��Ŀ����
13��ij��A 0.2mol�������г��ȼ�պ����ɻ�����B��C��1.2mol���Իش���1����A�ķ���ʽ��C6H12��
��2����ȡһ��������A���ȼ�պ�����B��C��3mol������42g��A�μ��˷�Ӧ��ȼ��ʱ���ı�״���µ�����100.8L��
��3������A��ʹ��ˮ��ɫ���ڴ�����������H2�����ӳɷ�Ӧ������
 ����A�Ľṹ��ʽΪ��CH3��3CCH=CH2��
����A�Ľṹ��ʽΪ��CH3��3CCH=CH2����4������A��ʹ��ˮ��ɫ���ҷ���������̼ԭ�ӹ�ƽ�棬��A�Ľṹ��ʽΪ��CH3��2C=C��CH3��2��
���� ��1��������C��H����Ԫ�أ�ij��A0.2mol�������ij��ȼ�պ����ɻ�����B��C��1.2mol��������CO2��H2O��1.2mol����1mol���к���6molC��12molHԭ�ӣ�A����ʽΪC6H12��
��2������ȼ�շ���ʽ����A�����ʵ������������������ʵ������ٸ���m=nM������A������������V=nVm�������������������
��3������A��ʹ��ˮ��ɫ���ڴ�����������H2�ӳ����� ��˵�������к���1��C=C������������̼ԭ�Ӷ�����Hԭ��ΪC=C˫��λ�ã��ݴ�ȷ��A�Ľṹ��ʽ��
��˵�������к���1��C=C������������̼ԭ�Ӷ�����Hԭ��ΪC=C˫��λ�ã��ݴ�ȷ��A�Ľṹ��ʽ��
��4������A��ʹ��ˮ��ɫ���ҷ���������̼ԭ�ӹ�ƽ�棬��C=C˫���в�����Cԭ�������ĸ������ݴ�ȷ��A�Ľṹ��ʽ��
��� �⣺��1��������C��H����Ԫ�أ�ij��A0.2mol�������ij��ȼ�պ����ɻ�����B��C��1.2mol��������CO2��H2O��1.2mol����1mol���к���6molC��12molHԭ�ӣ�����ʽΪC6H12��
�ʴ�Ϊ��C6H12��
��2��C6H12��ȫȼ�գ�����3molCO2��H2O����
C6H12+9O2$\frac{\underline{\;��ȼ\;}}{\;}$6CO2+6H2O��
1mol 9mol 6mol 6mol
0.5mol 4.5mol 3mol 3mol
������3molCO2��H2Oʱ����Ҫ0.5molC6H12��m��C6H12��=0.5mol��84g/mol=42g��
��Ҫ���������ΪV��O2��=4.5mol��22.4L/mol=100.8L��
�ʴ�Ϊ��42��100.8��
��3������A��ʹ��ˮ��ɫ���ڴ�����������H2�ӳ����� ��˵�������к���1��C=C������������̼ԭ�Ӷ�����Hԭ��ΪC=C˫��λ�ã���A�Ľṹ��ʽΪ����CH3��3CCH=CH2��
��˵�������к���1��C=C������������̼ԭ�Ӷ�����Hԭ��ΪC=C˫��λ�ã���A�Ľṹ��ʽΪ����CH3��3CCH=CH2��
�ʴ�Ϊ����CH3��3CCH=CH2��
��4������A��ʹ��ˮ��ɫ���ҷ���������̼ԭ�ӹ�ƽ�棬��C=C˫���в�����Cԭ�������ĸ�������A�Ľṹ��ʽΪ����CH3��2C=C��CH3��2��
�ʴ�Ϊ����CH3��2C=C��CH3��2��
���� ���⿼���л������ʽȷ������������ͬ���칹�����д����Ŀ�Ѷ��еȣ�ע������ϩ���Ľṹ�����ʣ�

| A�� | ���¡���ѹ | B�� | ���¡���ѹ | C�� | ���¡���ѹ | D�� | ���¡���ѹ |
| A�� | FeS2����ԭ������Ԫ�ر���ԭ | |
| B�� | Fe2O3��SO2�����������������ǻ�ԭ���� | |
| C�� | ÿ����1 mol Fe2O3����4mol������ | |
| D�� | ÿ1 mol��������ת�Ƶ���5.5mol |
| A�� | ��ѧ��Դ��һ�ε�ء����ε�غ�ȼ�ϵ�صȣ�һ�ε��ֻ�ܷŵ磬���ܳ�� | |
| B�� | Ǧ����Ӧ�ù㷺����һ�ֳ����Ŀɳ���أ��ŵ�����е������Һ�ܶ������� | |
| C�� | ȼ�ϵ�ؾ������������ʸߡ�������ʹ�ú���Ⱦ����ŵ� | |
| D�� | ﮵����һ�ָ��ܵ�أ����С�������ᣬ��λ�����缫�ų��ĵ��ܶ� |
| X | Y | |
| Z | W | |
| T |
| A�� | �⻯���ȶ���Y��Wǿ������ΪY���⻯���д������ | |
| B�� | XY2��XW2��ZY2�۷е������������ɹ��ۼ��γɵķ��Ӿ��� | |
| C�� | һ�������£�X��������ZY2��Ӧ����Z���ʣ�˵��X�ķǽ����Ա�Zǿ | |
| D�� | ����Ԫ�������ɣ������Ʋ�TԪ�صĵ��ʾ��а뵼�����ԣ�������T�γ�Na2T2������ |
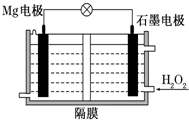 Mg-H2O2��ؿ������������˼�ʻ��DZ�������õ���Ժ�ˮΪ�������Һ��ʾ��ͼ���£��õ�ع���ʱ������˵����ȷ���ǣ�������
Mg-H2O2��ؿ������������˼�ʻ��DZ�������õ���Ժ�ˮΪ�������Һ��ʾ��ͼ���£��õ�ع���ʱ������˵����ȷ���ǣ�������| A�� | Mg�缫�Ǹõ�ص����� | B�� | H2O2��ʯī�缫�Ϸ���������Ӧ | ||
| C�� | ʯī�缫������Һ��pH���� | D�� | ���Ӵ�ʯī�缫����Mg�缫 |
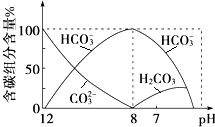 �ں���������ʵ���Һ�У������ж����ѧƽ��森
�ں���������ʵ���Һ�У������ж����ѧƽ��森