��Ŀ����
 ��ѧ��Ӧ�е���ЧӦ
��ѧ��Ӧ�е���ЧӦ��1����������������Ӧ����1molˮ��������241.8kJ��д���÷�Ӧ���Ȼ�ѧ����ʽ��
��2��ʵ������50mL 0.50mol?L-1���ᡢ50mL 0.55mol?L-1 NaOH��Һ����ͼ��ʾװ�ý��вⶨ�к��ȵ�ʵ�飬�õ����е����ݣ�
| ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶�t2/�� | |
| ���� | NaOH��Һ | ||
| 1 | 20.2 | 20.3 | 23.7 |
| 2 | 20.3 | 20.5 | 23.8 |
| 3 | 21.5 | 21.6 | 24.9 |
��ʵ��ʱ�û��β�����������Һ�ķ�����
������ͭ˿��������滷�β�������������
�ھ����ݴ�����t2-t1=3.4�森���ʵ���õ��к��ȡ�H=
������NaOH��Һ��Ϊ��ͬ�������ͬŨ�ȵİ�ˮ������к���Ϊ��H1�����H1���H�Ĺ�ϵΪ����H1
��3����֪��C��s��+H2O��g��=CO��g��+H2��g����H1 2C��s��+O2��g��=2CO��g����H2=-220kJ/molH-H��O=O��O-H���ļ��ֱܷ�Ϊ436��496��462kJ/mol�����H1=
���㣺�к��ȵIJⶨ,�Ȼ�ѧ����ʽ
ר�⣺��ѧ��Ӧ�е������仯
��������1�������Ȼ�ѧ����ʽ����дԭ��д������ȼ��������̬ˮ���Ȼ�ѧ����ʽ��ע�ⷽ���е������ͻ�ѧ������Ҫ��Ӧ������1mol��̬ˮת����Һ̬ˮ�ų������������������������Ӧ������̬ˮ�ķ�Ӧ�ȼ�������Һ̬ˮ�ķ�Ӧ�ȣ�������ȼ������ֵ1mol������ȫȼ������Һ̬ˮ�ų�����������λ��kJ/mol��
��2���ٻ��β�����������½���ʹ��Һ��ַ�Ӧ���������ȿ죬������ʧ�ࣻ
�ڸ��ݹ�ʽQ=cm��T���������0.05mol��ˮ�ų��������������к��ȵĸ��������Ӧ�ȣ�
�۸���������ʵ������ȷ�����
��3�����ݸ�˹���ɼ���ˮ�ֽⷴӦ���ʱ䣬��ѧ��Ӧ���ʱ��H=H����-H��Ӧ���ٽ�ϻ�ѧ���ܺ����������Ĺ�ϵ���ش�
��2���ٻ��β�����������½���ʹ��Һ��ַ�Ӧ���������ȿ죬������ʧ�ࣻ
�ڸ��ݹ�ʽQ=cm��T���������0.05mol��ˮ�ų��������������к��ȵĸ��������Ӧ�ȣ�
�۸���������ʵ������ȷ�����
��3�����ݸ�˹���ɼ���ˮ�ֽⷴӦ���ʱ䣬��ѧ��Ӧ���ʱ��H=H����-H��Ӧ���ٽ�ϻ�ѧ���ܺ����������Ĺ�ϵ���ش�
���
�⣺��1��������������Ӧ����1molˮ��������241.8kJ���÷�Ӧ���Ȼ�ѧ����ʽΪ��H2��g��+
O2��g��=H2O��g����H=-241.8kJ/mol��1gˮ����ת����Һ̬ˮ����2.44kJ����18gˮ����ת����Һ̬ˮ�ų�����2.5kJ��18=45kJ���ʷ�ӦH2��g��+
O2��g���TH2O��l���ķ�Ӧ�ȡ�H=-��241.8kJ/mol+45kJ/mol��=-286.8kJ/mol������2H2��g��+O2��g��=2H2O��1���ġ�H=-573.6kJ?mol-1��������ȼ����Ϊ286.8kJ/mol��
�ʴ�Ϊ��H2��g��+
O2��g��=H2O��g����H=-241.8kJ/mol��-573.6��-286.8��
��2���ٻ��β�����������½���ʹ��Һ��ַ�Ӧ�����ܽ����β����������Ϊͭ˿���������Ϊͭ˿��������ȵ������壬
�ʴ�Ϊ�����½�����ͭ���ȿ죬��ֹ������ʧ��
��50mL 0.50mol?L-1���ᡢ50mL 0.55mol?L-1 NaOH��Һ��������Ϊm=100mL��1g/mL=100g��c=4.18J/��g?�棩��t2-t1=3.4�棬���빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18J/��g?�棩��100g��3.4��=1421.2J=1.4212KJ��������0.025mol��ˮ�ų�����1.4212KJ����������1mol��ˮ�ų�����Ϊ
=56.8kJ������ʵ���õ��к��ȡ�H=-56.8kJ/mol��
�ʴ�Ϊ��-56.8kJ/mol��
��һˮ�ϰ�Ϊ����������Ϊ���ȹ��̣������ð�ˮ����ϡ����������Һ��Ӧ����Ӧ�ų�������С��56.8kJ�����ԡ�H1����H���ʴ�Ϊ������NH3?H2O�������ȣ�
��3����֪��C��s��+H2O��g���TCO��g��+H2��g������H1 ��
��2C��s��+O2��g���T2CO��g����H=-220kJ?mol-1
�١�2-�ڵã�2H2O��g���TO2��g��+2H2��g����H=��2��H1 +220��kJ?mol-1��
4��462-496-2��436=2��H1 +220��
��á�H1=+130��
�ʴ�Ϊ��+130��
| 1 |
| 2 |
| 1 |
| 2 |
�ʴ�Ϊ��H2��g��+
| 1 |
| 2 |
��2���ٻ��β�����������½���ʹ��Һ��ַ�Ӧ�����ܽ����β����������Ϊͭ˿���������Ϊͭ˿��������ȵ������壬
�ʴ�Ϊ�����½�����ͭ���ȿ죬��ֹ������ʧ��
��50mL 0.50mol?L-1���ᡢ50mL 0.55mol?L-1 NaOH��Һ��������Ϊm=100mL��1g/mL=100g��c=4.18J/��g?�棩��t2-t1=3.4�棬���빫ʽQ=cm��T������0.025mol��ˮ�ų�����Q=4.18J/��g?�棩��100g��3.4��=1421.2J=1.4212KJ��������0.025mol��ˮ�ų�����1.4212KJ����������1mol��ˮ�ų�����Ϊ
| 1.4212KJ��1mol |
| 0.025mol |
�ʴ�Ϊ��-56.8kJ/mol��
��һˮ�ϰ�Ϊ����������Ϊ���ȹ��̣������ð�ˮ����ϡ����������Һ��Ӧ����Ӧ�ų�������С��56.8kJ�����ԡ�H1����H���ʴ�Ϊ������NH3?H2O�������ȣ�
��3����֪��C��s��+H2O��g���TCO��g��+H2��g������H1 ��
��2C��s��+O2��g���T2CO��g����H=-220kJ?mol-1
�١�2-�ڵã�2H2O��g���TO2��g��+2H2��g����H=��2��H1 +220��kJ?mol-1��
4��462-496-2��436=2��H1 +220��
��á�H1=+130��
�ʴ�Ϊ��+130��
���������⿼���Ȼ�ѧ����ʽ����д���к��ȵIJⶨ����˹���ɵ�Ӧ���Լ���ѧ��Ӧ�������ͻ�ѧ������֮��Ĺ�ϵ����Ŀ�ѶȲ���ע���������㹫ʽ��Ӧ����c=4.18J/��g?�棩��Ҫע��������λ�Ļ��㣮

��ϰ��ϵ�д�
�����Ŀ
��ѧ������������أ�����˵����ȷ���ǣ�������
| A������N��PԪ�ص�������ˮ����ֱ���������ũ�� |
| B�����ۡ���ά�ء������ʡ������Ƕ�����Ȼ�߷��ӻ����� |
| C�����ع��͡���Ȼ�������к������ԡ��ع��͡����з�����Ƶ����͡�ú�ͣ��ﵽ���Ϊ����Ŀ�� |
| D�������ʡ���֬�����Ƕ���ˮ�⣬��ˮ����ﶼ����ͬ |
�ڼ��������ܷų���������Һ�У����ܴ���������ǣ�������
| A��NH4+��NO3-��SiO32-��Na+ |
| B��Na+��Ba2+��HCO3-��AlO2- |
| C��NO3-��Mg2+��K+��Cl- |
| D��NO3-��K+��AlO2-��OH- |
���л�ѧ��Ӧ�У������ڵ�������ԭ��Ӧ�ǣ�������
| A��CaO+H2O�TCa��OH��2 | ||||
| B��2Na2O2+2H2O�T4NaOH+O2�� | ||||
| C��2Na+2H2O�T2NaOH+H2�� | ||||
D��C+H2O
|
ij��Ӧ������ʽ��ʾ��mM+4H++O2=mM2++2H2O�������غ�ԭ����ȷ��mֵΪ��������
| A��2 | B��3 | C��4 | D��5 |
���л�ѧʽ�ܴ���һ�ִ�������ǣ�������
| A��C3H7Cl |
| B��C4H10 |
| C��C5H12 |
| D��CH2Br2 |
��ҵ�ϻ���й����ʵķ�����;����ȷ���ǣ�������
| A��������ͨ�����ʯ��ˮ�����Ư�� |
| B����ú���и���ý�̿��ú���͵Ȼ���ԭ�� |
| C����ʯ�ͷ������ϩ����ϩ��1��3-����ϩ |
| D����Ũ����Ͷ������̼��ȷ�Ӧ�Ʊ����� |
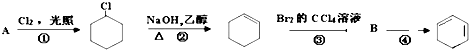


 ��R��R��Ϊ�������⣩
��R��R��Ϊ�������⣩ +R2OH
+R2OH +HCl��R��R��������
+HCl��R��R��������