��Ŀ����
9�� ��0.1320mol/L��HCl��Һ�ζ�δ֪Ũ�ȵ�NaOH��Һ��ʵ�����������ʾ��
��0.1320mol/L��HCl��Һ�ζ�δ֪Ũ�ȵ�NaOH��Һ��ʵ�����������ʾ��| ʵ���� | ����NaOH��Һ�����/mL | HCl��Һ�����/mL |
| 1 | 25.00 | 24.41 |
| 2 | 25.00 | 24.39 |
| 3 | 25.00 | 24.60 |
��1����ͼ�м�Ϊ��ʽ�ζ��ܣ���Ϊ��ʽ �ζ��ܣ����ʽ����ʽ����
��2��ʵ���У���Ҫ��ϴ�������ǣ����ᡢ��ʽ���ζ���
��3��ȡ����ҺNaOH��Һ25.00ml ����ƿ�У�ʹ�÷�̪��ָʾ�����ζ��յ���ж������Ǻ�ɫ��ȥ������Ӳ��ٳ��ֺ�ɫ
��4�����ζ�ǰ���ζ��ܼ�������ݣ��ζ���������ʧ����ʹ������ƫ��
���ƫ�ߡ���ƫ�͡������䡱����ͬ����������ʽ�ζ��ܶ���ʱ���ζ�ǰ���Ӷ������ζ�����ȷ��������������ƫ�ͣ�
��5��δ֪Ũ�ȵ�NaOH��Һ�����ʵ���Ũ��Ϊ0.1288 mol/l��
���� ��1�����������Ľṹ��������
��2�����ݵζ��ܲ��ô�ʢװ����Һ��ϴ���൱�ڸ���Һϡ�ͣ�ʢװ����Һ����ƿ����ʹ�ô���Һ��ϴ���������Һ�����ʵ���ƫ��
��3��������Һ��ɫ�仯�Ұ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣻
��4������c�����⣩=$\frac{c��������V������}{V�����⣩}$���жϣ�
��5���ȸ������ݵ���Ч�ԣ�Ȼ�����ƽ������V��HCl����Ȼ����ݹ�ϵʽ��NaOH��HCl���㼴�ɣ�
��� �⣺��1���ζ��ܵ��¶��Dz�����������������������Ϊ��ʽ�ζ��ܣ��ҵζ��ܵ��·����ܣ�������Ϊ��ʽ�ζ��ܣ�
�ʴ�Ϊ����ʽ����ʽ��
��2��ʵ���У���ʽ�ζ��ܡ���ʽ�ζ�����Ҫ�ô�ʢװ����Һ��ϴ������ᱻϡ�ͣ���ƿ�Ȳ���ʹ�ô���Һ��ϴ���������Һ�����ʵ���ƫ��
�ʴ�Ϊ�����ᡢ��ʽ���ζ��ܣ�
��3������Һ�еμ���ɫ�ķ�̪������ʹ��̪��죬�����÷�̪��ָʾ�����ζ��ﵽ�յ�������Ƿ�̪�ĺ�ɫ��ȥ�Ұ���Ӳ��ٳ��ֺ�ɫ��
�ʴ�Ϊ����ɫ��ȥ������Ӳ��ٳ��ֺ�ɫ��
��4�����ζ�ǰ���ζ��ܼ�������ݣ��ζ���������ʧ�����±�Һ�����ƫ����c�����⣩=$\frac{c��������V������}{V�����⣩}$������c�����⣩ƫ�ߣ�
��Һ����ʱ�����ζ�ǰ���ӣ��ζ����ӣ����±�Һ�����ƫС������c�����⣩=$\frac{c��������V������}{V�����⣩}$������c�����⣩ƫ�ͣ�
�ʴ�Ϊ��ƫ�ߣ�ƫ�ͣ�
��5���������ݿ�֪����ȥ���ϴ��22.60mL����V��HCl��=$\frac{24.41+24.39}{2}$mL=24.40mL���ɹ�ϵʽ��NaOH��HCl��֪��c��NaOH����25.00mL=0.1320mol/L��24.40mL ��ã�c��NaOH��=0.1288mol/L��
�ʴ�Ϊ��0.1288��
���� ������Ҫ�����˵ζ�������ע�������Լ����������ѶȲ����ر�ע�����ݵ�ȡ�ᣬ���ݿα�֪ʶ������ɣ�

| A�� | NaHBΪǿ����� | B�� | NaHB��Һ�У�c��Na+����c��HB-��+2c��B2-�� | ||
| C�� | H2BΪ������� | D�� | HB-�ĵ���̶�С��HB-��ˮ��̶� |
| A�� | HCL | B�� | HI | C�� | HF | D�� | H2SO4 |
| A�� | �Ҵ� | B�� | ������̼ | C�� | ���� | D�� | ��ȩ |
��1��ȷ��ȡ4.1g�ռ���Ʒ��������Ҫ������������ƽ��ҩ�ס��ձ���
��2������Ʒ���250mL����Һ����Ҫ���������ձ�����������250ml����ƿ����ͷ�ιܣ�
��3��ȡ10.00mL����Һ���ü�ʽ�ζ�����ȡ��
��4����0.2010mol•L-1������ζ������ռ���Һ���ζ�ʱ������ת��ʽ�ζ��ܵIJ������������ֲ�ͣ��ҡ����ƿ������ע����ƿ����Һ��ɫ�ı仯��ֱ���ζ��յ㣮
��5�������������ݣ���������ռ�Ĵ��ȣ�97.56%��
| �ζ����� | ����Һ��� ��mL�� | �����������mL�� | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 10.00 | 0.50 | 20.40 |
| �ڶ��� | 10.00 | 4.00 | 24.10 |
����ʽ�ζ���������ˮϴ����û���ñ�Һ��ϴ
�ڷ�ָ̪ʾ����ɫʱ����ֹͣ�ζ�
����ƿ������ˮϴ����û���ô���Һ��ϴ
�ܵζ�ǰ�ζ��ܼ�������ݣ��ζ���������ʧ
���յ����ʱ���ӣ���������������ȷ��
| A�� | ϡ���� | B�� | ϡ���� | C�� | ����ͭ | D�� | Ũ���� |
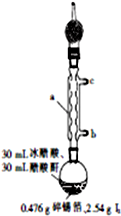 �ĵ⻯����SnI4����һ�ֳȺ�ɫ�ᾧ���۵�Ϊ144.5�棬�е�Ϊ364�棬��������ˮ�����ڴ��������ȷµȣ���ˮ��ˮ�⣬�����������Լ����л��ϳ��Լ���ʵ�����Ʊ��ĵ⻯������Ҫ�������£�
�ĵ⻯����SnI4����һ�ֳȺ�ɫ�ᾧ���۵�Ϊ144.5�棬�е�Ϊ364�棬��������ˮ�����ڴ��������ȷµȣ���ˮ��ˮ�⣬�����������Լ����л��ϳ��Լ���ʵ�����Ʊ��ĵ⻯������Ҫ�������£�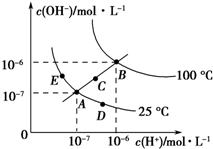 ��֪ˮ�ĵ���ƽ��������ͼ��ʾ���Իش��������⣺
��֪ˮ�ĵ���ƽ��������ͼ��ʾ���Իش��������⣺