��Ŀ����
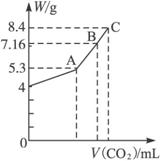
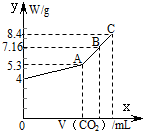
��100 mL NaOH��Һ��ͨ��CO2����ַ�Ӧ���ڼ�ѹ�ͽϵ��¶��£�С�Ľ���Һ���ɣ��ð�ɫ����M��ͨ��CO2�����V(��λ��mL���ڱ�״���²ⶨ����ͬ)��M������W(��λ��g)�Ĺ�ϵ��ͼ1-5-26��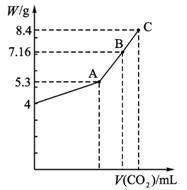
ͼ1-5-26
(1)A��ʱ����ɫ����M�Ļ�ѧʽΪ__________��ͨ��CO2�����Ϊ__________mL��
(2)C��ʱ����ɫ����M�Ļ�ѧʽΪ__________��ͨ��CO2�����Ϊ__________mL��
(3)����B��ʱM�����(�û�ѧʽ��ʾ)��ͨ��CO2����������
����:(1)��ͼ��֪NaOH����Ϊ4 g�����ʵ���Ϊ0.1 mol����ȫת��ΪNa2CO3ʱ��̼���Ƶ�����Ϊ5.3 g����CO2���Ϊ![]() ��22.4 L��mol-1=1.12 L��
��22.4 L��mol-1=1.12 L��
(2)��ȫת��ΪNaHCO3ʱ��NaHCO3����Ϊ8.4 g����CO2�����Ϊ0.1 mol��22.4 L��mol-1=2.24 L��?
(3)ͼ��B��ʱM������Ϊ7.16 g,5.3��7.16��8.4,֪M��Na2CO3��NaHCO3��ɡ�����B��ʱNa2CO3�����ʵ���Ϊx��NaHCO3�����ʵ���Ϊy�����У�
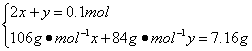 ���
���![]()
V(CO2)=(0.02+0.06) mol��22 400 mL��mol-1=1 792 mL
��:(1)Na2CO3 1 120 mL (2)NaHCO3 2 240 mL (3)Na2CO3��NaHCO3 1 792 mL

��ϰ��ϵ�д�
�����Ŀ
��100 mL 1mol/L��AlCl3��Һ�еμ�1mol/L��NaOH��Һ���õ�5.85g�����������NaOH��Һ�����������
| A��200 mL | B��225mL | C��300 mL | D��325 mL |