��Ŀ����
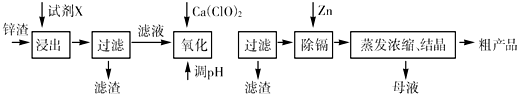
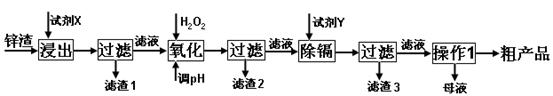
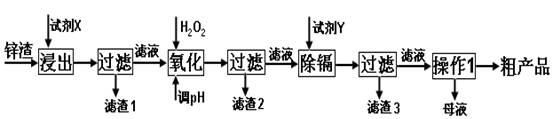
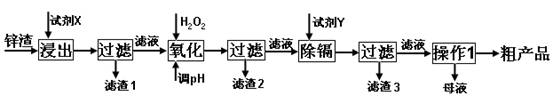
��16�֣���п�������Ĺ�ҵ��������п�������˺�Zn�⣬������Fe��Al��Cd��SiO2�����ʣ�������п����ȡ������ZnSO4��7H2O�ͽ�������һ������ij��ԣ����������£�
��֪��Fe3+��Al3+��Zn2+��Cd2+��Fe2+������������ȫ����ʱ��pH�ֱ�Ϊ��3.2��4.7��6.5��9.4��9.7��п�Ľ�����Ա���ǿ��
�Իش��������⣺
��1����������ʱ�õ��ġ��Լ�X��Ϊ (������)��
��2��д�������������̵����ӷ���ʽ ��
��3������pH�����̿���ѡ�� ����������ѡ��ѡ�� ����ţ�
A��H2SO4 B��ZnO C��NaOH
������2������Ҫ�ɷ��� (�ѧʽ����ͬ)��
��4�����Լ�Y����______________��������3���ijɷ�Ϊ______________________��
��5��������1���ķ�����___ ____���ڡ�����1��ʱ�������ȡ��ʵ���ʩ�� ��
��16�֣�ÿ��2�֣�
��1��������Һ ��2��2H++H2O2+2Fe2+ =2Fe3++2H2O
��3��B��Fe(OH)3 Al(OH)3 ��4��Zn ��Cd��Zn
��5������Ũ���������ᾧ�����ˣ�ϴ�ӣ���������Һһ�������
����������