��Ŀ����
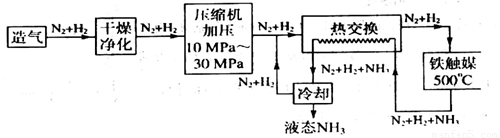
�������̣�
��11�֣������������γɵĸ�����ʯ��(CaSO4��2H2O)ת��Ϊ����ط��Ϻ��Ȼ���ˮ���ﴢ�Ȳ��ϣ����۴Ӿ���Ч�桢��Դ�ۺ����û��Ǵӻ��������Ƕȿ���������Ҫ���塣������ʯ��ת��Ϊ����غ��Ȼ��ƵĹ�������ʾ��ͼ��

(1)�����������õ�ԭ�ϳ�CaSO4��2H2O��KCl�⣬����Ҫ ��ԭ�ϡ�
(2)д��ʯ������Һ�м���̼�����Һ������Ӧ�����ӷ���ʽ�� _______ ��
(3)����I�������ù����У���CaCO3����� (�ѧʽ)�����ʣ��ù������������ˮ���ԭ�ϡ�
(4)����I����������Һ��(NH4)2SO4��Һ��������Һ�к���CO32 ���ķ����ǣ� ��
(5)�Ȼ��ƽᾧˮ����(CaCl2��6H2O)��Ŀǰ���õ������Ȳ��ϣ�ѡ���������
a���۵�ϵ�(29 ���ۻ�) b���ܵ��� c�������� d����
(6) ��������������������ɫ��ѧ������ǣ�___________________________��
�ο��𰸣���11�֣�(1)CaCO3�� NH3�� H2O�� ����1�֣�
(2)CaSO4��CO32 �� = CaCO3����SO42 ����2�֣� (3)CaSO4��1�֣�
(4)��Һ�еμ�����������ݡ� ��1�֣� (5) a�� d�� ����1�֣�
(6)̼���������ˮ��ԭ�ϡ�����ƣ��Ȼ���ת��Ϊ����غ��Ȼ��ƣ����ڹ�����ѭ��ʹ�õ�(ԭ�������ʸߣ�û���к������ŷŵ�������)��2�֣�