��Ŀ����
5�� ��ѧ��ȤС���趨����ʵ�鷽�����ⶨij�ѱ���Ϊ̼���Ƶ�С�մ���Ʒ��NaHCO3������������
��ѧ��ȤС���趨����ʵ�鷽�����ⶨij�ѱ���Ϊ̼���Ƶ�С�մ���Ʒ��NaHCO3����������������һ����ȡһ��������Ʒ�����������м��������أ���ȴ������ʣ��������������㣮
��1�������з�����Ӧ�Ļ�ѧ����ʽΪ��2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2��
��2��ʵ��������������ص�Ŀ���ǣ���֤NaHCO3�ֽ���ȫ
����������ȡһ��������Ʒ������С�ձ��У�������ˮ�ܽ⣻��С�ձ��м�������Ba��OH��2��Һ�����ˣ�ϴ�ӣ���������������������������㣮
��1�����˲����У������ձ���©���⣬��Ҫ�õ��IJ�������Ϊ������
��2��ʵ�����жϳ����Ƿ���ȫ�ķ�����ȡ�����ϲ���Һ���Թ��У��μ�һ��Ba��OH��2��Һ���۲��Ƿ��а�ɫ��������
������������ͼװ�ý���ʵ�飺
��1��Bװ������ʢ�Լ���Ũ���Dװ�õ������Ƿ�ֹ�����е�ˮ������������̼����Cװ�ã���Һ©���в��ܣ���ܡ����ܡ������������ϡ�������ʵ�飮
��2��ʵ��ǰ��ȡ17.9g��Ʒ��ʵ�����Cװ������8.8g������Ʒ��NaHCO3����������Ϊ70.4%��
��3�����ݴ�ʵ���õ����ݣ��ⶨ���������Ϊʵ��װ�û�����һ������ȱ���ǣ�ȱ��һ��A��Bװ���ڵ�CO2�������ϵ�Cװ���е�װ�ã�
���� [����һ]̼�����Ʋ��ȶ��������ֽ⣬���ݼ���ǰ����������仯�����ݲ�������̼�����Ƶ��������������̼���Ƶ�����������
��1��̼���������ȷֽ�����̼���ơ�ˮ�Ͷ�����̼��
��2�����Ⱥ��ر�֤̼��������ȫ�ֽ⣻
[������]̼���ƺ�̼�����ƶ�������������Ӧ����̼�ᱵ�������������ɳ���������������̼���Ƶ�����������
��1�����ݹ��˲�����������������
��2����ȡ�ϲ���Һ�������ӳ����������Ƿ����ɳ�����
[������]��3���ɲⶨ������ʵ���֪��A�з���Na2CO3+H2SO4=H2O+CO2��+Na2SO4��2NaHCO3+H2SO4=Na2SO4+2H2O+2CO2����B��ΪŨ��������ˮ�����������̼������Cװ�����ն�����̼��Dװ�÷�ֹ�����еĶ�����̼��ˮ����Cװ�ø��ź����ⶨ��
��4����Ϸ�Ӧ������ϵ��Ԫ���غ����õ����ʵ����������������ʺ�����
��5��������̼���岻��ȫ������Cװ�ñ����գ���Ҫ����һ��װ�ö�����̼�������װ��C��װ�ã�
��� �⣺[����һ]��1��̼���������ȷֽ�����̼���ơ�ˮ�Ͷ�����̼����Ӧ����ʽΪ��2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2����
�ʴ�Ϊ��2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2����
��2��ʵ��ԭ���Ǹ��ݼ���ǰ����������仯������̼�����ƣ���Ӧ��֤̼��������ȫ�ֽ⣬���Ⱥ�����̼��������ȫ�ֽ⣬
�ʴ�Ϊ����֤NaHCO3�ֽ���ȫ��
[������]��1������ʱ���ò������������ʴ�Ϊ����������
��2����ȡ�ϲ���Һ�������ӳ����������Ƿ����ɳ������������Ϊ��ȡ�ϲ���Һ�������Թ��У���������������Һ�����������ɣ��������ȫ��
�ʴ�Ϊ��ȡ�����ϲ���Һ��һ֧�Թ��У��μ�Ba��OH��2��Һ���۲��Ƿ��а�ɫ�������ɣ�
[������]��1��B��ΪŨ��������ˮ�����������̼�������е�ˮ�����Ͷ�����̼�ᱻ��ʯ�����գ���D�����������տ����е�ˮ�����Ͷ�����̼����ȷ��Cװ����������������ȷ�ԣ���Һ©�������������������ᣬ�����ӷ��������Ƶö�����̼�����к��Ȼ��⣬Ũ����������Ȼ��⣬���Ȼ��ⱻ��ʯ�����գ����²������̼����ƫ�ߣ�������̼���ƺ�̼�����ƣ�̼�����Ʋ���������̼�࣬��ᵼ��̼������ƫ�̼࣬����ƫС��
�ʴ�Ϊ��Ũ�����ֹ�����е�ˮ������������̼����Cװ�ã����ܣ�
��2����Na2CO3��NaHCO3�����ʵ����ֱ�Ϊx��y����
Na2CO3+H2SO4=H2O+CO2��+Na2SO4��
1 1
x x
2NaHCO3+H2SO4=Na2SO4+2H2O+2CO2��
2 2
y y
106x+84y=17.90
44x+44y=8.80
���x=0.05mol
y=0.15mol
����Ʒ��NaHCO3����������Ϊ=$\frac{0.15mol��84g/mol}{17.9g}$��100%=70.4%��
�ʴ�Ϊ��70.4%��
��3��ʵ��װ�û�����һ������ȱ��Ϊװ���еĶ�����̼���ܱ�Cȫ�����գ��������һ��װ�ý�A��B�е�CO2ȫ������C�����գ�
�ʴ�Ϊ��ȱ��һ��A��Bװ���ڵ�CO2�������ϵ�Cװ���е�װ�ã�
���� ���⿼��̼���ƺ����IJⶨʵ�飬Ϊ��Ƶ���㣬����ʵ��װ�õ����ü�ʵ��Ŀ��Ϊ���Ĺؼ������ط��������㼰ʵ���������ۺϿ��飬��Ŀ�Ѷ��еȣ�

 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д� �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�| A�� | ��״���£�22.4L SO3�к��еķ�����ΪNA�� | |
| B�� | 2L 0.5 mol•L-1��������Һ�к��е�H+������Ϊ2NA | |
| C�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.2NA | |
| D�� | �ܱ�������2mol NO��1mol O2��ַ�Ӧ������ķ�����Ϊ2NA |
| A�� |  ���������� | B�� |  ������������ | C�� | 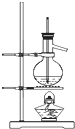 ʯ�͵����� | D�� | 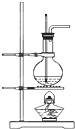 ʵ��������ϩ |
| A�� | 1molH3PO4������Ϊ98g•mol-1 | B�� | H3PO4��Ħ������Ϊ98g | ||
| C�� | 9.8g H3PO4����NA��H3PO4���� | D�� | NA��H3PO4���ӵ�����Ϊ98g |
| A�� | �������Ż�������ˮ���� | |
| B�� | ϡ��Ũ����ʱ����Ũ��������������ע��ˮ�� | |
| C�� | Ƥ���ϲ���մ��NaOH��Һ�������������ϴ | |
| D�� | ʵ����������촵��ƾ��� |
| A�� | ��ѧ����ʵ��Ϊ��������Ȼ��ѧ | |
| B�� | ��ѧ��һ�Ŵ����Եġ�ʵ���ԵĿ�ѧ | |
| C�� | ����Ļ�ѧʵ�����ʼ�ڽ��� | |
| D�� | �ִ���ѧ���ɳ�֮Ϊ21���͵����Ŀ�ѧ |
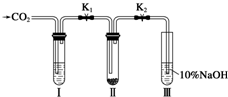 ij�����о�С���ͬѧ�ڲ�������ʱ��֪��Na2O2������CO2���ܷ�����Ӧ����������ˮ����ʱ��Na2O2����CO2������Ӧ����Na2CO3��O2��Ϊ��̽����������̼�Ƿ�����ˮ����ʱ������������Ʒ�Ӧ����ij�����о�С���ͬѧ���������ͼ��ʾ��ʵ��װ�ã��ֱ���мס�������ʵ�飺
ij�����о�С���ͬѧ�ڲ�������ʱ��֪��Na2O2������CO2���ܷ�����Ӧ����������ˮ����ʱ��Na2O2����CO2������Ӧ����Na2CO3��O2��Ϊ��̽����������̼�Ƿ�����ˮ����ʱ������������Ʒ�Ӧ����ij�����о�С���ͬѧ���������ͼ��ʾ��ʵ��װ�ã��ֱ���мס�������ʵ�飺