��Ŀ����
��15�֣�ij��ѧ�о�С��̽����ˮ������������K3[Fe(C2O4)3]���ȷֽ�IJ����������������������������֤������ͼװ�ý���ʵ�飨�г���������ȥ����
��1�����Ӻ�װ�ú���ҩƷǰ��Ӧ�ý��е�ʵ�������________________(���������)��
��2��ʵ�鿪ʼʱ����ͨ�뵪������һ��ʱ���Ժ��ȷ�Ӧ��A��E ������Ӧ��ȫ��ֹͣ���ȣ��Լ���ͨ������ֱ����Ӧ����ȴ��ֹͣ����ǰ�Ƿ���Ҫ�ȶϿ�A��B�����Ӵ���E��F�����Ӵ���Ϊʲô��_______________________________________________________________��
��3��ʵ���й۲쵽B�г���ʯ��ˮ����ǣ���˵������������� ��д��ѧʽ����
��֤���ֽ��������CO�������ɵ�ʵ�������� ��
��4����С��ͬѧ�������Ϻ��֪����������в�����+3��FeԪ�أ�����������ֻ��K2CO3��
�� ��֤��������м�Ԫ�ش��ڵķ����� �������� ��
�� �о�С��Ϊ̽����Ԫ�صĴ�����ʽ��ȡA�г�ַ�Ӧ�����ù������Թ��У�������������ˮ�����ˡ�ϴ�ӡ�����õ���ɫ��ĩ��ȡ4.4g��ɫ��ĩ��������ϡ�����У��ռ�����״����NO����1.12Lͨ�������жϺ�ɫ��ĩ�ijɷ�Ϊ ��д��ѧʽ����д����ɫ��ĩ��������ϡ���ᷢ����Ӧ�����ӷ���ʽ ��
��1�����װ�õ������ԣ�1�֣�
��2������Ҫ��1�֣� ��ΪN2����ͨ�룬B��Fƿ��Һ���ᵹ������1�֣�
��3��CO2��1�֣� E�к�ɫ�����ɺ�ɫ��F����Һ����ǣ�2�֣�
��4������ɫ��Ӧ��2�֣� ����ɫ���ܲ����۲쵽��ɫ���棨1�֣�
��FeO ��Fe 3FeO +10H+ +NO3��=3Fe3++NO��+5H2O
Fe+4H+ +NO3��=Fe3++NO��+2H2O ��ÿ��2�֣�
����


 2C��g�������еķ�ӦʽΪ��A��g��+B��g��
2C��g�������еķ�ӦʽΪ��A��g��+B��g�� 2C��g����
2C��g���� 2C��g�������еķ�ӦʽΪ��A��g��+B��g��
2C��g�������еķ�ӦʽΪ��A��g��+B��g�� 2C��g��
2C��g��
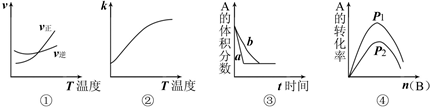


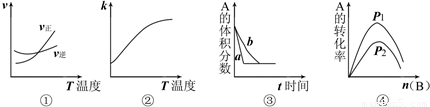
 2C��g�������еķ�ӦʽΪ��A��g��+B��g��
2C��g�������еķ�ӦʽΪ��A��g��+B��g�� 2C��g����
2C��g���� 2C��g�������еķ�ӦʽΪ��A��g��+B��g��
2C��g�������еķ�ӦʽΪ��A��g��+B��g�� 2C��g��
2C��g��