��Ŀ����
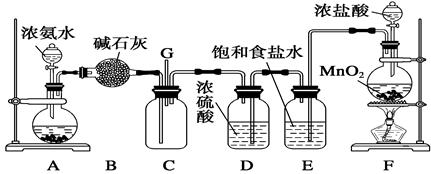
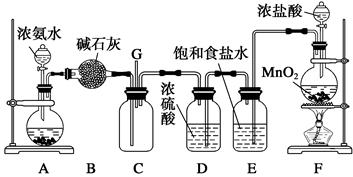
(��12��)ijѧ����������װ��̽�������백��֮��ķ�Ӧ������A��F�ֱ�Ϊ�����������ķ���װ�ã�CΪ��������������백����Ӧ��װ�á�ʵ���ҿ�����װ��F�Ʊ���������Ӧ�Ļ�ѧ����ʽΪ��MnO2��4HCl(Ũ) MnCl2��Cl2����2H2O
��ش��������⣺
(1)��װ��F�з�����Ӧ�����ӷ���ʽΪ_______________________________________��
(2)��Ϊ�˿����Ʊ�������װ��A����ƿ�п�װ�Լ�_____________________��
(3)��Bװ�õ�������_____________��(�����) A.ϴ��ƿ B.����� C.��Һ©��
Eװ�õ�������__________________________________________________________��
(4)ͨ��Cװ�õ�����������߽ϳ����ұ߽϶̣�Ŀ����________________________��
(5)װ��C�ڳ���Ũ��İ��̲��������ڱ����ᣬ��һ�������ǿ�������Ҫ�ɷ�֮һ����д����Ӧ�Ļ�ѧ����ʽ��_________________________________________________��
(6)��װ��C��G���ݳ���β���п��ܺ��л���ɫ���ж����壬��δ�����____________________________________________________________________________��
(1)MnO2��4H����2Cl��Mn2����Cl2����2H2O��2�֣�
(2).CaO��.NaOH��1�֣�
(3)B����1�֣� ��ȥ�����е��Ȼ��⣨2�֣�
(4)ʹ�ܶȴ���������ܶ�С�İ����Ͽ�ؾ��Ȼ�ϣ�2�֣�
(5)3Cl2��8NH3===N2��6NH4Cl��2�֣�
(6)��G���ӵ���ֱ��ͨ��ʢ���ռ���ձ��У�2�֣�
����:��1��F������ȡ�����ģ�����ʽΪMnO2��4H����2Cl��Mn2����Cl2����2H2O��
��2����ˮ���лӷ���Ũ��Խ��Խ�ӷ�����������ܽ�����¶ȵ����߶����ͣ����Կ��Խ���ˮ������ʯ�һ��������ƹ����С�
��3������Bװ���ص���жϣ��������Ǹ���ܡ���ȡ�������к����Ȼ������壬������Ҫ���ñ���ʯ��ˮ����ȥ�����Ȼ��⡣
��4���������ܶ�С�ڿ����ģ��������ܶȴ��ڿ����ģ�������������Ŀ����Ϊ��ʹ�����ֻ�ϡ�
��5����������ǿ�����ԣ�����������������ʵ��������ж��������ǵ������Ȼ�泥�����ʽΪ3Cl2��8NH3===N2��6NH4Cl��
��6�������ж�����Ҫβ���������������ü�Һ�����ն����������

 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�