��Ŀ����
11��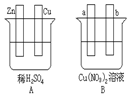 ��Ҫ���ͼ���������б�Ҫ�����Ӳ���գ�
��Ҫ���ͼ���������б�Ҫ�����Ӳ���գ���1����Aͼ�У�ʹͭƬ��ðH2���ݣ�����Ա�Ҫ���ӣ��缫��Ӧʽ��п�壺Zn-2e-=Zn2+��ͭ�壺2H++2e-=H2����
��2����Bͼ�У�a�� b����ʯī�缫��ʹa������ͭ�����Ա�Ҫ�����Ӻ缫��Ӧʽ��a��Cu2++2e-=Cu b��4OH--4e-=O2��+2H2O������һ��ʱ���ֹͣ��Ӧ�����һ������CuO��CuCO3����Һ�ָܻ�������ǰ��ȫһ�£�
���� ��1����Aͼ�У�ʹͭƬ��ðH2���ݣ������õ�������Zn��Cu�缫�γ�ԭ��ط�Ӧ��Zn������ʧ���ӷ���������Ӧ��ϡ������Һ����������ͭ���ϵõ����ӷ�����ԭ��Ӧ����������
��2��Bͼʹa������ͭ����b����O2��Ӧ���ӵ�Դ���缫��̼����a�缫��������b�缫���������γɵ��أ���Һ������������������ʧ��������������ͭ�����������õ���������ͭ����Ӧ�������������ᣬ��ҺpH���ͣ�������ʲô��ʲôʹ��Һ�ָ�������ǰ��ȫһ�£�
��� �⣺��1����Aͼ�У�ʹͭƬ��ðH2���ݣ������õ�������Zn��Cu�缫�γ�ԭ��ط�Ӧ��Zn������ʧ���ӷ���������Ӧ���缫��ӦΪ��Zn-2e-=Zn2+��ͭ��������ϡ������Һ����������ͭ���ϵõ����ӷ�����ԭ��Ӧ�����������缫��ӦΪ��2H++2e-=H2����
�ʴ�Ϊ��Zn-2e-=Zn2+��2H++2e-=H2����
��2��Bͼʹa������ͭ��b����O2��Ӧ���ӵ�Դ���缫��̼����a�缫��������b�缫���������γɵ��أ���Һ������������������b��ʧ���������������缫��ӦΪ��4OH--4e-�T2H2O+O2����ͭ����������a�ϵõ���������ͭ���缫��ӦΪ��2Cu2++4e-�T2Cu���ܷ�ӦΪ��2Cu��NO3��2+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2Cu+O2��+2 H2SO4����Ӧ�������������ᣬ���Ի���ǿ��������������ͭ�������ų�������������ʹ��Һ�ָ�������ǰ��ȫһ�£��ɼ���һ������CuO��CuCO3�ȣ�a����Cu2++2e-=Cu��b����4OH--4e-=O2��+2H2O������һ��ʱ���ֹͣ��Ӧ�����һ������CuO��CuCO3����Һ�ָܻ�������ǰ��ȫһ�£�
�ʴ�Ϊ��Cu2++2e-=Cu��4OH--4e-=O2��+2H2O��CuO��CuCO3��
���� ���⿼����ԭ��غ͵���ԭ������ȷ���ӷŵ�˳���ǽⱾ��ؼ���ע�������ƺ͵缫��Ӧ�ķ����жϣ���Ŀ�Ѷ��еȣ�

| A�� | ��֬�ǵ�λ��������������ߵ�Ӫ������ | |
| B�� | ʯ���ѽ��Ŀ�����������Һ��ȼ�ϵIJ��� | |
| C�� | �ƼغϽ��ܶ�С���۵�ͣ����Կ������˷�Ӧ�ѵĴ��Ƚ��� | |
| D�� | ����±Ũ���������������ȿ�����ˮ��������ȡ�õ���ˮ�е��� |
| A�� | ʯӢ��ˮ����ˮ�ࡢ���ά�����ʵ���Ҫ�ɷ���SiO2 | |
| B�� | ���ڳ�ʪ�Ŀ����з��ã�������ѧ��ʴ������ | |
| C�� | ҽ���ϣ�̼������������θ����һ��ҩ�� | |
| D�� | ����ˮ�������ó涣ҧ������ϴ�� |
| �� ���� | IA | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | �� | �� | ||||
| �� | �� | �� | �� | �� | �� | �� |
��2����������Ԫ�س�����ԭ�Ӱ뾶��С����Cl����Ԫ�ط��ţ�����ЩԪ�ص�����������Ӧˮ�����м�����ǿ����NaOH���ѧʽ����ͬ�����������Ե���Al��OH��3��
��3���ݢޢߵ��ʵĻ�������ǿ������˳��ΪNa��Mg��Al ����Ԫ�ط��ţ����жϵ�ʵ�������ǵ��ʷֱ���ˮ���ᷴӦ�ľ��ҳ̶ȣ���������������Ӧ��ˮ����ļ���ǿ���� ��д��һ�֣���
��4���ߡ���ĵ��ʷֱ���ݵ�����������ˮ���ﷴӦ�����ӷ���ʽ����Ϊ2Al+2H2O+2OH-=2AlO2-+3H2����Cl2+2OH-=Cl-+ClO-+H2O��
| A�� | HCl��NaOH��Ӧ���к��ȡ�H=-57.3kJ/mol����H2SO4��Ca��OH��2��Ӧ���к���Ϊ2����-57.3��kJ/mol | |
| B�� | 500�桢30MPa�£���0.5molN2��1.5mol H2�����ܱ������г�ַ�Ӧ����NH3��g��������19.3kJ�����Ȼ�ѧ����ʽΪ��N2��g��+3H2��g�� $?_{500�桢30MPa}^{����}$2NH3��g����H=-38.6kJ•mol-1 | |
| C�� | CO��g����ȼ������283.0 kJ/mol�����ʾCOȼ���ȵ��Ȼ�ѧ����ʽΪCO��g��+$\frac{1}{2}$O2��g���TCO2 ��g����H=-283.0 kJ/mol | |
| D�� | ��Ҫ���Ȳ��ܷ����ķ�Ӧһ�������ȷ�Ӧ |
| A�� | �ɱ��еķ��Ӽ��������͵��ʵ��еķ��Ӽ������� | |
| B�� | CsCl��������һ��Cl-���ڵ�Cs+����NaCl��������һ��Cl-���ڵ�Na+�� | |
| C�� | �������Si-Si���ļ��ܺͽ��ʯ��C-C���ļ��� | |
| D�� | ˮ������O-H���ļ��Ǻͼ��������C-H���ļ��� |
 ��D�ļ���̬�⻯��Ŀռ乹��Ϊ�����Σ�
��D�ļ���̬�⻯��Ŀռ乹��Ϊ�����Σ�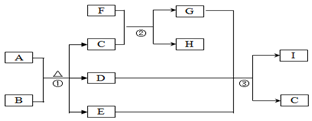 �й����ʵ�ת����ϵ��ͼ��ʾ�������������뷴Ӧ��������ȥ����A�dz����ķǽ������嵥�ʣ�F���Ϻ�ɫ�Ľ������ʣ�B��C�dz�����ǿ�ᣬD��G��I�dz��������壬D��I�����Ԫ����ͬ����D����Է���������I�Ĵ�16��E���������ɫҺ�壮
�й����ʵ�ת����ϵ��ͼ��ʾ�������������뷴Ӧ��������ȥ����A�dz����ķǽ������嵥�ʣ�F���Ϻ�ɫ�Ľ������ʣ�B��C�dz�����ǿ�ᣬD��G��I�dz��������壬D��I�����Ԫ����ͬ����D����Է���������I�Ĵ�16��E���������ɫҺ�壮 ��
��