζ���2��3�Σ����������ϴ��ʽ�ζ���2��3�Σ�
E���ô��������ϴ��ƿ2��3�Σ�
F��������ʹ�ζ��ܼ��������Һ���ѵζ��̶ܹ��ã�
G���ζ��ܼ�©����ˮ��ϴ�������������ã�
H��ͨ����ʽ�ζ�������ƿע��20.00mL������ᣬ������2-3��ָʾ����
��1�������ӷ���ʽ��ʾ��ʵ���ԭ��CH3COOH+OH-=CH3COO-+H2O��
��2��������ʵ�鲽����ѡ����ȷ�IJ��������Ⱥ��������ʵ��˳��Ϊ��������ĸ��ʾ��G��D��A��F��B��H��C��
��3��ʵ���ҳ��õ����ָʾ���м��ȡ�ʯ��ͷ�̪����ѡ���ָʾ���Ƿ�̪������������ǡ����ȫ�к����ɴ����ƣ���ˮ��Һ�ʼ��ԣ�����ѡ���ڼ��Է�Χ��ɫ����ɫ�����жϵķ�̪��ָʾ���жϵζ��յ㣻�ζ��յ����������ɫ��Һ��dz��ɫ�Ұ�����ڲ���ɫ��
��4��ijͬѧʵ���õ������������
| �ζ����� ʵ������ | 1 | 2 | 3 | 4 | 5 |
| V��NaOH��/mL���������� | 0.00 | 0.20 | 0.00 | 0.10 | 0.05 |
| V��NaOH��/mL���ն����� | 15.75 | 15.20 | 14.98 | 15.12 | 15.05 |
��5���������в����Բⶨ�����Ӱ�죬�á�ƫ�͡�����ƫ�ߡ���Ӱ�족��գ�
�ٵζ�ǰ��ʽ�ζ��ܳ�����Һ���ζ�����촦������ƫ�ͣ�
�ڶ�ȡ����Һ���ʱ���ζ�ǰƽ�ӣ��ζ�������ƫ�ߣ�
| A�� |  | B�� | 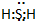 | C�� |  | D�� | 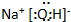 |
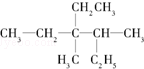 ��ϵͳ������������3��4-����-3-�һ����飮
��ϵͳ������������3��4-����-3-�һ����飮