��Ŀ����
6������������Ҫ�Ĺ������ϣ��÷���м�Ʊ������������£��ش��������⣺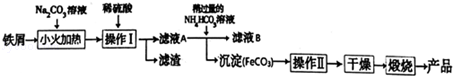
��1���ټ�̼������Һ��С����ȵIJ���Ŀ����ȥ�����ۣ�
�ڲ�����������ǹ��ˣ��������������ϴ�ӣ�
��2���������������ķ�ӦΪFeSO4+2NH4HCO3=FeCO3��+��NH4��2SO4+CO2��+H2O�����У������Թ�����NH4HCO3��Һ����Ҫ������Һ��Ϊ6.8��7.2֮�䣮�����Թ�����Ŀ����ʹ��Һ������������ȫת��ΪFeCO3��������Һ��pH���ܹ��͵�ԭ����̼����������ᷴӦ��̼��������������
�ڼ�����ҺB�к���NH4+�ķ�����ȡ����B��Һ���Թ��У���������Ũ����������Һ�����ȣ�������ʹʪ��ĺ�ɫʯ����ֽ���������壬֤����ҺB�к���笠����ӣ�
��3����Щͬѧ��ΪKMnO4��Һ�ζ��ܽ�����Ԫ�غ����IJⶨ��
a����ȡ2.85g�̷���FeSO4•7H2O����Ʒ�����250mL��Һ��
b����ȡ25.00mL������Һ����ƿ�У�
c�������ữ��0.0100mol/L KMnO4��Һ�ζ����յ㣬����KMnO4��Һ�����ƽ��ֵΪ20.00mL��
��д������KMnO4��Һ��FeSO4��Һ��Ӧ�����ӷ���ʽ5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O��
����KMnO4����Һ�ζ�ʱӦѡ����ʽ�ζ��ܣ����ʽ����ʽ������
�ۼ���������Ʒ��FeSO4•7H2O����������Ϊ97.54%��
���� ̼������Һ�Լ��ԣ����Գ�ȥ��м��������ۣ���ϡ�����ܽ�������ͨ�����˷���õ���ҺA����ҺA�м���̼����泥�����Һ����������ת��ΪFeCO3��������ͨ�����˷�����ҺB��FeCO3����ҺB�к�������泥�FeCO3���渽�����ʣ��ڸ���ǰӦ��ϴ�ӳ�ȥ���ʣ��ڿ��������շֽ⣬��+2�����ڿ����м����ױ�����Ϊ+3�ۣ����յõ�Fe2O3��
��1����̼������Һ�Լ��ԣ����Գ�ȥ��м��������ۣ�
�ڲ���I��õ���������Һ��ӦΪ���ˣ���������ǰӦ��ϴ�ӳ�ȥ���ʣ�
��2���ټ����ٹ�����̼����泥�ʹ��Һ������������ȫת��ΪFeCO3������pH���ͣ�̼����������ᷴӦ��̼�������������٣�
������笠�������Ӧ���ɰ�����������ʹʪ��ĺ�ɫʯ����ֽ������
��3���ٸ����������ǿ�����ԣ��������Ӿ��л�ԭ�ԣ����������£����������������������Ϊ�����ӣ���������ԭΪ�����ӣ�ͬʱ��ˮ���ɣ�
�ڸ��������Һ����ǿ�����ԣ����Ը�ʴ��Ƥ�ܣ�Ӧʢ������ʽ�ζ����У�
�۸���n=cV����25mL��Ʒ��Һ���ĸ�����ص����ʵ�������������250mL��Һ�������ĸ�����ص����ʵ������ٸ��ݢ������ӷ���ʽ�����������ӵ����ʵ�������FeԪ���غ�ɵ�FeSO4•7H2O�����ʵ���������m=nM����FeSO4•7H2O����������������������������
��� �⣺̼������Һ�Լ��ԣ����Գ�ȥ��м��������ۣ���ϡ�����ܽ�������ͨ�����˷���õ���ҺA����ҺA�м���̼����泥�����Һ����������ת��ΪFeCO3��������ͨ�����˷�����ҺB��FeCO3����ҺB�к�������泥�FeCO3���渽�����ʣ��ڸ���ǰӦ��ϴ�ӳ�ȥ���ʣ��ڿ��������շֽ⣬��+2�����ڿ����м����ױ�����Ϊ+3�ۣ����յõ�Fe2O3��
��1����̼������Һ�Լ��ԣ��ٽ�����ˮ�⣬���Գ�ȥ��м��������ۣ�
�ʴ�Ϊ��ȥ�����ۣ�
�ڲ���I���뻥�����ܵĹ�����Һ�壬��ȡ���ˣ�̼���������渽�����ʣ���������ǰӦ��ϴ�ӳ�ȥ���ʣ�
�ʴ�Ϊ�����ˣ�ϴ�ӣ�
��2���ټ����ٹ�����̼����泥�ʹ��Һ������������ȫת��ΪFeCO3������pH���ͣ�̼����������ᷴӦ��̼�������������٣�
�ʴ�Ϊ��ʹ��Һ������������ȫת��ΪFeCO3������̼����������ᷴӦ��̼�������������٣�
�ڼ���笠����ӵ�ʵ�鷽��Ϊ��ȡ����B��Һ���Թ��У���������Ũ����������Һ�����ȣ�������ʹʪ��ĺ�ɫʯ����ֽ���������壬֤����ҺB�к���笠����ӣ�
�ʴ�Ϊ��ȡ����B��Һ���Թ��У���������Ũ����������Һ�����ȣ�������ʹʪ��ĺ�ɫʯ����ֽ���������壬֤����ҺB�к���笠����ӣ�
��3���ٸ����������ǿ�����ԣ��������Ӿ��л�ԭ�ԣ����������£����������������������Ϊ�����ӣ���������ԭΪ�����ӣ�ͬʱ��ˮ���ɣ���Ӧ���ӷ���ʽΪ��5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O��
�ʴ�Ϊ��5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O��
�ڸ��������Һ����ǿ�����ԣ����Ը�ʴ��Ƥ�ܣ���Ϊ�����ữ��Ӧʢ������ʽ�ζ����У��ʴ�Ϊ����ʽ��
��25mL��Ʒ��Һ���ĸ�����ص����ʵ���Ϊ0.02L��0.0100mol/L=2��10-4mol����֪250mL��Һ�������ĸ�����ص����ʵ���Ϊ2��10-4mol��$\frac{250mL}{25mL}$=2��10-3mol����5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O����֪250mL��Һ�������������ӵ����ʵ���Ϊ2��10-3mol��5=0.01mol����FeԪ���غ�ɵ�FeSO4•7H2O�����ʵ���Ϊ0.01mol��
��FeSO4•7H2O����������Ϊ��$\frac{0.01mol��278g/mol}{2.85g}$��100%=97.54%��
�ʴ�Ϊ��97.54%��
���� ����ͨ����ѧ�������̿���̽��������ɡ��������ʺ����ķ�������Ŀ�Ѷ��еȣ���ȷʵ��Ŀ�ġ�ʵ��ԭ��Ϊ���ؼ���ע������Լ��ߡ������ߡ���Ӧ�ߣ��Ƕ�ѧ���ۺ������Ŀ��飬��Ҫѧ���߱���ʵ�Ļ�����

| A�� | �������� | B�� | ���� | C�� | ���� | D�� | ֲ���� |
| A�� | �ᴿ�������ʵĹ�ҵ�Ҵ������õķ��������� | |
| B�� | �����£���������̼ͨ�뱽������Һ����Һ������ | |
| C�� | ������ɳ�ı����ᣬ��ȥ���ʵķ����ǽ����ܽ����ˣ��������ᾧ���� | |
| D�� | ���ͱ���ϡ��Һ�ֱ���Ũ��ˮ��ϣ����߲�����ɫ���� |
| A�� | ���顢�Ҵ������ᶼ�������Ʒ�Ӧ�������� | |
| B�� | ʯ�ͷ���ɻ�ü��顢��ϩ������ | |
| C�� | ������ϩ������ʹ������Ȼ�̼��Һ��ɫ | |
| D�� |  ��֪�Ϻ������������Ա�־����֮-�Ϻ������ᣨ��ͼ�����ϲ����Ĥ�ṹETFE�ɵ�����ϩ���ķ���ϩ�Ӿ۶��ɣ���ṹ��ʽΪ�� 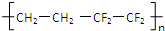 |

| A�� | ����W���� | |
| B�� | b�����Դ���������� | |
| C�� | ���ӽ���ĤcΪ�����ӽ���Ĥ | |
| D�� | a���ĵ缫��ӦΪSO2+2H2O-2e-=SO42-+4H+�� |
| A�� | ����ɫ��Ӧ�ɼ���NaCl��KCl | |
| B�� | ���������ڿ�ʴ������������SiO2��������������� | |
| C�� | ����ͨ����ˮ����ͭ����ĩ������֤��ԭ�����к���ˮ���� | |
| D�� | ��ʳƷ��װ���з���轺��ֹʳƷ���ʣ������ù轺����ˮ�� |
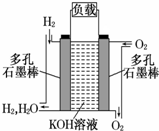 ����Ϊ�ı�δ�������ʮ���¿Ƽ�֮һ��ȼ�ϵ�ؾ�������Ⱦ������������Ч �ʵ��ص㣮��ͼΪ����ȼ�ϵ�صĽṹʾ��ͼ���������ҺΪKOH ��Һ���缫����Ϊ���ɶ��ʯī�����������������ֱ��� �����ϵش�����������ͨ��ȼ�ϵ��ʱ������ڱպϻ�·�в��ϵز����������Իش��������⣺
����Ϊ�ı�δ�������ʮ���¿Ƽ�֮һ��ȼ�ϵ�ؾ�������Ⱦ������������Ч �ʵ��ص㣮��ͼΪ����ȼ�ϵ�صĽṹʾ��ͼ���������ҺΪKOH ��Һ���缫����Ϊ���ɶ��ʯī�����������������ֱ��� �����ϵش�����������ͨ��ȼ�ϵ��ʱ������ڱպϻ�·�в��ϵز����������Իش��������⣺ ��
��