��Ŀ����
1����������ʵ��������������õ��Ľ�����ȷ���ǣ�������| ѡ�� | ʵ����������� | ���� |
| A | ��ijʳ����Һ�еμӵ�����Һ����Һ��ɫû�б�Ϊ��ɫ | ���Ǽӵ�ʳ�� |
| B | ij��Һ�м��������ữ��Ba��NO3��2��Һ���������ٵμ�AgNO3��Һ���а�ɫ�������� | ��Һ��һ������Cl- |
| C | �����£���pH��ֽ�ֱ��0.100mol•L-1 CH3COONa��Һ��0.100mol•L-1 Na2S��Һ��pH������pH�� | ���ԣ�H2S��CH3COOH |
| D | ������X�ֱ�ͨ����ˮ�����Ը��������Һ������Һ����ɫ | ����Xһ����C2H4 |
| A�� | A | B�� | B | C�� | C | D�� | D |
���� A��ʹ���۱������ǵⵥ�ʵ����ʣ����ǵ�Ԫ�ص����ʣ�
B�����������ữ��Ba��NO3��2��Һ���ų��˸������ӣ��ٵμ�AgNO3��Һ�����ɵİ�ɫ����ֻ��ΪAgCl��
C��Na2S��Ӧ����Ϊ��������ӣ�Ӧ�ó̶ȵ�Ũ�ȵ�NaHS��Һ��pH��
D������̼̼˫����̼̼�������л��������ʹ��ˮ�����Ը��������Һ��ɫ��
��� �⣺A������ʳ���еĵ�Ϊ�������еĵ�Ԫ�أ����ǵⵥ�ʣ�������ijʳ����Һ�еμӵ�����Һ����Һ��ɫ�����Ϊ��ɫ����A����
B��ij��Һ�м��������ữ��Ba��NO3��2��Һ���������ų�������������ӡ���������ӵȸ��ţ��ٵμ�AgNO3��Һ���а�ɫ�������ɣ��ó���һ��ΪAgCl��֤����Һ��һ������Cl-����B��ȷ��
C�������£���pH��ֽ�ֱ��0.100mol•L-1 CH3COONa��Һ��0.100mol•L-1 Na2S��Һ��pH������pH������ Na2S��Ӧ����ΪHS-��ֻ��֤��CH3COOH�����Դ���HS-���ӣ����Ƚ�H2S��CH3COOH�����ԣ���C����
D��������X�ֱ�ͨ����ˮ�����Ը��������Һ������Һ����ɫ��֤��֤��X�к��в����ͼ���X��һ����C2H4������Ϊ��������̼̼˫����̼̼�������л����D����
��ѡB��
���� ���⿼���˻�ѧʵ�鷽�������ۣ���Ŀ�Ѷ��еȣ��漰���Ӽ��顢����ǿ���Ƚϡ��л�������֪ʶ����ȷ����Ԫ�ؼ��仯�������ʼ��ɽ���������������ѧ���ķ������������Ӧ��������

 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�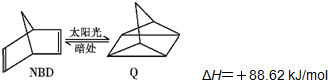 �������й�˵����ȷ���ǣ�������
�������й�˵����ȷ���ǣ�������| A�� | NBD��Q����Ϊͬ���칹�� | |
| B�� | ����NBD�ķ���ʽΪC7H7 | |
| C�� | ����Q��һ��ȡ������2�� | |
| D�� | һ�������£�1molNBD������2molBr2�����ӳɷ�Ӧ |
| A�� | ����ɫ��ѧ���ĺ�����Ӧ�û�ѧԭ���Ի�����Ⱦ�������� | |
| B�� | �������������е����ʵ�����ǽ���Ԫ�ص���ɫ��Ӧ | |
| C�� | �����ʫ�ơ�������¯�����̡�������д����������� | |
| D�� | ú����������Һ������������仯���̣�����ת��Ϊ�����Դ |
| A | B | C | D | |
| ʵ��װ�� | 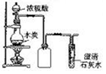 | 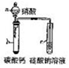 | 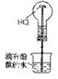 | 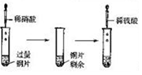 |
| ʵ������ | �Թ��г��ְ�ɫ���� | cװ���г��ְ�ɫ���� | ��ɫ��Ȫ | �μ�ϡ���ᡢͭƬ�ܽ� |
| ʵ����� | ��ɫ����CaCO3 | �ǽ����ԣ�N��C��Si | HCl��������ˮ | ����������NO3-������ǿ |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ���л���X�ļ���ʽΪ
���л���X�ļ���ʽΪ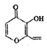 �����й����л���X��˵������ȷ���ǣ�������
�����й����л���X��˵������ȷ���ǣ�������| A�� | X�ķ���ʽΪC7H8O3 | |
| B�� | X����������������ͬ�Ĺ����� | |
| C�� | X����������KMnO4��Һ�����ӳɷ�Ӧ����ɫ | |
| D�� | �л���Y��X��ͬ���칹�壬����̼�ᵪ����Һ��Ӧ�Һ��б�������Y�Ľṹ��3�� |
| A�� | ��������ɫ���������۵���Խϵ͡��ܶȱ�ˮС�Ľ������� | |
| B�� | ���������Ż�ʱ������ɳ������ | |
| C�� | �������ڿ����г��ڷ��ã����ձ�Ϊ̼���� | |
| D�� | ���������ǰ�ɫ���� |
| ��ѡ�� | ��ʵ����� | ��ʵ������ | ������ |
| ��A | ����ij��Һ�м��������ữ��BaCl2��Һ | ���ɰ�ɫ���� | ����Һ�в�һ������SO42- |
| ��B | FeCl2��Һ�У��μ�KSCN��Һ����ˮ | �μ�KSCN��Һ�������Ա仯���μ���ˮ����Һ��ΪѪ��ɫ | �����ԣ�Br2��Fe3+ |
| ��C | ȡ���õ�Na2O2��ĩ�������еμӹ��������� | ������ɫ���� | ��Na2O2û�б��� |
| ��D | ��һƬ�������ھƾ������������� | �����ۻ��������� | ��������������Al2O3��Ĥ����Al2O3�۵����Al |
| A�� | A | B�� | B | C�� | C | D�� | D |