��Ŀ����
 ��֪A��B��C��D���ֶ�����Ԫ�أ����ǵĺ˵������������Aԭ�ӡ�Cԭ�ӵ�I���ܲ��У���������δ�ɶԵĵ��ӣ�C��Dͬ���壮E��F���ǵ�������Ԫ�أ�Eԭ�Ӻ�����4��δ�ɶԵ��ӣ�Fԭ�ӳ������ܲ�ֻ��1�������⣬������ܲ��Ϊȫ����������������Ϣ�ش�
��֪A��B��C��D���ֶ�����Ԫ�أ����ǵĺ˵������������Aԭ�ӡ�Cԭ�ӵ�I���ܲ��У���������δ�ɶԵĵ��ӣ�C��Dͬ���壮E��F���ǵ�������Ԫ�أ�Eԭ�Ӻ�����4��δ�ɶԵ��ӣ�Fԭ�ӳ������ܲ�ֻ��1�������⣬������ܲ��Ϊȫ����������������Ϣ�ش���1����̬Dԭ���У�����ռ�ݵ�����ܲ����
��2��E2+���ӵļ۲�����Ų�ͼ��
��3��AԪ�ص�����������Ӧ��ˮ��������ԭ�Ӳ�ȡ�Ĺ���ӻ���ʽΪ
VSEPRģ��Ϊ
��4��������AC2��B2C��������DAB-��Ϊ�ȵ����壬���ǽṹ���ƣ�DAB-�ĵ���ʽΪ
��5��ij�ֻ�������D��E��F����Ԫ����ɣ��侧����ͼ��ʾ��lpm=10-10cm�������仯ѧʽΪ
���㣺�����ļ���,λ�ýṹ���ʵ����ϵӦ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��,��ѧ���뾧��ṹ
������A��B��C��D�������ڱ��еĶ�����Ԫ�أ����ǵĺ˵������������Aԭ�ӡ�Cԭ�ӵ�L�ܲ��ж�������δ�ɶԵĵ��ӣ���Aԭ�Ӻ�������Ų�Ϊ1s22s22p2��Cԭ�Ӻ�������Ų�Ϊ1s22s22p4����AΪ̼Ԫ�ء�CΪ��Ԫ�أ�Bԭ����������C��O֮�䣬��BΪ��Ԫ�أ�C��Dͬ���壬��DΪSԪ�أ�E��F���ǵ�������Ԫ�أ�Eԭ�Ӻ�����4��δ�ɶԵ��ӣ�ԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p63d64s2����EΪFe��Fԭ�ӳ������ܲ�ֻ��1�������⣬������ܲ��Ϊȫ������Fԭ�Ӻ��������=2+8+18+1=29����FΪCuԪ�أ��ݴ˽��
���
�⣺A��B��C��D�������ڱ��еĶ�����Ԫ�أ����ǵĺ˵������������Aԭ�ӡ�Cԭ�ӵ�L�ܲ��ж�������δ�ɶԵĵ��ӣ���Aԭ�Ӻ�������Ų�Ϊ1s22s22p2��Cԭ�Ӻ�������Ų�Ϊ1s22s22p4����AΪ̼Ԫ�ء�CΪ��Ԫ�أ�Bԭ����������C��O֮�䣬��BΪ��Ԫ�أ�C��Dͬ���壬��DΪSԪ�أ�E��F���ǵ�������Ԫ�أ�Eԭ�Ӻ�����4��δ�ɶԵ��ӣ�ԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p63d64s2����EΪFe��Fԭ�ӳ������ܲ�ֻ��1�������⣬������ܲ��Ϊȫ������Fԭ�Ӻ��������=2+8+18+1=29����FΪCuԪ�أ�
��1����̬Sԭ���е���ռ�ݵ�����ܲ�Ϊ��3�ܲ㣬����M�����ܲ���1��s�����3��p�����5��d���������9 ��ԭ�ӹ�����ʴ�Ϊ��M��9��
��2��Fe2+���ӵļ۲�����Ų�3d6����۲�����Ų�ͼ�� ��FΪCuԪ�أ�ԭ�Ӻ�����29�����ӣ�ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1���ʴ�Ϊ��
��FΪCuԪ�أ�ԭ�Ӻ�����29�����ӣ�ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1���ʴ�Ϊ�� ��1s22s22p63s23p63d104s1��
��1s22s22p63s23p63d104s1��
��3��̼Ԫ�ص�����������Ӧ��ˮ����ΪH2CO3������Cԭ�ӳ�3���Ҽ���û�йµ��Ӷԣ�Cԭ�Ӳ�ȡsp2�ӻ���ʽ��B����̬�⻯��ΪNH3��NH3������Nԭ�Ӽ۲���Ӷ���=3+
=4����VSEPRģ��Ϊ�����壬�ʴ�Ϊ��sp2�������壻
��4����CO2��Ϊ�ȵ����壬��֪SCN-����ʽ��CO2��ͬ������CO2�ĵ���ʽд������ʽΪ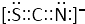 ���ʴ�Ϊ��
���ʴ�Ϊ��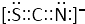 ��
��
��5��������D��S��ԭ��λ�ھ����ڲ���ԭ����ĿΪ8����E��Fe��ԭ��6��λ�����ϡ�4��λ�����ϣ�E��Fe��ԭ����Ŀ=6��
+4��
=4��F��Cu��ԭ��λ��4��λ�����ϡ�1���ڲ���8�������ϣ�ԭ����Ŀ=4��
+1+8��
=4��������Cu��Fe��Sԭ����Ŀ֮��=4��4��8=1��1��2���ʸþ��廯ѧʽΪCuFeS2���þ�����ܶ�d=
=4.66g/cm3��
�ʴ�Ϊ��CuFeS2��4.66��
��1����̬Sԭ���е���ռ�ݵ�����ܲ�Ϊ��3�ܲ㣬����M�����ܲ���1��s�����3��p�����5��d���������9 ��ԭ�ӹ�����ʴ�Ϊ��M��9��
��2��Fe2+���ӵļ۲�����Ų�3d6����۲�����Ų�ͼ��
 ��FΪCuԪ�أ�ԭ�Ӻ�����29�����ӣ�ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1���ʴ�Ϊ��
��FΪCuԪ�أ�ԭ�Ӻ�����29�����ӣ�ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1���ʴ�Ϊ�� ��1s22s22p63s23p63d104s1��
��1s22s22p63s23p63d104s1����3��̼Ԫ�ص�����������Ӧ��ˮ����ΪH2CO3������Cԭ�ӳ�3���Ҽ���û�йµ��Ӷԣ�Cԭ�Ӳ�ȡsp2�ӻ���ʽ��B����̬�⻯��ΪNH3��NH3������Nԭ�Ӽ۲���Ӷ���=3+
| 5-1��3 |
| 2 |
��4����CO2��Ϊ�ȵ����壬��֪SCN-����ʽ��CO2��ͬ������CO2�ĵ���ʽд������ʽΪ
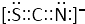 ���ʴ�Ϊ��
���ʴ�Ϊ��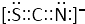 ��
�� ��5��������D��S��ԭ��λ�ھ����ڲ���ԭ����ĿΪ8����E��Fe��ԭ��6��λ�����ϡ�4��λ�����ϣ�E��Fe��ԭ����Ŀ=6��
| 1 |
| 2 |
| 1 |
| 4 |
| 1 |
| 2 |
| 1 |
| 8 |
| 4��56+4��64+8��32 |
| 6.02��1023��500��500��1050��10-30 |
�ʴ�Ϊ��CuFeS2��4.66��
�����������ǶԿ������ʽṹ�Ŀ��飬�漰��������Ų����ӻ����ۡ��۲���ӶԻ������ۡ�������������ȣ���Ҫѧ���߱���ʵ�Ļ������Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
����������Һ�������ͺ��Ȼ�����Һ ��39%���Ҵ���Һ ���Ȼ��ƺ͵������ˮ��Һ���������ϸ����Һ����ȷ���������ǣ�������
| A����Һ��������ȡ |
| B����ȡ������Һ |
| C����Һ����ȡ������ |
| D��������ȡ����Һ |
���Ӵ��Ǻϳɹ�����ά�ĵ��壬�ṹ��ʽ��ͼ�������йؽ��Ӵ���˵������ȷ���ǣ�������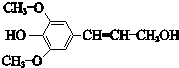
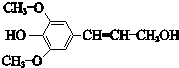
| A������ʽΪC11H14O4 |
| B������˳���칹���� |
| C���ܷ���ˮ�ⷴӦ |
| D��������ˮ��Ӧ |
���з�Ӧԭ�������Ϲ�ҵұ������ʵ��������ǣ�������
A��Cu2S+O2
| ||||
B��2Ag2O
| ||||
C��Fe2O3+3CO
| ||||
D��2AlCl3�����ڣ�
|
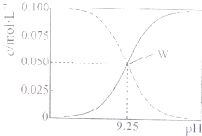 25��ʱ������ˮ���Ȼ����Һ��ϵõ�c��NH3?H2O��+c��NH4+��=0.1mol?L-1�Ļ����Һ����Һ��c��NH3?H2O����c��NH4+����pH�Ĺ�ϵ��ͼ��ʾ�������й�����Ũ�ȹ�ϵ����һ����ȷ���ǣ�������
25��ʱ������ˮ���Ȼ����Һ��ϵõ�c��NH3?H2O��+c��NH4+��=0.1mol?L-1�Ļ����Һ����Һ��c��NH3?H2O����c��NH4+����pH�Ĺ�ϵ��ͼ��ʾ�������й�����Ũ�ȹ�ϵ����һ����ȷ���ǣ�������| A��W���ʾ��Һ�У�c��NH4+��+c��H+��=c��Cl-��+c��OH-�� |
| B��pH=10.5��Һ��c��Cl-��+c��OH-��+c��NH3?H2O����0.1 mol?L-1 |
| C��pH=9.5��Һ��c��NH3?H2O����c��NH4+����c��OH-����c��H+�� |
| D����W������ʾ1L��Һ�м���0.05molNaOH���壨��Һ����仯�ɺ��ԣ���c��Cl-����c��Na+����c��OH-����c��NH4+����c��H+�� |