��Ŀ����
������M��һ��ҽ�ø߷��Ӳ��ϣ����������������۾���������M���л�������ԭ��H ���ϳ���N���ĺϳ�·�ߣ�
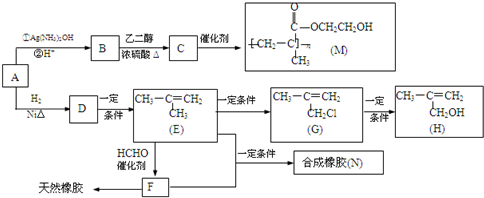
��1��A �к��еĹ������� ��д���ƣ���
��2��д����Ӧ���ͣ�D��E ��F����Ȼ��
��3��д����ѧ��Ӧ����ʽ��B��C�� ��E+F��N ��
��4��H��һ���������ܱ�����Ϊ��������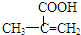 ���ò��������ж���ͬ���칹�壬��д�����������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ ����д2�֣���
���ò��������ж���ͬ���칹�壬��д�����������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ ����д2�֣���

��1��A �к��еĹ�������
��2��д����Ӧ���ͣ�D��E
��3��д����ѧ��Ӧ����ʽ��B��C��
��4��H��һ���������ܱ�����Ϊ��������
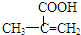 ���ò��������ж���ͬ���칹�壬��д�����������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ
���ò��������ж���ͬ���칹�壬��д�����������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������������ϳɷ�������M�Ľṹ��ʽ��֪CΪ��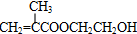 ��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��
��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��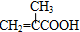 ��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��
��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��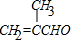 ��
��
A�����������ӳɷ�Ӧ����D��A��̼̼˫����̼��˫�����ܹ������������ӳɷ�Ӧ������Dһ���������ܹ�����E��֪��DΪ2-��-1-��������F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��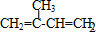 �����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ��
�����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ�� ��
��
�������Ϸ������1������3��
��4������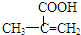 �ķ�����ɡ�ͬ���칹�����дԭ������Ҫ��д�������������л���Ľṹ��ʽ��
�ķ�����ɡ�ͬ���칹�����дԭ������Ҫ��д�������������л���Ľṹ��ʽ��
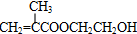 ��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��
��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��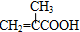 ��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��
��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��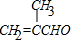 ��
��A�����������ӳɷ�Ӧ����D��A��̼̼˫����̼��˫�����ܹ������������ӳɷ�Ӧ������Dһ���������ܹ�����E��֪��DΪ2-��-1-��������F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��
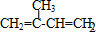 �����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ��
�����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ�� ��
���������Ϸ������1������3��
��4������
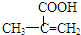 �ķ�����ɡ�ͬ���칹�����дԭ������Ҫ��д�������������л���Ľṹ��ʽ��
�ķ�����ɡ�ͬ���칹�����дԭ������Ҫ��д�������������л���Ľṹ��ʽ�����
�⣺����M�Ľṹ��ʽ��֪CΪ��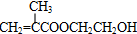 ��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��
��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��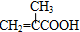 ��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��
��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��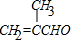 ��
��
A�����������ӳɷ�Ӧ����D��A��̼̼˫����̼��˫�����ܹ������������ӳɷ�Ӧ������Dһ���������ܹ�����E��֪��DΪ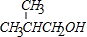 ����F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��
����F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��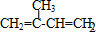 �����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ��
�����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ�� ��
��
��1�����ݷ�����֪��A�Ľṹ��ʽΪ��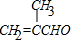 ������A�к��й�����Ϊ��ȩ����̼̼˫����
������A�к��й�����Ϊ��ȩ����̼̼˫����
�ʴ�Ϊ��ȩ����̼̼˫����
��2��DΪ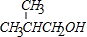 ��Dͨ����ȥ��Ӧ����E��
��Dͨ����ȥ��Ӧ����E��
FΪ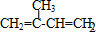 �������ϩͨ���Ӿ۷�Ӧ���������ϩ����Ȼ����
�������ϩͨ���Ӿ۷�Ӧ���������ϩ����Ȼ����
�ʴ�Ϊ����ȥ��Ӧ���Ӿ۷�Ӧ��
��3��BΪ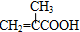 ��B���Ҷ�����Ӧ�Ļ�ѧ����ʽΪ��
��B���Ҷ�����Ӧ�Ļ�ѧ����ʽΪ�� ��
��
E��F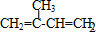 ��Ӧ�Ļ�ѧ����ʽΪ��
��Ӧ�Ļ�ѧ����ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
 ��
��
��4��H��һ���������ܱ�����Ϊ��������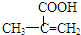 ���ò��������ж���ͬ���칹�壬���������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ�У�
���ò��������ж���ͬ���칹�壬���������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ�У�
HCOOC��CH3��=CH2��HCOOCH2-CH=CH2��HCOOCH=CH-CH3��CH3COOCH=CH2��CH2=CH-COOCH3��
�ʴ�Ϊ��HCOOC��CH3��=CH2��HCOOCH2-CH=CH2����HCOOCH=CH-CH3��CH3COOCH=CH2��CH2=CH-COOCH3����
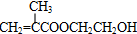 ��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��
��B���Ҷ�����Ӧ����C����B�Ľṹ��ʽΪ��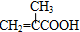 ��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��
��Aͨ����������Ӧ������H+��Ӧ����B����A�Ľṹ��ʽΪ��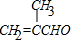 ��
��A�����������ӳɷ�Ӧ����D��A��̼̼˫����̼��˫�����ܹ������������ӳɷ�Ӧ������Dһ���������ܹ�����E��֪��DΪ
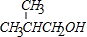 ����F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��
����F�ܹ�������Ȼ����F�Ľṹ��ʽΪ��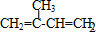 �����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ��
�����ݼӾ۷�Ӧԭ������д��E��F��Ӧ�Ļ�ѧ����ʽΪ�� ��
����1�����ݷ�����֪��A�Ľṹ��ʽΪ��
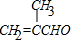 ������A�к��й�����Ϊ��ȩ����̼̼˫����
������A�к��й�����Ϊ��ȩ����̼̼˫�����ʴ�Ϊ��ȩ����̼̼˫����
��2��DΪ
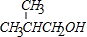 ��Dͨ����ȥ��Ӧ����E��
��Dͨ����ȥ��Ӧ����E��FΪ
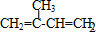 �������ϩͨ���Ӿ۷�Ӧ���������ϩ����Ȼ����
�������ϩͨ���Ӿ۷�Ӧ���������ϩ����Ȼ�����ʴ�Ϊ����ȥ��Ӧ���Ӿ۷�Ӧ��
��3��BΪ
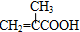 ��B���Ҷ�����Ӧ�Ļ�ѧ����ʽΪ��
��B���Ҷ�����Ӧ�Ļ�ѧ����ʽΪ�� ��
��E��F
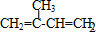 ��Ӧ�Ļ�ѧ����ʽΪ��
��Ӧ�Ļ�ѧ����ʽΪ�� ��
���ʴ�Ϊ��
 ��
�� ��
����4��H��һ���������ܱ�����Ϊ��������
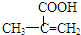 ���ò��������ж���ͬ���칹�壬���������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ�У�
���ò��������ж���ͬ���칹�壬���������Һ���̼̼˫����ͬ���칹��Ľṹ��ʽ�У�HCOOC��CH3��=CH2��HCOOCH2-CH=CH2��HCOOCH=CH-CH3��CH3COOCH=CH2��CH2=CH-COOCH3��
�ʴ�Ϊ��HCOOC��CH3��=CH2��HCOOCH2-CH=CH2����HCOOCH=CH-CH3��CH3COOCH=CH2��CH2=CH-COOCH3����
���������⿼�����л�����ƶϣ���Ŀ�Ѷ��еȣ���ȷ����ϳ�����Ϊ���ؼ����������ճ����л���ṹ������Ϊ��������Ļ�����ע����ϳɷ��ڽ���л��ƶ����е�Ӧ�÷����������ֿ����˵�ѧ���ķ������������������Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A��22.4L�����к�������ӵ���ĿΪNA |
| B��1 mol������O3���к�����ԭ�ӵ���ĿΪ2NA |
| C�����³�ѹ�£�14g�������е�ԭ����ĿΪNA |
| D��0.5 mol/LFe2��SO4��3��Һ�У�SO42-����ĿΪ1.5NA |
��ͼ�������Ƽ�Ĺ�ҵ����ʾ��ͼ����������ȥ�������з�����ȷ���ǣ�������
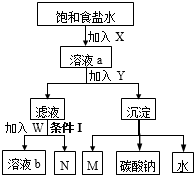

| A��X��M��ͬ������ |
| B��W���Ȼ��ƹ����X |
| C����Һb��N��ѭ������ |
| D���������Ǽ��� |
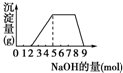 ij��Һ�п��ܺ���H+��NH4+��Mg2+��Fe3+��Al3+��SO42-�����ӣ��������Һ�м���ijŨ�ȵ�NaOH��Һʱ���������ɳ��������ʵ�����������Һ��NaOH�����ʵ����ı仯��ͼ��ʾ��[��֪��Mg2++2OH-�TMg��OH��2����Fe3++3OH-�TFe��OH��3������Mg��OH��2��Fe��OH��3������ˮ��NH4++OH-�TNH3?H2O]�ɴ˿�֪������Һ�п϶����е�������
ij��Һ�п��ܺ���H+��NH4+��Mg2+��Fe3+��Al3+��SO42-�����ӣ��������Һ�м���ijŨ�ȵ�NaOH��Һʱ���������ɳ��������ʵ�����������Һ��NaOH�����ʵ����ı仯��ͼ��ʾ��[��֪��Mg2++2OH-�TMg��OH��2����Fe3++3OH-�TFe��OH��3������Mg��OH��2��Fe��OH��3������ˮ��NH4++OH-�TNH3?H2O]�ɴ˿�֪������Һ�п϶����е�������