��Ŀ����
 ��A�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ����ֻ��ϼۣ�����A��Ԫ�صĻ��������о�����������������Ҫ��;����ش��������⣺
��A�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ����ֻ��ϼۣ�����A��Ԫ�صĻ��������о�����������������Ҫ��;����ش��������⣺��1����Ԫ�����γɷ�����������д��һ����CO2�ȵ��ӵĻ�����
��2����Na2O��SiO2��P2O5���������ﰴ�۷е��ɸߵ���˳������
��3��ԭ�ӵĵ�һ��������ָ��̬�����Ի�̬ԭ��ʧȥһ������ת��Ϊ��̬��̬����������Ҫ�����������O��S��Seԭ�ӵĵ�һ�������ɴ�С��˳��Ϊ
��4��Seԭ�ӻ�̬������ӵ��Ų�ʽΪ
��5��SO32-��������ԭ�ӵ��ӻ���ʽ
��6��ij����Ԫ��A���������������������������ϡ������������������ľ����ṹ��ͼ��ʾ�����������Ļ�ѧʽΪ
���㣺�����ļ���,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�жϼ��ӻ����ӵĹ���,���ȵ���ԭ������Ӧ��,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺Ԫ����������Ԫ�����ڱ�ר��,��ѧ���뾧��ṹ
��������1�����ݵȵ���ԭ���жϣ�
��2�����ݾ��������жϣ�
��3�����ݵ�һ�����ܵݱ�����жϣ�
��4������ԭ�������������������жϣ�
��5�����ݼۼ����������жϣ�
��6�����ݾ�̯�����м��㣮
��2�����ݾ��������жϣ�
��3�����ݵ�һ�����ܵݱ�����жϣ�
��4������ԭ�������������������жϣ�
��5�����ݼۼ����������жϣ�
��6�����ݾ�̯�����м��㣮
���
�⣺��1���ȵ�����Ϊԭ������ͬ�Ҽ۵�������ͬ��ԭ���ţ����Ը���S��Oͬ�����뵽��CS2������ͬ��������Ԫ���뵽N2O���ʴ�Ϊ��CS2��N2O��
��2��SiO2Ϊԭ�Ӿ��壬�۷е�ȡ���ڹ��ۼ����ܣ��۷е�ܸߣ�Na2OΪ���Ӿ��壬�۷е�ȡ�����侧���ܣ��۷е�ϸߣ�P2O5Ϊ���Ӿ��壬�۷е�ȡ���ڷ��Ӽ����������۷е�ϵͣ��ʴ�Ϊ��SiO2��Na2O��P2O5��
��3��ͬ����Ԫ�ش��ϵ��£�ԭ�Ӱ뾶Խ��Խ��ԭ�Ӻ˶Ժ�����ӵ�������Խ��Խ������һ��������С���ʴ�Ϊ��O��S��Se��
��4��Seԭ������Ϊ34�������������ԭ������������ԭ���ͺ��ع���д��Ϊ[Ar]3d104s24p4�����߾��ɷ��ӹ��ɣ��۷е�ȡ���ڷ��Ӽ������������Ӽ�������ȡ������Է���������H2Se����Է���������H2S�����H2Se���Ӽ�������ǿ��H2S��
�ʴ�Ϊ��[Ar]3d104s24p4��H2Se���Ӽ�������ǿ��H2S��
��5����SO32-���Ӽ۵��Ӷ���Ϊ
=4���γ�4���µ��ӻ������������sp3�ӻ����µ��Ӷ���=�۵��Ӷ���-��λԭ����=4-3=1������һ�Թ¶Ե��ӣ�ռ��һ���ӻ��������S-O�����ų����ã�S��O���������Σ��ʴ�Ϊ��sp3��������
��6���ھ����ṹ�У�����ɫ���������λ�����ڣ���4��������ɫ���������8��λ�ڶ����6��λ�����ģ�����Ϊ8��
+6��
=4����ѧʽ��дΪAO���ʴ�Ϊ��AO��
��2��SiO2Ϊԭ�Ӿ��壬�۷е�ȡ���ڹ��ۼ����ܣ��۷е�ܸߣ�Na2OΪ���Ӿ��壬�۷е�ȡ�����侧���ܣ��۷е�ϸߣ�P2O5Ϊ���Ӿ��壬�۷е�ȡ���ڷ��Ӽ����������۷е�ϵͣ��ʴ�Ϊ��SiO2��Na2O��P2O5��
��3��ͬ����Ԫ�ش��ϵ��£�ԭ�Ӱ뾶Խ��Խ��ԭ�Ӻ˶Ժ�����ӵ�������Խ��Խ������һ��������С���ʴ�Ϊ��O��S��Se��
��4��Seԭ������Ϊ34�������������ԭ������������ԭ���ͺ��ع���д��Ϊ[Ar]3d104s24p4�����߾��ɷ��ӹ��ɣ��۷е�ȡ���ڷ��Ӽ������������Ӽ�������ȡ������Է���������H2Se����Է���������H2S�����H2Se���Ӽ�������ǿ��H2S��
�ʴ�Ϊ��[Ar]3d104s24p4��H2Se���Ӽ�������ǿ��H2S��
��5����SO32-���Ӽ۵��Ӷ���Ϊ
| 6+2 |
| 2 |
��6���ھ����ṹ�У�����ɫ���������λ�����ڣ���4��������ɫ���������8��λ�ڶ����6��λ�����ģ�����Ϊ8��
| 1 |
| 8 |
| 1 |
| 2 |
�����������ۺϿ������ʽṹ�����ʣ��漰��������Ų����ɡ������ܡ��ӻ����ۡ����ӽṹ�������ṹ�ȣ������жϿռ乹�����׳�����

��ϰ��ϵ�д�
 �������ϵ�д�
�������ϵ�д�
�����Ŀ
���з�Ӧ�У�������Ϊ���������ǣ�������
| A��Zn+2HCl�TZnCl2+H2�� | ||||
| B��FeS+2HCl�TFeCl2+H2S�� | ||||
| C��NH3+HCl�TNH4Cl | ||||
D��MnO2+4HCl
|
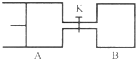 ��ͼ��ʾ����A��B�ж�����2mol X��2mol Y����ʼʱVA=0.8aL��VB=aL������ͬ�¶Ⱥ��д����������£��������и��Է������з�ӦX��g��+Y��g��?2Z��g��+W��g����H��0���ﵽƽ��ʱ��VA=0.9aL��������˵��������ǣ�������
��ͼ��ʾ����A��B�ж�����2mol X��2mol Y����ʼʱVA=0.8aL��VB=aL������ͬ�¶Ⱥ��д����������£��������и��Է������з�ӦX��g��+Y��g��?2Z��g��+W��g����H��0���ﵽƽ��ʱ��VA=0.9aL��������˵��������ǣ�������| A���������дﵽƽ���ʱ��A��B |
| B��A������X��ת����Ϊ25%���ұ�B������X��ת����С |
| C����Kһ��ʱ��ﵽƽ��ʱ��A�����Ϊ0.8aL����ͨ��������������ƣ� |
| D����K�ﵽ��ƽ�������B�����¶ȣ�A�������һ����С |
ij��ɫ����������Һ���ܴ��������һ�������ǣ�������
| A��Na+��SO42-��Al3+��Cl- |
| B��Na+��HCO3-��K+��SO42- |
| C��MnO4-��K+��SO42-��Na+ |
| D��Fe3+��Na+��SO42-��Cl- |

 ����ͼ��ʾ��װ���У���ֱͨ����5minʱ��ͭ�缫��������2.16g���Իش�
����ͼ��ʾ��װ���У���ֱͨ����5minʱ��ͭ�缫��������2.16g���Իش�
 ���������仯��������Դ�����ϵȹ�ҵ�ж�����Ҫ��;��
���������仯��������Դ�����ϵȹ�ҵ�ж�����Ҫ��;��