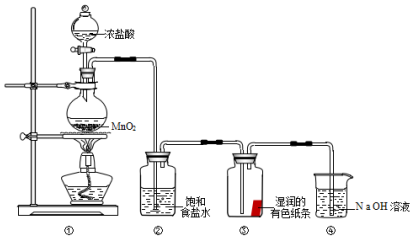
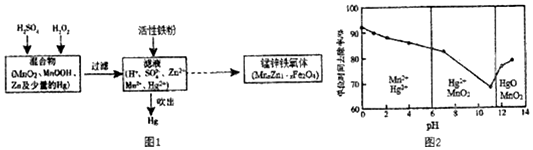
��Ŀ����
19��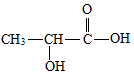 �����������Ϲ��ϵ������л���֮һ����Ӧ�÷dz��㷺��������ʳƷ��ζ����ҽҩ����������ҵpH���ڼ��ȣ�����Ľṹ��ʽ��ͼ��ʾ��
�����������Ϲ��ϵ������л���֮һ����Ӧ�÷dz��㷺��������ʳƷ��ζ����ҽҩ����������ҵpH���ڼ��ȣ�����Ľṹ��ʽ��ͼ��ʾ����ش�
��1����������к��������ŵ��������ǻ����Ȼ���
��2������ķ���ʽ��C3H6O3��
��3������ۺϵõ��ľ�������Գ��˿�ߣ������������õ�ҽ����������ߣ�������һ�������¾ۺ����ɾ�����Ļ�ѧ����ʽ��
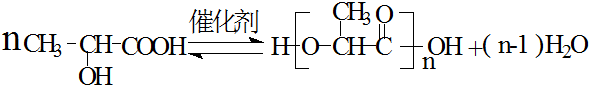 ��
����4�������ж���ͬ���칹�壬��д������һ����������������ͬ���칹��Ľṹ��ʽ��HCOOCH��OH��CH3��
�ٺ��������ܷ���������Ӧ
����������Ʒ�Ӧ����������
���� ��1�����ݹ����ŵĽṹ�ص�����ƽ���л���Ľṹ��ʽ���ش�
��2�������л���Ľṹ��ʽ��Ϸ���ʽ����д���ش�
��3������ͨ��������Ӧ���е����۷�Ӧ���ɾ����
��4���ٺ��������ܷ���������Ӧ��˵���Ǽ���ij���Ľṹ��
����������Ʒ�Ӧ����������˵�������ǻ���
��� �⣺��1����������к��������ŵ��������ǻ����Ȼ����ʴ�Ϊ���ǻ����Ȼ���
��2�������л�������Ľṹ��ʽ���õ��������ʽΪ��C3H6O3���ʴ�Ϊ��C3H6O3��
��3��������һ�������·�����Ӧ�õ�������Ļ�ѧ����ʽ�ǣ�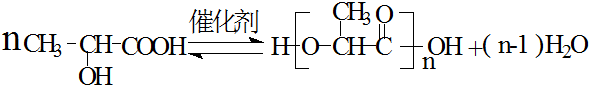 ��
��
�ʴ�Ϊ��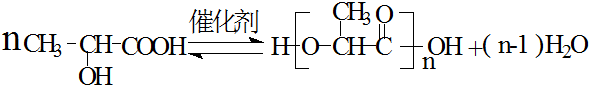 ��
��
��4���ٺ��������ܷ���������Ӧ��˵���Ǽ���ij��HCOO-�Ľṹ��
����������Ʒ�Ӧ����������˵�������ǻ�-OH������ʽΪC3H6O3����ṹ��ʽ������HCOOCH��OH��CH3���ʴ�Ϊ��HCOOCH��OH��CH3��
���� ���⿼��ѧ���л���ṹ��ʽ�ͷ���ʽ����д��ͬ���칹�����д�����ʵ����ʺ����ŵĹ�ϵ��֪ʶ���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
9������Ǧ���ص�˵����ȷ���ǣ�������
| A�� | �ڷŵ�ʱ�����������ķ�Ӧ��Pb+SO42-�TPbSO4+2e- | |
| B�� | �ڷŵ�ʱ���õ�صĸ���������Ǧ�� | |
| C�� | �ڳ��ʱ������������Ũ�Ȳ��ϱ�С | |
| D�� | �ڳ��ʱ�����������ķ�Ӧ��PbSO4+2e-�TPb+SO42- |
10������������ŷ���ȫ���ů����Ҫԭ���������������У������л�����ǣ�������
| A�� | O3 | B�� | CO2 | C�� | N2O | D�� | CH4 |
7������˵���У���ȷ���ǣ�������
| A�� | 1 mol CO2��������44g | |
| B�� | 17 g NH3�������22.4 L | |
| C�� | 100 mL 1 mol/L NaOH��Һ�к���1 mol OH- | |
| D�� | ��״���£�11.2 L O2�к���6.02��1023�������� |
4�������£�������Һ��pH����7���ǣ�������
| A�� | NH4Cl | B�� | Al2��SO4��3 | C�� | NaHCO3 | D�� | Na2SO4 |
9��X��Y��Z��W�Ƕ���������Ԫ�أ�Xԭ�����������������ڲ��������3����Y��ԭ����������������������6����Z��һ�ֵ�������Ȼ������Ӳ�����ʣ���Ȼ���У�W�ĵ��ʶ�����ڻ�ɽ�ڸ�������Ϊ����ɫ���壮����������ȷ���ǣ�������
| A�� | ԭ�Ӱ뾶�Ĵ�С��Z��X��Y | |
| B�� | Z���⻯��ķе�һ����X �ĵ� | |
| C�� | W������������Ӧˮ�����������ͬ��������ǿ | |
| D�� | Y�ĵ�����Z��X�γɵĻ����ﷴӦ��ұ������ |