��Ŀ����
ͼ����ĸ�����������ʾ�Ϊ��ѧ��ѧ�������ʡ�����A���ճ������в���ȱ�ٵ����ʣ�Ҳ�ǻ��������ϵ���Ҫԭ�ϣ�������C��D��HΪ���嵥�ʡ�����E��M��NΪ������N�ǵؿ��к������Ľ���Ԫ�ء�Y�Ǻ��ɫ��������Щ������һ�������´�������ת�� ��ϵ��������Щ��Ӧ����������Ѿ���ȥ���Իش��������⣺
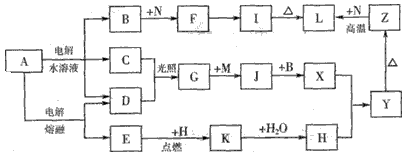
��1�� ��ҵ�ϣ��ڵ��A��Һ���豸�н�����������������________________������
��2��Z��L��Ӧ��������____________________��K�ĵ���ʽΪ__________________��
��3��д��B��F�����ӷ���ʽ________________________��
��4��д��K��CO2��Ӧ�Ļ�ѧ����ʽ___________________��
��5�� Y��NaClO��B�����Һ���ã����Ʊ���ɫˮ������(Na2MO4)��һ�ַ�������д���йط�Ӧ�����ӷ���ʽ________________________________��
��2��Z��L��Ӧ��������____________________��K�ĵ���ʽΪ__________________��
��3��д��B��F�����ӷ���ʽ________________________��
��4��д��K��CO2��Ӧ�Ļ�ѧ����ʽ___________________��
��5�� Y��NaClO��B�����Һ���ã����Ʊ���ɫˮ������(Na2MO4)��һ�ַ�������д���йط�Ӧ�����ӷ���ʽ________________________________��
��1�������ӽ���Ĥ��
��2�����ȷ�Ӧ��
��3��
��4��Fe(OH)3+3ClO-+4OH-===2FeO42-+3Cl-+5H2O
��2�����ȷ�Ӧ��

��3��

��4��Fe(OH)3+3ClO-+4OH-===2FeO42-+3Cl-+5H2O

��ϰ��ϵ�д�
�����Ŀ



