��Ŀ����
16���ߴ�������[��ѧʽ��Sr��NO3��2]���������źŵơ���ѧ�����ȣ���1����ҵ���������г���������ơ����ᱵ�����ʣ���������ƿ�����Ũ���ᣬ�������ȡ����ᱵ������Ũ���ᣮ���������Ϣ�����������ᴿ�����ȵ�����ʵ�鲽�裺
��ȡ�����ʵ���������Ʒ�������м���ŨHNO3�����裮
�ڹ��ˣ�����ŨHNO3ϴ��������
�۽���������ˮ�У����Թ�������ʹBa2+���������ú�����£�N2H4�����������ỹԭ������pH=7��8�����ˣ�
�ܽ���Һ���������pH=2��3������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӣ�
�ݽ��õ���Sr��NO3��2•2H2O������100�������¸���õ��ߴ������ȣ�
��2��Sr��NO3��2�����ֽ⣬����Sr��NO2��2��O2����500��ʱSr��NO2��2��һ���ֽ�����SrO���������ȡһ��������Sr��NO2��2��Sr��NO3��2��Ʒ����������ȫ�ֽ⣬�õ�5.20g SrO�����5.08g������壮�������Ʒ��Sr��NO3��2������������д��������̣���
���� ��1��������Ϣ������ƿ�����Ũ���ᣬ�������ȡ����ᱵ������Ũ���ᡱ����ȷ���ᴿ�����ȵIJ��裺
�ٽ���Ʒ����ŨHNO3�У����裻
�ڹ��ˣ�����ŨHNO3ϴ��������ȡ���������������ȡ����ᱵ����
�ܽ������е����ᱵ���������ȥ��
��ȡ��Һ���������ȣ�����ͨ������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӱ�ɵõ��ϴ��������Ⱦ��壻
��2�����������غ㶨�ɼ�����Ʒ����������Srԭ���غ㡢Sr��NO2��2��Sr��NO3��2���������з��̼���������ʵ���������������Ʒ��Sr��NO3��2������������
��� �⣺��1��������Ϣ������ƿ�����Ũ���ᣬ�������ȡ����ᱵ������Ũ���ᡱ����ȷ���ᴿ�����ȵIJ��裺
�ٽ���Ʒ����ŨHNO3�У����裻
�ڹ��ˣ�����ŨHNO3ϴ��������ȡ���������������ȡ����ᱵ����
�ܽ������е����ᱵ���������ȥ��
��ȡ��Һ���������ȣ�����ͨ������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӱ�ɵõ��ϴ��������Ⱦ��壬
�ʴ�Ϊ���������м���ŨHNO3���ڹ��ˣ�����ŨHNO3ϴ��������������Ũ������ȴ�ᾧ��
��2��m����Ʒ��=5.20g+5.08g=10.28g
n��SrO��=$\frac{5.20g}{104g/mol}$=0.05mol
n[Sr��NO3��2]��212g•mol-1+n[Sr��NO2��2]��180g•mol-1=10.28g
n[Sr��NO3��2]+n[Sr��NO2��2]=0.05mol
���n[Sr��NO3��2]=0.04mol��n[Sr��NO2��2]=0.01mol
�ʸ���Ʒ��Sr��NO3��2����������Ϊ$\frac{0.04mol��212g/mol}{10.28g}$��100%=82.49%��
�𣺸���Ʒ��Sr��NO3��2����������Ϊ82.49%��
���� ���⿼�黯ѧʵ�鷽�����������ļ���ȣ��Ƕ�ѧ���ۺ������Ŀ��飬ע�⣨2�����غ㷨��Ӧ�ã�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | �����¶ȣ�v���������v���棩��С | |
| B�� | ���º��ݣ�ƽ��ǰ����������ܶȱ��ֲ��� | |
| C�� | ���º�ѹ������N2��ƽ�ⲻ�ƶ� | |
| D�� | ���º��ݣ�����O2��O2��ת�������� |
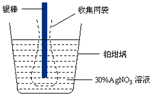 ��ȷ�������������ڶ��Ե缫���ԶƲ���ʽ�����Ľ�������������ȷ����������ͨ�����صĵ�����ʵ�ʲ����г��õ��������ƽṹ��ͼ��ʾ������˵������ȷ���ǣ�������
��ȷ�������������ڶ��Ե缫���ԶƲ���ʽ�����Ľ�������������ȷ����������ͨ�����صĵ�����ʵ�ʲ����г��õ��������ƽṹ��ͼ��ʾ������˵������ȷ���ǣ�������| A�� | �������е�����Ӧ���Դ�������������������Ϸ����ĵ缫��Ӧ�ǣ�Ag++e-=Ag | |
| B�� | ʵ���У�Ϊ�˱������ܽ�����п��ܲ����Ľ����������������������²������������缫��������һ���ռ���������û���ռ����������������ƫ�� | |
| C�� | ��֪ÿĦ�����ӵĵ���Ϊ965 00 C�����������ǰ�������������仯ֵΪ108.0 mg�����������ͨ�����صĵ���Ϊ96.5 C | |
| D�� | ��Ҫ�ⶨ��⾫��ͭʱͨ���ĵ������ɽ������������е������������صĴ�ͭ�缫���� |
| A�� | �ù㷺pH��ֽ���0.05mol•L-1 H2SO4��Һ��pH=1 | |
| B�� | ����ɫ��Ӧʵ��ʱ������ɫ�ܲ������۲쵽������ɫΪ��ɫ��֤����������һ��Ϊ���� | |
| C�� | �ζ������У��۾�Ҫʼ��ע����ƿ��Һ�����ɫ�仯 | |
| D�� | ����FeSO4��Һʱ���Ƚ�FeSO4�����ܽ���ϡ�����У�Ȼ��ϡ�͵�����Ũ�ȣ�����ټ����������� |
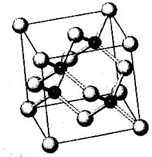 N��P��As��Ga��Cr��Ԫ�ػ���������࣬������Ҫ���о���ֵ��Ӧ�ü�ֵ����ش��������⣺
N��P��As��Ga��Cr��Ԫ�ػ���������࣬������Ҫ���о���ֵ��Ӧ�ü�ֵ����ش��������⣺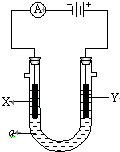 ���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ������ֱ����Դ��������ش��������⣺
���ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�ã���ͼ��ʾһ�����أ�װ�е��Һa��X��Y������缫�壬ͨ������ֱ����Դ��������ش��������⣺