��Ŀ����
AԪ��ԭ��M���Ӳ�����7�����ӣ�BԪ����AԪ��λ��ͬһ���ڣ�BԪ�ص�ԭ��������Ӳ�ֻ��1�����ӣ�
��1������BԪ�ص�ԭ�ӽṹʾ��ͼ
��2��A��B��Ԫ���γɻ������������ ������ʽ�ǣ� ��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ�Ԫ�أ�AΪ BΪ CΪ D������������������ȣ�D ��ԭ����ɷ���Ϊ�� ����������������Ӧ��ˮ��� ��
��3���õ���ʽ��ʾMgCl2���γɹ��� �õ���ʽ��ʾH2O�γɹ��� ��
��1������BԪ�ص�ԭ�ӽṹʾ��ͼ
��2��A��B��Ԫ���γɻ������������
��3���õ���ʽ��ʾMgCl2���γɹ���
���㣺λ�ýṹ���ʵ����ϵӦ��,Ԫ�����ڱ��Ľṹ����Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
������AԪ��ԭ��M���Ӳ�����7�����ӣ���AΪCl��BԪ����AԪ��λ��ͬһ���ڣ�BԪ�ص�ԭ��������Ӳ�ֻ��1�����ӣ���BΪNa��
��1��Na��ԭ������Ϊ11��ԭ�ӽṹ����3�����Ӳ㣬����������Ϊ1��
��2��A��B��Ԫ���γɻ�����ΪNaCl��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��
��3��MgCl2Ϊ���ӻ����H2OΪ���ۻ����
��1��Na��ԭ������Ϊ11��ԭ�ӽṹ����3�����Ӳ㣬����������Ϊ1��
��2��A��B��Ԫ���γɻ�����ΪNaCl��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��
��3��MgCl2Ϊ���ӻ����H2OΪ���ۻ����
���
�⣺AԪ��ԭ��M���Ӳ�����7�����ӣ���AΪCl��BԪ����AԪ��λ��ͬһ���ڣ�BԪ�ص�ԭ��������Ӳ�ֻ��1�����ӣ���BΪNa��
��1��Na��ԭ������Ϊ11��ԭ�ӽṹ����3�����Ӳ㣬����������Ϊ1��ԭ�ӽṹʾ��ͼΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2��A��B��Ԫ���γɻ�����ΪNaCl��Ϊ�Ȼ��ƣ�����ʽΪ ��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��D������������������ȣ�D ��ԭ����ɷ���Ϊ
��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��D������������������ȣ�D ��ԭ����ɷ���Ϊ
S��������������Ӧ��ˮ����ΪH2SO4��
�ʴ�Ϊ���Ȼ��ƣ� ��Cl��Na��O��
��Cl��Na��O��
S��H2SO4��
��3��MgCl2Ϊ���ӻ�����õ���ʽ��ʾ���γɹ���Ϊ ��H2OΪ���ۻ������ԭ�Ӵﵽ�ȶ��ṹ���õ���ʽ��ʾ�γɹ���Ϊ
��H2OΪ���ۻ������ԭ�Ӵﵽ�ȶ��ṹ���õ���ʽ��ʾ�γɹ���Ϊ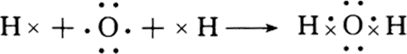 ��
��
�ʴ�Ϊ�� ��
��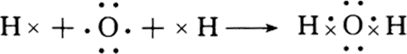 ��
��
��1��Na��ԭ������Ϊ11��ԭ�ӽṹ����3�����Ӳ㣬����������Ϊ1��ԭ�ӽṹʾ��ͼΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����2��A��B��Ԫ���γɻ�����ΪNaCl��Ϊ�Ȼ��ƣ�����ʽΪ
 ��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��D������������������ȣ�D ��ԭ����ɷ���Ϊ
��B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ������һ�ֵ���ɫ�Ĺ���E��EΪ�������ƣ���CΪO��D��L�����������K��M�������Ӳ��ϵ�����֮�ͣ���M�������Ϊ8-2=6����DΪS��D������������������ȣ�D ��ԭ����ɷ���Ϊ32 16 |
�ʴ�Ϊ���Ȼ��ƣ�
 ��Cl��Na��O��
��Cl��Na��O��32 16 |
��3��MgCl2Ϊ���ӻ�����õ���ʽ��ʾ���γɹ���Ϊ
 ��H2OΪ���ۻ������ԭ�Ӵﵽ�ȶ��ṹ���õ���ʽ��ʾ�γɹ���Ϊ
��H2OΪ���ۻ������ԭ�Ӵﵽ�ȶ��ṹ���õ���ʽ��ʾ�γɹ���Ϊ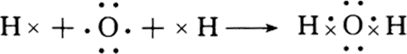 ��
���ʴ�Ϊ��
 ��
��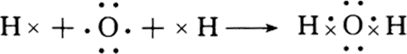 ��
��
���������⿼��λ�á��ṹ�����ʵ��ۺ�Ӧ�ã�Ϊ��Ƶ���㣬����ԭ�ӽṹ�ƶ�Ԫ��Ϊ���Ĺؼ������ػ�ѧ����Ŀ��飬ע�ػ���֪ʶ�ĺ�ʵ��ѵ������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
�����Ŀ
2g AO32-�к������������������3.01��1022������Ԫ��A��Ħ�������ǣ�������
| A��12g/mol |
| B��32g |
| C��80g/mol |
| D��32g/mol |

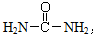 ����д�����ص�ͬ���칹���к������Ӽ��Ļ�ѧʽ
����д�����ص�ͬ���칹���к������Ӽ��Ļ�ѧʽ