��Ŀ����
19����ͼ��һЩ�����ĵ��ʡ�������֮���ת����ϵͼ����Щ��Ӧ�еIJ������ʱ���ȥ�����³�ѹ�£�BΪ����ɫ��ĩ��A��CΪ�������ʣ�IΪ����ɫ���壮��ش��������⣺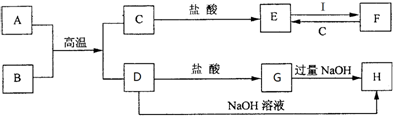
��1��B�Ļ�ѧʽFe2O3
��2��I�ĵ���ʽ

��3��A+B��C+D�Ļ�ѧ����ʽFe2O3+2 Al $\frac{\underline{\;����\;}}{\;}$ Al2O3+2Fe
��4��F+C��E�����ӷ���ʽ2Fe3++Fe�T3Fe2+��
���� BΪ����ɫ��ĩ��A��CΪ�������ʣ���A��B��Ӧ�õ�C��D��Ӧ�����ȷ�Ӧ��BΪFe2O3��AΪAl��CΪFe��DΪAl2O3��IΪ����ɫ���壬��IΪCl2����ת����ϵ��֪��EΪFeCl2��FΪFeCl3��GΪAlCl3��HΪNaAlO2���ݴ˽��
��� �⣺BΪ����ɫ��ĩ��A��CΪ�������ʣ���A��B��Ӧ�õ�C��D��Ӧ�����ȷ�Ӧ��BΪFe2O3��AΪAl��CΪFe��DΪAl2O3��IΪ����ɫ���壬��IΪCl2����ת����ϵ��֪��EΪFeCl2��FΪFeCl3��GΪAlCl3��HΪNaAlO2��
��1��B�Ļ�ѧʽΪ��Fe2O3���ʴ�Ϊ��Fe2O3��
��2��IΪCl2������ʽΪ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��A+B��C+D�Ļ�ѧ����ʽ��Fe2O3+2 Al $\frac{\underline{\;����\;}}{\;}$ Al2O3+2Fe���ʴ�Ϊ��Fe2O3+2 Al $\frac{\underline{\;����\;}}{\;}$ Al2O3+2Fe��
��4��F+C��E�����ӷ���ʽ��2Fe3++Fe�T3Fe2+���ʴ�Ϊ��2Fe3++Fe�T3Fe2+��
���� ���⿼��������ƶϣ����ʵ���ɫ���ƶ�ͻ�ƿڣ���������Ԫ�ػ��������ʣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
9��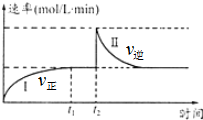 ��һ����������ɱ���ܱ������з������·�Ӧ��A��g��+B��g��?2C��g����H��0��t1ʱ�̴ﵽƽ�����t2ʱ�̸ı�ijһ��������ƽ�⣬�䷴Ӧ������ͼ������˵���д�����ǣ�
��һ����������ɱ���ܱ������з������·�Ӧ��A��g��+B��g��?2C��g����H��0��t1ʱ�̴ﵽƽ�����t2ʱ�̸ı�ijһ��������ƽ�⣬�䷴Ӧ������ͼ������˵���д�����ǣ�
 ��һ����������ɱ���ܱ������з������·�Ӧ��A��g��+B��g��?2C��g����H��0��t1ʱ�̴ﵽƽ�����t2ʱ�̸ı�ijһ��������ƽ�⣬�䷴Ӧ������ͼ������˵���д�����ǣ�
��һ����������ɱ���ܱ������з������·�Ӧ��A��g��+B��g��?2C��g����H��0��t1ʱ�̴ﵽƽ�����t2ʱ�̸ı�ijһ��������ƽ�⣬�䷴Ӧ������ͼ������˵���д�����ǣ�| A�� | 0��t2ʱ��v����v�� | |
| B�� | t2ʱ�̸ı�����������ǼӴ��� | |
| C�� | �������̴ﵽƽ��ʱ��A����������� | |
| D�� | �������̴ﵽƽ��ʱ��ƽ�ⳣ��I���� |
7�����л�ѧ�����ʾ��ȷ���ǣ�������
| A�� | �Ҵ��Ľṹ��ʽ��C2H6O | |
| B�� | �����ӵĽṹʾ��ͼ��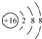 | |
| C�� | �����ĵ���ʽ��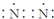 | |
| D�� | ���������ĵ��뷽��ʽ��Ba��OH��2�TBa2++��OH-��2 |
14��������Ԫ��X��Y��Z��ԭ��������������Yԭ�ӵ������������Ǵ�����3����XY2Ϊ����ɫ���壬Z����ɫ��Ӧ�ʻ�ɫ������������ȷ���ǣ�������
| A�� | ԭ�Ӱ뾶�Ĵ�С˳��r��X����r��Y����r��Z�� | |
| B�� | ��̬�⻯������ȶ��ԣ�X��Y | |
| C�� | Ԫ��X������������Ӧ��ˮ������ǿ�� | |
| D�� | ������XY��Z2Y��Z2Y2�л�ѧ����������ͬ |
11���������ӷ���ʽ��ȷ���ǣ�������
| A�� | �����£�����Ƭ��������Ũ�����У�Fe+4H++2NO3-�TFe2++2NO2��+2H2O | |
| B�� | ����������Һ��ͨ������SO2��Ca2++2ClO-+SO2+H2O�TCaSO3��+2HClO | |
| C�� | ����Һ��FeI2������3��4��Ӧ��2Fe2++6I-+4Cl2�T2Fe3++3I2+8Cl- | |
| D�� | ��Ba��OH��2��Һ�мӹ�����NH4HSO4��Һ��Ba2++2OH-+SO42-+H++NH4+�TBaSO4��+H2O+NH3•H2O |
8����NAΪ�����ӵ�������ֵ�������й�������ȷ���ǣ�������
| A�� | 28g��ϩ�������õ��Ӷ���ĿΪ4NA | |
| B�� | 1mol C4H10�����й��ۼ�����Ϊ13NA | |
| C�� | 1 mol�������ĵ�������Ϊ7NA | |
| D�� | ��״���£�11.2 L��������������Ϊ0.5 NA |
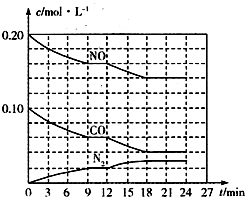 Ϊ��Ч�������������ػ�����ȡ��ʩ���ƴ�����������Ч���ƿ����е������̼����������������Ե���Ϊ��Ҫ��
Ϊ��Ч�������������ػ�����ȡ��ʩ���ƴ�����������Ч���ƿ����е������̼����������������Ե���Ϊ��Ҫ��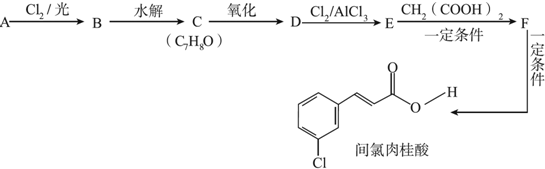
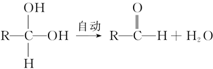
 +Cl2$\stackrel{����}{��}$
+Cl2$\stackrel{����}{��}$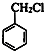 +HCl��
+HCl��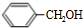 +CH3COOH$��_{��}^{Ũ����}$
+CH3COOH$��_{��}^{Ũ����}$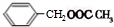 +H2O��
+H2O��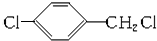 ��
��