��Ŀ����
16��CH3COOH��CH3COONa�����Һ�д�������ƽ�⣺CH3COOH��aq��?CH3COO-��aq��+H+��aq����H=+1.3kJ•mol-1��
CH3COO-��aq��+H2O��l��?CH3COOH��aq��+OH-��aq����H=+56.0kJ•moL-1��
���й������ʵ���Ũ�Ⱦ�Ϊ0.1mol•L-1��CH3COOH��CH3COONa�Ļ�ϵ�˵����ȷ���ǣ�������
| A�� | H+��aq��+OH-��aq���TH2O��l����H=+57.3kJ•mol-1 | |
| B�� | ������������ˮ��ƽ��ٺ͢ھ������ƶ� | |
| C�� | ��Һ��c��CH3COOH����c��Na+����c��CH3COO-����c��OH-����c��H+�� | |
| D�� | ��������CaCl2��Һ��������Һ�У�c��CH3COOH��+2c��OH-��=c��CH3COO-�� |
���� A���кͷ�ӦΪ���ȷ�Ӧ��
B����ˮϡ�ʹٽ������ˮ�⣻
C��CH3COOH��CH3COONa�Ļ����Һ�����ԣ�
D�����ݵ���غ�������غ������
��� �⣺A���кͷ�ӦΪ���ȷ�Ӧ����H+��aq��+OH-��aq���TH2O��l����H=-57.3kJ•mol-1����A����
B����ˮϡ�ʹٽ�����ĵ��룬Ҳ�ٽ���������ӵ�ˮ�⣬����ƽ��ٺ͢ھ������ƶ�����B��ȷ��
C��CH3COOH��CH3COONa�Ļ����Һ�����ԣ�����ĵ���̶ȴ��ڴ�������ӵ�ˮ��̶ȣ�����Һ����Ũ�ȴ�С��ϵΪ��c��CH3COO-����c��Na+����c��CH3COOH����c��H+����c��OH-������C����
D����Һ�еĵ���غ�Ϊ��c��Na+��+c��H+��+c��Cl-��=c��CH3COO-��+c��OH-��+2c��Ca2+��������c��Cl-��=2c��Ca2+������c��Na+��+c��H+��=c��CH3COO-��+c��OH-������Һ�������غ�Ϊ��c��CH3COOH��+c��CH3COO-��=2c��Na+������c��CH3COO-��+2c��OH-��=c��CH3COOH��+2c��H+������D����
��ѡB��
���� ���⿼������Һ������Ũ�ȴ�С�ıȽϣ���Ŀ�Ѷ��еȣ����ݵ���غ�ȷ����Һ������Լ���ĵ���̶Ⱥ��������ˮ��̶���Դ�С���ٽ�ϵ���غ�������غ���ע�����õ���غ������

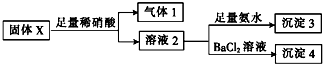
��������ʵ�飬����˵����ȷ���ǣ�������
| A�� | ����1һ����NO���ܺ�CO2 | |
| B�� | ����3һ����Mg��OH��2һ������Al��OH��3 | |
| C�� | ����4����ΪBaCO3��BaSO3�������� | |
| D�� | �����ĩX��һ����MgCl2��Na2SO3��������KAlO2 |
| A�� | �������Ũ�ȵ�Ba��OH��2ϡ��Һ��NH4HCO3ϡ��Һ��ϣ�Ba2++2OH-+NH4++HCO3-=BaCO3��+NH3•H2O+H2O | |
| B�� | FeI2��Һ��ͨ�����Cl2��2Fe2++2I-+2Cl2�T2Fe3++I2+4Cl- | |
| C�� | ������SO2ͨ��NaOH��Һ�У�SO2+2OH-�TSO32-+H2O | |
| D�� | ��CH3COOH�ܽ�CaCO3��CaCO3+2H+�TCa2++H2O+CO2�� |
| A�� | һ����S2-��SO32-��Na+ | B�� | һ��û��Ag+��Al3+ | ||
| C�� | ����ֻ��Na+��S2-��CO32- | D�� | һ����Na+��S2-��NO3 |
| A�� | 118��Ԫ�ص����ԭ������Ϊ293 | |
| B�� | 118��Ԫ�ص�һ��ͬλ��${\;}_{118}^{293}$Pb��������Ϊ293 | |
| C�� | 118��Ԫ�ص�һ��ͬλ��${\;}_{118}^{293}$Pb��������Ϊ293 | |
| D�� | 118��Ԫ��ӦΪ���ڱ��е�8���ڵ�Ԫ�� |
| A�� | Sԭ�ӵĵ��Ӳ�����Clԭ�ӵĵ��Ӳ����� | |
| B�� | Cl���⻯��ķе��S���⻯��е�� | |
| C�� | Cl����̬�⻯���S����̬�⻯���ȶ� | |
| D�� | Cl��ʧ����������Sǿ |
| A�� | ī�ӣ����ʵķָ����������� | B�� | ��ķ�������Ѹ����ģ�� | ||
| C�� | ���ӿ����أ��ŵ�ԭ���� | D�� | �����ն���ԭ�ӽṹ������ģ�� |
 ����
���� ��
��
