��Ŀ����
7����������ΪLiCoO2������ӵ���ѱ��㷺������Яʽ��Դ�����ܵ���Դ�ѷ����������һ����չ����1�����ʯ��LiFePO4��һ��DZ�ڵ�����ӵ���������ϣ�������ͨ����NH4��2Fe��SO4��2��H3PO4��LiOH��Һ������������Ӧ�����ó�����80����ո�����³��Ͷ��Ƶã�
�ٹ�������ӦͶ��ʱ��������NH4��2Fe��SO4��2��LiOH��Һֱ�ӻ�ϵ�ԭ����Fe2+�ڼ��������¸��ױ�������
�ڹ�������Ӧ�Ļ�ѧ����ʽΪ��NH4��2Fe��SO4��2+LiOH+H3PO4=LiFePO4��+2NH4HSO4+H2O��
�۸��³���ǰ������LiFePO4�м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ��LiFePO4�ĵ��������⣬�����������O2��Ӧ����ֹLiFePO4�е�Fe2+��������
��2���Ͼ�����ӵ�ص�����������������Ҫ����LiCoO2������AI��Fe�ȣ���ͨ������ʵ�鷽�������ܡ�ﮣ�
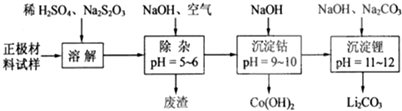
���������ܽ�����У�S2O32-��������SO42-��LiCoO2���ܽ�����з�Ӧ�Ļ�ѧ����ʽΪ8LiCoO2+Na2S2O3+11H2SO4=4Li2SO4+8CoSO4+Na2SO4+11H2O��
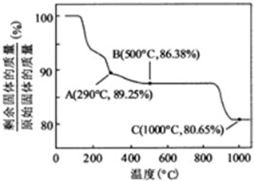
��Co��OH��2�ڿ����м���ʱ��������������¶ȵı仯������ͼ��ʾ����֪�ܵ��������������290��ʱ����ȫ��ˮ����1000��ʱ��ʣ�����ijɷ�ΪCoO����ѧʽ������350��400�淶Χ�ڣ�ʣ�����ijɷ�ΪCo2O3��Co3O4���ѧʽ����
���� ��1���ٲ���ֱ�ӻ�ϵ�ԭ����Fe2+�ڼ��������¸����ױ�������
�ڸ����������Ϣ�������ķ�ӦΪ��NH4��2Fe��SO4��2+LiOH+H3PO4=LiFePO4+2NH4HSO4+H2O��
�����Ŀ����е�O2������Fe2+����ֹFe2+��������
��2����������ΪLiCoO2������ӵ�أ�����������������Ҫ����LiCoO2������AI��Fe�ȣ�������ϡ�����ܽ���������ܽ�����������������������������������ƣ�S2O32-��������SO42-�����ɵ���Һ�к�������ﮡ������ܡ������ƣ���������������Һ��ͨ�����������������Ϊ�����ӣ��γ������ӡ������ӵij�������Һ�м��������������Ƶ�����ҺPH�������ܣ����˵õ��������ܣ���Һ�м����������ơ�̼���Ƶ�����ҺPH����������γ�̼��ﮣ�
��ͨ�������Ϣ��֪LiCoO2��Na2S2O3������������ԭ��Ӧ����ӦΪ8LiCoO2+Na2S2O3+11H2SO4=4Li2SO4+8CoSO4+Na2SO4+11H2O��
�ڸ��������غ㶨�ɣ��ڱ仯�����У�Co������û�б䣬ͨ��������ݿ�����1000����Co��OH��2��ȫ�ֽ⣬�����CoO��
��350-400��ʱ�������������89.25%-86.38%֮�䣬����ͨ��������з�����
��290�棬n��Co����n��O��=$\frac{100}{93}$��[��89.25-100��$\frac{59}{93}$����16]=2��3���仯ѧʽΪCo2O3��
��500�棬n��Co����n��O��=$\frac{100}{93}$��[��86.38-100��$\frac{59}{93}$����16]=3��4���仯ѧʽΪCo3O4��
���Կ���ȷ����350-400��ʱ�Ļ�ѧʽΪCo2O3��Co3O4��
��� �⣺��1����NH4��2Fe��SO4��2��LiOH��Һ��Ӧ����Fe��OH��2��Fe��OH��2�ױ��������������Բ��ܽ�ֱ�ӻ�ϣ�
�ʴ�Ϊ��Fe2+�ڼ��������¸��ױ�������
�ڸ��������Ϣ����NH4��2��H3PO4��LiOH��Һ������������Ӧ����LiFePO4��NH4HSO4��H2O����Ӧ�Ļ�ѧ����ʽΪ����NH4��2Fe��SO4��2+LiOH+H3PO4=LiFePO4+2NH4HSO4+H2O��
�ʴ�Ϊ����NH4��2Fe��SO4��2+LiOH+H3PO4=LiFePO4��+2NH4HSO4+H2O��
�۸��³���ǰ������LiFePO4�м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ��LiFePO4�ĵ��������⣬�������Ŀ����е�����������Fe2+����ֹFe2+��������
�ʴ�Ϊ���������O2��Ӧ����ֹLiFePO4�е�Fe2+��������
��2����������������Ҫ����LiCoO2������Al��Fe�ȣ�����ϡH2SO4��Na2S2O3��S2O32-��������SO42-�����л�ԭ�ԣ�����������ֻ��LiCoO2���������ԣ��뷴ӦNa2S2O3��Ӧ����CoSO4����Ӧ��ѧ����ʽΪ��8LiCoO2+Na2S2O3+11H2SO4=4Li2SO4+8CoSO4+Na2SO4+11H2O��
�ʴ�Ϊ��8LiCoO2+Na2S2O3+11H2SO4=4Li2SO4+8CoSO4+Na2SO4+11H2O��
�ڸ��������غ㶨�ɣ��ڱ仯�����У�Co������û�б䣬����ԭʼ��������Ϊ100g����n��Co��=$\frac{100}{93}$mol��m��Co��=100��$\frac{59}{93}$g��
��1000��ʱ�������������ٱ仯��˵��Co��OH��2��ȫ�ֽ⣬n��Co����n��O��=$\frac{100}{93}$��[��80.65-100��$\frac{59}{93}$����16]=1��1��ʣ�����ɷ�ΪCoO��
��350-400��ʱ�������������89.25%-86.38%֮�䣬����ͨ��������з�����
��290�棬n��Co����n��O��=$\frac{100}{93}$��[��89.25-100��$\frac{59}{93}$����16]=2��3���仯ѧʽΪCo2O3��
��500�棬n��Co����n��O��=$\frac{100}{93}$��[��86.38-100��$\frac{59}{93}$����16]=3��4���仯ѧʽΪCo3O4��
���Կ���ȷ����350-400��ʱ�Ļ�ѧʽΪCo2O3��Co3O4��
�ʴ�Ϊ��CoO��Co2O3��Co3O4��
���� ���⿼���֪ʶ�Ƚ�ɢ���漰����Դ���ã��������ʡ��������̷�����ͼ��������������ȽϹ㣬��Ŀ�ѶȽϴ�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | һ����̼ | B�� | �Ȼ��� | C�� | ��������̼ | D�� | ���� |
 ͼ
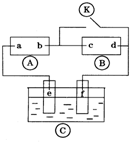
ͼ Ϊֱ����Դ��
Ϊֱ����Դ�� Ϊ�������Ȼ�����Һ�ͷ�̪��Һ����ֽ��
Ϊ�������Ȼ�����Һ�ͷ�̪��Һ����ֽ�� Ϊ��Ʋۣ���ͨ��·����
Ϊ��Ʋۣ���ͨ��·���� �ϵ�c���Ժ�ɫ��Ϊʵ�����϶�п����ͨ
�ϵ�c���Ժ�ɫ��Ϊʵ�����϶�п����ͨ ��ʹc��d�����·������������ȷ���ǣ�������
��ʹc��d�����·������������ȷ���ǣ�������| A�� | aΪֱ����Դ�ĸ��� | B�� | f�缫Ϊп�� | ||
| C�� | e��������ԭ��Ӧ | D�� | c�������ķ�ӦΪ2 H++2e-=H2�� |
| A�� | 0.1mol•L-1Na2S��Һ�к���0.2NA��Na+ | |
| B�� | ��״���£�22.4L��H2O����2NA����ԭ�� | |
| C�� | �����£�44gCO2����NA����ԭ�� | |
| D�� | 0.1molNa������O2��Ӧ����Na2O��ת��0.1NA������ |
| A�� | ���³�ѹ�£�16g14CH4����������ĿΪ8NA | |
| B�� | ��״���£�7.8g���к�̼̼˫������ĿΪ0.3NA | |
| C�� | ��0.2NA�������ӵ�Na2O2��ˮ��ȫ��Ӧת��0.2mol���� | |
| D�� | 5.6g�����������������г��ȼ�գ��õ�������Ϊ0.3NA |
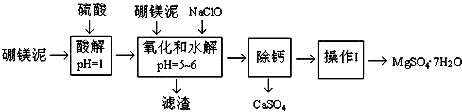
��������ش��������⣺
��1�����������У���ӿ�������ʣ���������ֿ��еĴ�ʩ���¡�����þ����顢����ȣ�
��2����������Ҫ�ɷݳ�����Fe��OH��3��Al��OH��3��MnO2�⣬����SiO2��
��3�������NaClO����Mn2+��Ӧ����MnO2�������÷�Ӧ�����ӷ���ʽ��Mn2++ClO-+H2O�TMnO2��+2H++Cl-���ڵ���pH������ˮ��֮ǰ������һ������Ҳ�ᱻNaClO�������÷�Ӧ�����ӷ���ʽΪ2Fe2++ClO-+2H+�T2Fe3++Cl-+H2O��
��4��Ϊ�˼�����Һ��Fe3+�Ƿ�������ѡ�õ��Լ���ȡ������Һ�������м������軯����Һ�������Һ�����ɫ��˵����Һ�в���Fe3+�������Һ���ɫ��˵����Һ�к�Fe3+��
A��KSCN B������-KI��Һ C��H2O2 D��KMnO4ϡ��Һ
��5����֪MgSO4��CaSO4���ܽ�������
| �¶�/�� | 40 | 50 | 60 | 70 |
| MgSO4 | 30.9 | 33.4 | 35.6 | 36.9 |
| CaSO4 | 0.210 | 0.207 | 0.201 | 0.193 |
��6������l��60t��þ�࣬����MgSO4•7H2O�����������̵IJ���Ϊ50%������������MgSO4•7H2O��Ʒ1.97t ��������������λ��Ч���֣���
| A�� | ���� | B�� | ������ | C�� | ��ά�� | D�� | ����ϩ |
 ��͵�Ԫ���ڻ�ѧ���к���Ҫ�ĵ�λ���ش��������⣺
��͵�Ԫ���ڻ�ѧ���к���Ҫ�ĵ�λ���ش��������⣺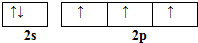 ��Ԥ����2017�귢��ġ��϶���š�̽�������õij���5�����ػ��ȼ��Ϊƫ������[��CH3��2NNH2]����CH3��2NNH2��Nԭ�ӵ��ӻ���ʽΪsp3��
��Ԥ����2017�귢��ġ��϶���š�̽�������õij���5�����ػ��ȼ��Ϊƫ������[��CH3��2NNH2]����CH3��2NNH2��Nԭ�ӵ��ӻ���ʽΪsp3��