��Ŀ����
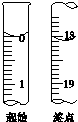 Na2SO3���ױ�������Na2SO4�����ʣ�ij��ѧ��ȤС��Ϊ�˲ⶨʵ���Ҵ�ŵ�Na2SO3�Ĵ��ȣ����費���������ʣ�����������ʵ�飮
Na2SO3���ױ�������Na2SO4�����ʣ�ij��ѧ��ȤС��Ϊ�˲ⶨʵ���Ҵ�ŵ�Na2SO3�Ĵ��ȣ����費���������ʣ�����������ʵ�飮ȷ��ȡ��Ʒm g��������ˮ�ܽ⣬���250mL��Һ��ȷ��ȡ25.00mL����0.1000mol/L��ı���Һ�ζ����ζ�������Ϊ���²��裺
��ȷ��ȡ25.00mL�����Na2SO3��Һע��ྻ����ƿ��������2��3��ָʾ��
���ñ�Һ��Һ��ϴ�ζ���2��3��
�۰�ʢ�б���Һ����ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ
��ȡ������Һע����ʽ�ζ�����0�̶�����
�ݵ���Һ����0��0�̶����£����¶���
�ް���ƿ���ڵζ��ܵ��·����ñ�����Һ�ζ����յ㣬���µζ���Һ��Ŀ̶��ظ����ϲ���2��3�Σ�
�ش��������⣺
��1����ȷ����˳���ǣ�
��2������ҺӦ����
��3��ѡ��
��4������һ�εζ�����ʼ���յ�Һ��������ʾ�������ı���Һ�����V=mL����Ʒ��Na2SO3����������Ϊ
��5������ʧ�������ʹʵ����ƫ�����
A���ζ�ǰ���ô���Һ��ϴ��ƿ
B���ζ�ǰ��δ�ô���Һ��ϴ�ζ���
C���ζ�����ʱ�����Ӷ���
D�����Ʊ�����Һ����ʱ�����ӿ̶���
��6�����ζ�����ʱ���������ʹʵ����ƫС��ԭ���ǣ�
���û�ѧ��Ӧ����ʽ���ͣ���
��7�����������һ��ʵ�鷽�����ⶨNa2SO3�Ĵ��ȣ���Ҫ˵��ʵ�鲽�衢�����Լ��Լ��������ݣ�
���㣺�к͵ζ�,̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ����
��������1���ζ�ʵ���������©��ϴ�ӡ���ϴ��װҺ��ȡ����Һ��ָʾ�����ζ��Ȳ�����
��2��Na2SO3����Һ��ˮ���Լ��ԣ��ü�ʽ�ζ�����ȡ��
��3���ⵥ���������ۻ����ɫ���õ�����ָʾ�����ⵥ����Na2SO3��Ӧ����Na2SO3��ȫ��Ӧ���ټ�һ�ε���Һ����Һ����ɫ��
��4�����ı���Һ�����Ϊ���ζ��յ�Ķ���-�ζ�ǰ�Ķ��������ݷ�Ӧ�ķ���ʽ�����Na2SO3���ʵ��������������������������
��5����Na2SO3+I2+H2O=Na2SO4+2HI��֪n��Na2SO3��=n��I2��=cV��
A���ζ�ǰ���ô���Һ��ϴ��ƿ������ƿ��Na2SO3���ʵ���ƫ��
B���ζ�ǰ��δ�ô���Һ��ϴ�ζ��ܣ���Na2SO3Ũ�ȱ�С��
C���ζ�����ʱ�����Ӷ���������ƫС��
D�����Ʊ�����Һ����ʱ�����ӿ̶��ߣ�����Һ���ƫ���Ũ��ƫС��
��6��Na2SO3����Һ���ױ�����������
��7��ȷ��ȡ��Ʒm1g���ܽ����Һ����������Ȼ������������ᱵ�������������ᱵ��������ԭ���غ�����������������������Na2SO3������������������
��2��Na2SO3����Һ��ˮ���Լ��ԣ��ü�ʽ�ζ�����ȡ��
��3���ⵥ���������ۻ����ɫ���õ�����ָʾ�����ⵥ����Na2SO3��Ӧ����Na2SO3��ȫ��Ӧ���ټ�һ�ε���Һ����Һ����ɫ��
��4�����ı���Һ�����Ϊ���ζ��յ�Ķ���-�ζ�ǰ�Ķ��������ݷ�Ӧ�ķ���ʽ�����Na2SO3���ʵ��������������������������
��5����Na2SO3+I2+H2O=Na2SO4+2HI��֪n��Na2SO3��=n��I2��=cV��
A���ζ�ǰ���ô���Һ��ϴ��ƿ������ƿ��Na2SO3���ʵ���ƫ��
B���ζ�ǰ��δ�ô���Һ��ϴ�ζ��ܣ���Na2SO3Ũ�ȱ�С��
C���ζ�����ʱ�����Ӷ���������ƫС��
D�����Ʊ�����Һ����ʱ�����ӿ̶��ߣ�����Һ���ƫ���Ũ��ƫС��
��6��Na2SO3����Һ���ױ�����������
��7��ȷ��ȡ��Ʒm1g���ܽ����Һ����������Ȼ������������ᱵ�������������ᱵ��������ԭ���غ�����������������������Na2SO3������������������
���
�⣺��1���ζ�ʵ�������
���ñ�Һ��Һ��ϴ�ζ���2��3�Σ�
��ȡ������Һע����ʽ�ζ�����0�̶����ϣ�
�۰�ʢ�б���Һ����ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ��
�ݵ���Һ����0��0�̶����£����¶�����
��ȷ��ȡ25.00mL�����Na2SO3��Һע��ྻ����ƿ��������2��3��ָʾ����
�ް���ƿ���ڵζ��ܵ��·����ñ�����Һ�ζ����յ㣬���µζ���Һ��Ŀ̶��ظ����ϲ���2��3�Σ�����ȷ����˳���ǣ��ڢܢۢݢ٢ޣ�
�ʴ�Ϊ���ڢܢۢݢ٢ޣ�
��2��Na2SO3����Һ��ˮ���Լ��ԣ�������Һ�ü�ʽ�ζ�����ȡ���ʴ�Ϊ���
��3����0.1000mol/L��ı���Һ�ζ�Na2SO3��Һʱ���ⵥ���������ۻ����ɫ���õ�����ָʾ�����ζ�ʱ�ⵥ����Na2SO3��Ӧ����Na2SO3��ȫ��Ӧ���ټ�һ�ε���Һ����ƿ�е���Һ������ɫ����30s�ڲ���ɫ��
�ʴ�Ϊ�����ۣ����������һ�ε�Һʱ����ƿ�е���Һ������ɫ����30s�ڲ���ɫ��
��4�����ı���Һ�����Ϊ���ζ��յ�Ķ���-�ζ�ǰ�Ķ�������18.10ml-0ml=18.10ml��
���ĵĵ�����ʵ���Ϊ��0.1000mol/L��V��10-3L����Na2SO3+I2+H2O=Na2SO4+2HI��֪��n��Na2SO3��=0.1000��V��10-3mol��
��250mL��Һ��Na2SO3�����ʵ���Ϊ��0.1000��V��10-3mol��
=V��10-3mol��������������Ϊ
��100%=
%��
�ʴ�Ϊ��18.10��
%��
��5����Na2SO3+I2+H2O=Na2SO4+2HI��֪n��Na2SO3��=n��I2��=cV
A���ζ�ǰ���ô���Һ��ϴ��ƿ������ƿ��Na2SO3���ʵ���ƫ�ζ�ʱ���ĵĵ���Һ�����ƫ����õ���n��Na2SO3��ƫ������������ƫ��A��ȷ��
B���ζ�ǰ��δ�ô���Һ��ϴ�ζ��ܣ���Na2SO3Ũ�ȱ�С���ζ�ʱ���ĵĵ���Һ�����ƫС������õ���n��Na2SO3��ƫС������������ƫС����B����
C���ζ�����ʱ�����Ӷ���������ƫС�������ĵĵ���Һ�����ƫС������õ���n��Na2SO3��ƫС������������ƫС����C����
D�����Ʊ�����Һ����ʱ�����ӿ̶��ߣ�����Һ���ƫ���Ũ��ƫС���ζ�ʱ���ĵĵ���Һ�����ƫ����õ���n��Na2SO3��ƫ������������ƫ��A��ȷ��
�ʴ�Ϊ��AD��
��6�����ζ�����ʱ�������Na2SO3����Һ�лᱻ������������ʹʵ����ƫС�������ķ�ӦΪ��2Na2SO3+O2=2Na2SO4��
�ʴ�Ϊ��2Na2SO3+O2=2Na2SO4��
��7��ȷ��ȡ��Ʒm1g��������ˮ�ܽ⣬��������μ��������ữ���Ȼ�����Һ��������ȫ���������ᱵ�����ˡ�ϴ�ӡ���ɲ��Ƶó���Ϊm2g��������ԭ���غ�����������������������Na2SO3������������������
�ʴ�Ϊ��ȷ��ȡ��Ʒm1g��������ˮ�ܽ⣬��������μ��������ữ���Ȼ�����Һ��������ȫ�����ˡ�ϴ�ӡ���ɲ��Ƶó���Ϊm2g��
���ñ�Һ��Һ��ϴ�ζ���2��3�Σ�
��ȡ������Һע����ʽ�ζ�����0�̶����ϣ�
�۰�ʢ�б���Һ����ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ��
�ݵ���Һ����0��0�̶����£����¶�����
��ȷ��ȡ25.00mL�����Na2SO3��Һע��ྻ����ƿ��������2��3��ָʾ����
�ް���ƿ���ڵζ��ܵ��·����ñ�����Һ�ζ����յ㣬���µζ���Һ��Ŀ̶��ظ����ϲ���2��3�Σ�����ȷ����˳���ǣ��ڢܢۢݢ٢ޣ�
�ʴ�Ϊ���ڢܢۢݢ٢ޣ�
��2��Na2SO3����Һ��ˮ���Լ��ԣ�������Һ�ü�ʽ�ζ�����ȡ���ʴ�Ϊ���
��3����0.1000mol/L��ı���Һ�ζ�Na2SO3��Һʱ���ⵥ���������ۻ����ɫ���õ�����ָʾ�����ζ�ʱ�ⵥ����Na2SO3��Ӧ����Na2SO3��ȫ��Ӧ���ټ�һ�ε���Һ����ƿ�е���Һ������ɫ����30s�ڲ���ɫ��
�ʴ�Ϊ�����ۣ����������һ�ε�Һʱ����ƿ�е���Һ������ɫ����30s�ڲ���ɫ��
��4�����ı���Һ�����Ϊ���ζ��յ�Ķ���-�ζ�ǰ�Ķ�������18.10ml-0ml=18.10ml��
���ĵĵ�����ʵ���Ϊ��0.1000mol/L��V��10-3L����Na2SO3+I2+H2O=Na2SO4+2HI��֪��n��Na2SO3��=0.1000��V��10-3mol��
��250mL��Һ��Na2SO3�����ʵ���Ϊ��0.1000��V��10-3mol��
| 250ml |
| 25ml |
| V��10-3mol��126g/mol |
| mg |
| 12.6V |
| m |
�ʴ�Ϊ��18.10��
| 12.6V |
| m |
��5����Na2SO3+I2+H2O=Na2SO4+2HI��֪n��Na2SO3��=n��I2��=cV
A���ζ�ǰ���ô���Һ��ϴ��ƿ������ƿ��Na2SO3���ʵ���ƫ�ζ�ʱ���ĵĵ���Һ�����ƫ����õ���n��Na2SO3��ƫ������������ƫ��A��ȷ��
B���ζ�ǰ��δ�ô���Һ��ϴ�ζ��ܣ���Na2SO3Ũ�ȱ�С���ζ�ʱ���ĵĵ���Һ�����ƫС������õ���n��Na2SO3��ƫС������������ƫС����B����
C���ζ�����ʱ�����Ӷ���������ƫС�������ĵĵ���Һ�����ƫС������õ���n��Na2SO3��ƫС������������ƫС����C����
D�����Ʊ�����Һ����ʱ�����ӿ̶��ߣ�����Һ���ƫ���Ũ��ƫС���ζ�ʱ���ĵĵ���Һ�����ƫ����õ���n��Na2SO3��ƫ������������ƫ��A��ȷ��
�ʴ�Ϊ��AD��
��6�����ζ�����ʱ�������Na2SO3����Һ�лᱻ������������ʹʵ����ƫС�������ķ�ӦΪ��2Na2SO3+O2=2Na2SO4��
�ʴ�Ϊ��2Na2SO3+O2=2Na2SO4��
��7��ȷ��ȡ��Ʒm1g��������ˮ�ܽ⣬��������μ��������ữ���Ȼ�����Һ��������ȫ���������ᱵ�����ˡ�ϴ�ӡ���ɲ��Ƶó���Ϊm2g��������ԭ���غ�����������������������Na2SO3������������������
�ʴ�Ϊ��ȷ��ȡ��Ʒm1g��������ˮ�ܽ⣬��������μ��������ữ���Ȼ�����Һ��������ȫ�����ˡ�ϴ�ӡ���ɲ��Ƶó���Ϊm2g��
���������⿼�������ʺ����IJⶨ���ζ�ʵ��IJ�����յ��жϷ�������������Ӧ�õȣ���Ŀ�ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ������ڿ���ѧ����ʵ��̽�����������ݴ���������

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
������ˮΪ֮��������ˮ������ˮ����ɱ��Ĺ��̵�������ȷ���ǣ�������
| A����H��0����S��0 |
| B����H��0����S��0 |
| C����H��0����S��0 |
| D����H��0����S��0 |
��a��Fe2O3 ��Al2O3 ��Ʒ�ܽ��ڹ�����200mL c��H+��=0.1mol?L-1 ��������Һ�У�Ȼ�������м���NaOH��ҺʹFe3+��Al3+ �պ���ȫ��������ȥNaOH��Һ100mL����NaOH ��Һ��Ũ��Ϊ��������
| A��0.1 mol?L-1 |
| B��0.2 mol?L-1 |
| C��0.4 mol?L-1 |
| D��0.8 mol?L-1 |
���й������ʷ����˵����ȷ���ǣ�������
| A�����ᡢ��ˮ�����ᱵ������������ʡ� |
| B��ͭ�Ͻ����Ͻ𡢸��������ڽ������� |
| C�������͡�ֲ���͡������;������������ |
| D��ϡ������ϡ���ᡢ�轺�����ڽ��� |
�����и�����������Һ�У�һ���ܴ���������������ǣ�������
| A��ij��Һ�У�Ca2+��H+��Cl-��HSO3- |
| B����ʹPH��ֽ�ʺ�ɫ����Һ��Na+��NH4+��Fe2+��NO3- |
| C��Na2CO3��Һ��K+��Fe3+��SO42-��NO3- |
| D��Kw/c��H+��=0.1mol/L����Һ��Na+��K+��S2-��NO3- |
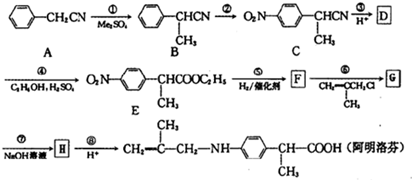
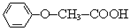 ������������Ӧ�IJ��
������������Ӧ�IJ��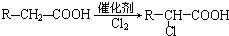 ��R-ONa
��R-ONa
 �˴Ź���������
�˴Ź���������