��Ŀ����
25�棬����ĵ���ƽ�ⳣ��Ka�����ʾ������˵����ȷ���ǣ�������
| ���� | CH3COOH | HCN | H2CO3 |
| Ka | 1.8��10-5 | 4.9��10-10 | K1��4.3��10-7 K2��5.6��10-11 |
| A��pH��ͬ��������Һ���ʵ���Ũ�ȹ�ϵ��c��Na2CO3����c��NaCN����c��CH3COONa�� |
| B��a L 0.1mol?L-1CH3COOH��b L 0.1mol?L-1NaOH��Һ��Ϻ�������Һ��pH��7����aһ��С�ڻ����b |
| C�����ʵ���Ũ�Ⱥ��������ͬ��CH3COONa��NaOH������Һ����֪ǰ����Һ��������ĿΪn1��������Һ��������ĿΪn2����n1��n2 |
| D�����ʵ���Ũ����ͬNaHCO3��Na2CO3�Ļ����Һ�У�2c��H+��-2c��OH-��=c��CO32-��-c��HCO3-��-c��H2CO3�� |
���㣺����Ũ�ȴ�С�ıȽ�
ר�⣺
������A������ĵ���ƽ�ⳣ��Խ������Խǿ����֮����ƽ�ⳣ��ԽС������Խ������Ӧ�������������ˮ��̶�Խ����Һ��PHԽ����PH��ͬʱ��Ӧ���ε�Ũ��ԽС��
B��a L 0.1mol?L-1CH3COOH��b L 0.1mol?L-1NaOH��Һ��Ϻ�������Һ��pH��7����Һ�ʼ��ԣ����ݵ���غ�c��H+��+c��Na+��=c��OH-��+c��CH3COO-������c��Na+����c��CH3COO-����a��b��a�Tb��a������bҲ���ϣ�
C�����ݵ���غ�CH3COONa��Һ��c��H+��+c��Na+��=c��OH-��+c��CH3COO-����������ĿΪ�����ӵ�2������NaOH��Һ�е���غ��֪c��H+��+c��Na+��=c��OH-����������ĿΪ�����ӵ�2������CH3COONa��ˮ�ĵ�����ٽ����ã������������������ã�����CH3COONa��Һ��������Ũ�ȴ���NaOH��Һ�������ӵ�Ũ�ȣ�
D�����ݵ���غ�������غ�������
B��a L 0.1mol?L-1CH3COOH��b L 0.1mol?L-1NaOH��Һ��Ϻ�������Һ��pH��7����Һ�ʼ��ԣ����ݵ���غ�c��H+��+c��Na+��=c��OH-��+c��CH3COO-������c��Na+����c��CH3COO-����a��b��a�Tb��a������bҲ���ϣ�
C�����ݵ���غ�CH3COONa��Һ��c��H+��+c��Na+��=c��OH-��+c��CH3COO-����������ĿΪ�����ӵ�2������NaOH��Һ�е���غ��֪c��H+��+c��Na+��=c��OH-����������ĿΪ�����ӵ�2������CH3COONa��ˮ�ĵ�����ٽ����ã������������������ã�����CH3COONa��Һ��������Ũ�ȴ���NaOH��Һ�������ӵ�Ũ�ȣ�
D�����ݵ���غ�������غ�������
���
�⣺A�����ݵ���ƽ�ⳣ����֪����ǿ��˳��Ϊ��CH3COOH��H2CO3��HCN��HCO3-������Խ������Ӧ�������������ˮ��̶�Խ����Һ��PHԽ�����ʵ���Ũ����ͬ�ĸ���ҺpH��ϵΪ��pH��CH3COONa����pH��NaCN����pH��Na2CO3��������pH��ͬ��������Һ���ʵ���Ũ�ȹ�ϵ��CH3COONa��NaCN��Na2CO3����A����
B��a L 0.1mol?L-1CH3COOH��b L 0.1mol?L-1NaOH��Һ��Ϻ�������Һ��pH��7����Һ�ʼ��ԣ����ݵ���غ�c��H+��+c��Na+��=c��OH-��+c��CH3COO-������c��Na+����c��CH3COO-����a��b��a�Tb�����ϣ��������������NaOHҲ���Եģ���B����
C�����ݵ���غ�CH3COONa��Һ��c��H+��+c��Na+��=c��OH-��+c��CH3COO-����������ĿΪ�����ӵ�2������NaOH��Һ�е���غ��֪c��H+��+c��Na+��=c��OH-����������ĿΪ�����ӵ�2������CH3COONa��ˮ�ĵ�����ٽ����ã������������������ã������ƺ�NaOH��������Ũ����ȣ���������������Ũ�ȴ���NaOH�У�����CH3COONa��Һ��������Ũ�ȴ���NaOH��Һ�������ӵ�Ũ�ȣ�����n1��n2����C��ȷ��
D����Һ�д��ڵ���������Na+��H+����������OH-��HCO3-��CO32-��������Һ�ʵ����ԣ�����C��Na+��+c��H+��=c��OH-��+c��HCO3-��+2c��CO32-�����٣��������غ��֪��2C��Na+��=3c��HCO3-��+3c��CO32-��+3c��H2CO3�����ڣ���2����-�ڵã�2c��H+��=2c��OH-��-c��HCO3-��+c��CO32-��-3c��H2CO3��������2c��H+��-2c��OH-��=c��CO32-��-c��HCO3-��-3c��H2CO3������D����
��ѡ��C��
B��a L 0.1mol?L-1CH3COOH��b L 0.1mol?L-1NaOH��Һ��Ϻ�������Һ��pH��7����Һ�ʼ��ԣ����ݵ���غ�c��H+��+c��Na+��=c��OH-��+c��CH3COO-������c��Na+����c��CH3COO-����a��b��a�Tb�����ϣ��������������NaOHҲ���Եģ���B����
C�����ݵ���غ�CH3COONa��Һ��c��H+��+c��Na+��=c��OH-��+c��CH3COO-����������ĿΪ�����ӵ�2������NaOH��Һ�е���غ��֪c��H+��+c��Na+��=c��OH-����������ĿΪ�����ӵ�2������CH3COONa��ˮ�ĵ�����ٽ����ã������������������ã������ƺ�NaOH��������Ũ����ȣ���������������Ũ�ȴ���NaOH�У�����CH3COONa��Һ��������Ũ�ȴ���NaOH��Һ�������ӵ�Ũ�ȣ�����n1��n2����C��ȷ��
D����Һ�д��ڵ���������Na+��H+����������OH-��HCO3-��CO32-��������Һ�ʵ����ԣ�����C��Na+��+c��H+��=c��OH-��+c��HCO3-��+2c��CO32-�����٣��������غ��֪��2C��Na+��=3c��HCO3-��+3c��CO32-��+3c��H2CO3�����ڣ���2����-�ڵã�2c��H+��=2c��OH-��-c��HCO3-��+c��CO32-��-3c��H2CO3��������2c��H+��-2c��OH-��=c��CO32-��-c��HCO3-��-3c��H2CO3������D����
��ѡ��C��
���������⿼��������ʵĵ���ƽ�⣬�漰����Ũ�ȴ�С�Ƚϣ�������ע����������Ե���ƽ���Ӱ�죬���õ���غ㡢�����غ��������Ŀ��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
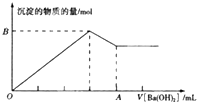 ��֪Ba[Al��OH��4]2������ˮ��ͼ�б�ʾ����100mL0.02mol��L-1KAl��SO4��2��Һ�������0.05mol��L-1Ba��OH��2��Һʱ��25�棩�����ɳ��������ʵ��������Ba��OH��2��Һ������Ĺ�ϵ������˵������ȷ���ǣ�������
��֪Ba[Al��OH��4]2������ˮ��ͼ�б�ʾ����100mL0.02mol��L-1KAl��SO4��2��Һ�������0.05mol��L-1Ba��OH��2��Һʱ��25�棩�����ɳ��������ʵ��������Ba��OH��2��Һ������Ĺ�ϵ������˵������ȷ���ǣ�������| A�����۵�Ba��OH��2��Һ��c��OH-��=0.1mol��L- |
| B��A���ֵ��80mL |
| C��B���ֵ��0.005mol |
| D����V[Ba��OH��2]ʱ�����ɳ�����������0.699g |
���й�������;��˵���У���ȷ���ǣ�������
| A�����������Ե���� |
| B��Һ������������� |
| C���Ȼ��Ƴ������к�θ�� |
| D����������������ʢװϡ���� |
���ڷ�ӦCaH2+2H2OCa��OH��2+2H2�� �������жϣ������ж���ȷ���ǣ�������
��H2 ֻ���������
��H2O ����������
��CaH2�е���Ԫ�ر���ԭ��
�ܴ˷�Ӧ�����������뻹ԭ�����ԭ�Ӹ�������1��1��
��H2 ֻ���������
��H2O ����������
��CaH2�е���Ԫ�ر���ԭ��
�ܴ˷�Ӧ�����������뻹ԭ�����ԭ�Ӹ�������1��1��
| A���٢� | B���ۢ� | C���٢� | D���ڢ� |
������Һ�е�Cl-Ũ����50mL 1mol/L AlCl3��Һ�е�Cl-Ũ����ȵ��ǣ�������
| A��150mL 1mol/L NaCl |
| B��75mL 2mol/L NH4Cl |
| C��150mL 3mol/L KClO3 |
| D��75mL 1.5mol/L MgCl2 |
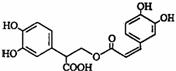 �Ե������Ǵӷ仨��ֲ������ȡ�õ����������ʣ���ṹ����ͼ������������ȷ���ǣ�������
�Ե������Ǵӷ仨��ֲ������ȡ�õ����������ʣ���ṹ����ͼ������������ȷ���ǣ�������| A���Ե��������ڷ����� |
| B��1mol�Ե���������ܺ�1mol���������ӳɷ�Ӧ |
| C���Ե�������Է���ˮ�ⷴӦ�����ܷ���������Ӧ |
| D���Ե�������ֻ��һ������̼ԭ�� |
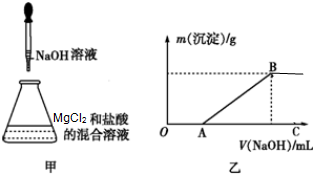 ijͬѧ��ȡһ������MgCl2���������Һ���ڸ���Һ���ȼ���һ������ϡ���ᣬȻ����μ���NaOH��Һ����ͼ����ʾ��
ijͬѧ��ȡһ������MgCl2���������Һ���ڸ���Һ���ȼ���һ������ϡ���ᣬȻ����μ���NaOH��Һ����ͼ����ʾ��