��Ŀ����
 ��1����ѧ�̲���ͼʾ��NaCl����ṹ��������ά�ռ����쵽�������壮NiO��������������Ľṹ��NaCl��ͬ��Ni2+�����ڽ���O2-�˼����Ϊa��10-8 cm������NiO������ܶȣ���֪NiO��Ħ������Ϊ74.7g/mol��
��1����ѧ�̲���ͼʾ��NaCl����ṹ��������ά�ռ����쵽�������壮NiO��������������Ľṹ��NaCl��ͬ��Ni2+�����ڽ���O2-�˼����Ϊa��10-8 cm������NiO������ܶȣ���֪NiO��Ħ������Ϊ74.7g/mol����2����Ȼ�ĺ;����˹��Ʊ��ľ��嶼���ڸ���ȱ�ݣ�������ij��NiO�����оʹ�����ͼ��ʾ��ȱ�ݣ�һ��Ni2+��ȱ����������Ni2������Ni3+��ȡ�������������Գʵ����ԣ�����������Ni��O�ı�ֵȴ�����˱仯��ij��������Ʒ���ΪNi0.97O���Լ��㾧����Ni 3+��Ni 2+��������֮��
���㣺�����ļ���
ר�⣺
��������1������Ni2+�����ڽ�O2-�ĺ˼�������1cm3�к��е��������������������������ӵĹ�ϵ������Ӷ������ٸ����ܶȹ�ʽ���㣻
��2�������������������ӵ����ȼ���Ni3+��Ni2+�ĸ����ȣ�
��2�������������������ӵ����ȼ���Ni3+��Ni2+�ĸ����ȣ�
���
�⣺��1����NiO�����У�ÿ4��Ni������4��O 2-�������һ�������壬�����Ϊ��a��10-8 cm��3��ÿ��Ni����������8��С���������У�1molNiO������
��8 ������С�����壬����1molNiO�������Ϊ��a��10-8 cm��3��12.04��1023cm3=1.204��a3cm3�������ܶ�=
g?cm-3=
g?cm-3��
�ʴ�Ϊ��
g?cm-3��
��2����1mol Ni0.97O�к�Ni3+xmol��Ni2+Ϊ��0.97-x��mol�����ݾ����Գ����ԣ���֪ 3x+2����0.97-x��=2��1��
x=0.06mol Ni2+Ϊ��0.97-x��mol=0.91mol����������֮�� Ni3+��Ni2+=0.06��0.91=6��91��
�ʴ�Ϊ��6��91��
| 6.02��1023 |
| 4 |
| 74.7 |
| 1.204��a3 |
| 62.0 |
| a3 |
�ʴ�Ϊ��
| 62.0 |
| a3 |
��2����1mol Ni0.97O�к�Ni3+xmol��Ni2+Ϊ��0.97-x��mol�����ݾ����Գ����ԣ���֪ 3x+2����0.97-x��=2��1��
x=0.06mol Ni2+Ϊ��0.97-x��mol=0.91mol����������֮�� Ni3+��Ni2+=0.06��0.91=6��91��
�ʴ�Ϊ��6��91��
���������⿼���˾����ļ��㣬����ʱҪע���������ӵĹ�ϵ�����Ӷ������ٸ����ܶȹ�ʽ�������ܶȣ��Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
���и߾�������������ֵ������۶��ɵ��ǣ�������
A�� |
B��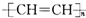 |
C�� |
D�� |
��֪25��ʱ��Ka��HF��=3.6��10-4��Ksp��CaF2��=1.46��10-10������1mL pH=2 ��HF��Һ�м���1m L 0.2mol?L-1 CaCl2��Һ��������˵������ȷ���ǣ�������
| A��Ka��HF����Ksp��CaF2���������¶Ⱥ�Ũ�ȵı仯���仯 |
| B��25��ʱpH=2 ��HF��Һ��HF����Ũ��Ϊ0.01mol?L-1 |
| C������HF��Һ��CaCl2��Һ��Ϻ�Ӧ�����˳��� |
| D��������Ӧ������ӹ�������Һ�������Ӵ��� |
����˵����ȷ���ǣ�������
| A��SiO2����������ˮ��ˮ��Һ��������? |
| B��SiO2�������������������������� |
| C��Na2SiO3��ˮ��Һ��Ϊ��ˮ���������京���Dz�����ˮ��Һ����˼? |
| D������ʱSiO2��Na2CO3��Ӧ�ų�CO2�����Ʋ����ķ�Ӧ֮һ�����ɵ�Na2SiO3�Dz����ɷ�֮һ |
���������ĸ�ƿ��Ϊ��ֹ��ʴ��װ����ǰ���루������
| A����ϴ�ɾ� | B������������� |
| C����ָ��� | D����ȥ���� |
���ɱ��ۻ�������ʱ�����и����з����仯���ǣ�������
| A�������ڻ�ѧ�� |
| B�����Ӽ���� |
| C�����ӹ��� |
| D�����Ӽ������� |
A��B���Ƕ�����Ԫ�أ�ԭ�Ӱ뾶B��A�����ǿ��γɻ�����AB2���ɴ˿��Եó�����ȷ�ж��ǣ�������
| A��ԭ��������A��B |
| B��A��B������ͬһ���� |
| C��A�����ڵ�2���ڢ�A�� |
| D��A�϶��ǽ���Ԫ�� |
