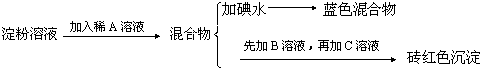
��Ŀ����
��1����������98%��ŨH2SO4����=1.84g/cm3�����Ƴ�0.5mol/L��ϡH2SO4 500mL�IJ������밴Ҫ����գ�
������ŨH2SO4�����Ϊ ��
�����ʵ������15mL��20mL��50mL��Ͳ��Ӧѡ�� mL��Ͳ��ã�
�۽���ȡ��ŨH2SO4���ձ��ڱ�����ע��ʢ��Լ100mLˮ���ձ�������Ͻ��裬Ŀ���� ��
�ܼ�ˮ��Һ��ӽ��̶���1-2cmʱ������ ��ˮ��ʹ��Һ�İ�Һ�����ø��̶�����ƽ��
��2��ijͬѧ������NaOH��Һʱ�����������������������Һ�����ʵ���Ũ���к�Ӱ�죨�ƫ�ߡ�����ƫ�͡����䡱����
�ٶ���ʱ�۲�Һ�温�ӣ� ����NaOH�������沿�ֱ��ʣ� ��
��3��Fe��OH��3������Ʊ�����Ϊ�� ��
������ŨH2SO4�����Ϊ
�����ʵ������15mL��20mL��50mL��Ͳ��Ӧѡ��
�۽���ȡ��ŨH2SO4���ձ��ڱ�����ע��ʢ��Լ100mLˮ���ձ�������Ͻ��裬Ŀ����
�ܼ�ˮ��Һ��ӽ��̶���1-2cmʱ������
��2��ijͬѧ������NaOH��Һʱ�����������������������Һ�����ʵ���Ũ���к�Ӱ�죨�ƫ�ߡ�����ƫ�͡����䡱����
�ٶ���ʱ�۲�Һ�温�ӣ�
��3��Fe��OH��3������Ʊ�����Ϊ��
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺ʵ����
��������1�����ȼ����Ũ��������ʵ���Ũ��Ϊc=
��Ȼ�������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
�ڸ��ݡ����������ԭ������Ҫ��ȡ��Ũ����������ѡ����ʵ���Ͳ��
�۸���Ũ����ϡ�ͷ�����������
�ܶ���Ҫ�ý�ͷ�ιܣ�
��2������c=
��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��3������ʵ�����Ʊ����������������ڷ��ڵ�����ˮ�м��뱥���Ȼ�����Һ��
| 1000�Ѧ� |
| M |
�ڸ��ݡ����������ԭ������Ҫ��ȡ��Ũ����������ѡ����ʵ���Ͳ��
�۸���Ũ����ϡ�ͷ�����������
�ܶ���Ҫ�ý�ͷ�ιܣ�
��2������c=
| n |
| V |
��3������ʵ�����Ʊ����������������ڷ��ڵ�����ˮ�м��뱥���Ȼ�����Һ��
���
�⣺��1����Ũ��������ʵ���Ũ��Ϊc=
=
=18.4mol/L������Ҫ��ŨH2SO4�����ΪVml��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��18.4mol/L��Vml=0.5mol/L��500mL�����V=13.6mL��
�ʴ�Ϊ��13.6mL��
�ڸ��ݡ����������ԭ������Ҫ��ȡ��Ũ��������Ϊ13.6mL����ѡ����ʵ���ͲΪ20mL���ʴ�Ϊ��20��
��Ũ����ϡ�ͷ��ȣ���Ҫ�ò��������Ͻ��裬Ŀ����ʹ��Һ��Ͼ��ȣ�ʹ����Ѹ����ɢ���ʴ�Ϊ��ʹ��Һ��Ͼ��ȣ�ʹ����Ѹ����ɢ��
�ܶ��ݵIJ������ȼ�ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμ�����Һ����̶������У��ʴ�Ϊ����ͷ�ιܣ�
��2���ٶ���ʱ�۲�Һ�温�ӣ��ᵼ����Һ���ƫС��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��NaOH�������沿�ֱ��ʣ�����������ʧ��Ũ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��3��Fe��OH��3������Ʊ������ǣ��������Ȼ�����Һ��ε����ˮ�У�������У�����Һ��Ϊ���ɫ��ֹͣ���ȣ��ʴ�Ϊ���������Ȼ�����Һ��ε����ˮ�У�������У�����Һ��Ϊ���ɫ��ֹͣ���ȣ�
| 1000�Ѧ� |
| M |
| 1000��98%��1.84g/cm3 |
| 98g/mol |
�ʴ�Ϊ��13.6mL��
�ڸ��ݡ����������ԭ������Ҫ��ȡ��Ũ��������Ϊ13.6mL����ѡ����ʵ���ͲΪ20mL���ʴ�Ϊ��20��
��Ũ����ϡ�ͷ��ȣ���Ҫ�ò��������Ͻ��裬Ŀ����ʹ��Һ��Ͼ��ȣ�ʹ����Ѹ����ɢ���ʴ�Ϊ��ʹ��Һ��Ͼ��ȣ�ʹ����Ѹ����ɢ��
�ܶ��ݵIJ������ȼ�ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμ�����Һ����̶������У��ʴ�Ϊ����ͷ�ιܣ�
��2���ٶ���ʱ�۲�Һ�温�ӣ��ᵼ����Һ���ƫС��Ũ��ƫ�ߣ��ʴ�Ϊ��ƫ�ߣ�
��NaOH�������沿�ֱ��ʣ�����������ʧ��Ũ��ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��3��Fe��OH��3������Ʊ������ǣ��������Ȼ�����Һ��ε����ˮ�У�������У�����Һ��Ϊ���ɫ��ֹͣ���ȣ��ʴ�Ϊ���������Ȼ�����Һ��ε����ˮ�У�������У�����Һ��Ϊ���ɫ��ֹͣ���ȣ�
���������⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

��ϰ��ϵ�д�
 �ο�������ϵ�д�
�ο�������ϵ�д� ������ѧ��ʱ��ҵϵ�д�
������ѧ��ʱ��ҵϵ�д� ���������ʱ��ѵϵ�д�
���������ʱ��ѵϵ�д� �㽭�¿γ���άĿ�������ʱ��ѵϵ�д�
�㽭�¿γ���άĿ�������ʱ��ѵϵ�д�
�����Ŀ
����Ƭ�ֱ�����������ʵ���Һ�У���ַ�Ӧ����Һ�����ȷ�Ӧǰ������ǣ�������
| A��CuSO4 |
| B��Fe2��SO4��3 |
| C��HCl |
| D��FeSO4 |
���������Ҫ�ɷ�Ϊ���������������Ͷ������裬��ҵ�Ͼ������й��տ���ұ��������������˵���д�����ǣ�������
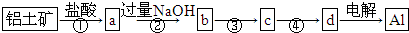
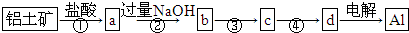
| A���٢��г����Լ��⣬����Ҫ���й��˲��� |
| B��a��b����Ԫ�صĻ��ϼ۲���ͬ |
| C��������Ҫͨ�������CO2 |
| D���ܽ��еIJ����Ǽ��ȣ�����dһ���������� |
����ʵ�������ȷ���ǣ�������
| A��ϡ��Ũ����ʱ���轫�������ձ���ע��ˮ�У��������ò��������� |
| B����������ҩƷʱ��������������ֱ�ӳ��� |
| C����ȡ��ϩʱ���¶ȼƵ�λ��Ӧ�ڻ��Һ�����Ϸ� |
| D���������Դ���ڲ��������� |
����˵������ȷ���ǣ�������
| A�����ۡ���ά�غ���֬��������Ȼ�߷��ӻ����� |
| B���ϳ���ά��������ά��̼��ά�������л��߷��Ӳ��� |
| C�����ǡ����ۡ���֬����ˮ������Ϊ�ǵ���� |
| D��ʯ���ǻ���������Ʒ����ҲΪ����� |
�������������彡����ɽϴ�Σ�����ǣ�������
| A������ˮ��ͨ������Cl2��������ɱ�� |
| B����SO2Ư��ʳƷ |
| C����ʳ����ϴ��ˮƿ���ڱڸ��ŵ�ˮ�� |
| D����С�մ�����������ͷ |
��ͼ��ʾ��ͨ���A��������Ag���Ը�װ�õ��й�������ȷ���ǣ�������
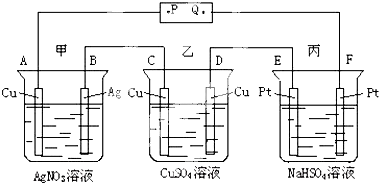

| A��P�ǵ�Դ������ |
| B��F���Ϸ����ķ�ӦΪ��4OH--4e-=2H2O+O2�� |
| C�����ʱ���ס��ҡ��������У���E��F�����⣬����缫���μ��˷�Ӧ |
| D��ͨ��׳ص�PH��С�����ҡ���������Һ��PH���� |