��Ŀ����
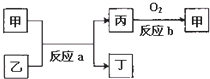
��֪ͨ��״���¼ס��ҡ���������Ϊ���嵥�ʣ�A��B��C��D��E��F��G��H��Ϊ���������A��B��E��G��Ϊ���壬CΪ����Һ�壮��Ӧ�٢ڢ۶�����Ҫ�Ļ�����Ӧ����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ���йص�ת����ϵ����ͼ��ʾ����Ӧ����������ȥ����

��ش��������⣺
��1����Ӧ�ܵĻ�ѧ����ʽΪ ��
��2��B��E��һ�������¿ɷ�����Ӧ������һ������ʵ������ķ�Ӧ��������E�Ի�������Ⱦ���÷�Ӧ���������뻹ԭ��������ʵ���֮��Ϊ ��
��3��0.1mol?L-1A��Һ��0.1mol?L-1B��Һ�������ϣ���Һ�� �ԣ�ԭ���� �������ӷ���ʽ˵������
��4�������ʵ�����D�����е������ӣ� ��
��5��pH��ͬ��A��D��H������Һ����ˮ�������c��OH-���Ĵ�С��ϵ�ǣ���A��D��H��ʾ���� ��
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1A����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣���ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1H��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣����������H��Һ������� mL��

��ش��������⣺
��1����Ӧ�ܵĻ�ѧ����ʽΪ
��2��B��E��һ�������¿ɷ�����Ӧ������һ������ʵ������ķ�Ӧ��������E�Ի�������Ⱦ���÷�Ӧ���������뻹ԭ��������ʵ���֮��Ϊ
��3��0.1mol?L-1A��Һ��0.1mol?L-1B��Һ�������ϣ���Һ��
��4�������ʵ�����D�����е������ӣ�
��5��pH��ͬ��A��D��H������Һ����ˮ�������c��OH-���Ĵ�С��ϵ�ǣ���A��D��H��ʾ����
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1A����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣���ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1H��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣����������H��Һ�������
���㣺������ƶ�
ר�⣺�ƶ���
������CΪ����Һ�壬ӦΪH2O���������ӦΪH2��O2�е����ʣ���B�ܺͱ���Ӧ����C��˵��BӦΪ�⻯���ΪH2����ΪO2���ɷ�Ӧ�٢ڢۿ�֪������������Ԫ�ش��ڶ��ֻ��ϼۣ���E��G��Ϊ���������G����ˮ��Ӧ����E��˵������������ԭ��Ӧ�����֪��ΪN2��BΪNH3��EΪNO��GΪNO2��HΪHNO3����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ��������NH3��ӦΪNH4Cl��Ca��OH��2�ķ�Ӧ����DΪNH4Cl��FΪCa��OH��2��IΪCaCl2�����ΪCl��AΪHCl����϶�Ӧ���ʵ������Լ���ĿҪ������⣮
���
�⣺CΪ����Һ�壬ӦΪH2O���������ӦΪH2��O2�е����ʣ���B�ܺͱ���Ӧ����C��˵��BӦΪ�⻯���ΪH2����ΪO2���ɷ�Ӧ�٢ڢۿ�֪������������Ԫ�ش��ڶ��ֻ��ϼۣ���E��G��Ϊ���������G����ˮ��Ӧ����E��˵������������ԭ��Ӧ�����֪��ΪN2��BΪNH3��EΪNO��GΪNO2��HΪHNO3����Ӧ������Ҫ��ʵ������ȡ����ķ�Ӧ��������NH3��ӦΪNH4Cl��Ca��OH��2�ķ�Ӧ����DΪNH4Cl��FΪCa��OH��2��IΪCaCl2�����ΪCl��AΪHCl��
��1����Ӧ��Ϊʵ�����Ʊ������ķ�Ӧ������ʽΪ2NH4Cl+Ca��OH��2
CaCl2+2NH3��+2H2O���ʴ�Ϊ��2NH4Cl+Ca��OH��2
CaCl2+2NH3��+2H2O��
��2��BΪNH3��EΪNO���������Ϣ��֪��Ӧ�ķ���ʽΪ4NH3+6NO=5N2+6H2O���ɷ���ʽ��֪���������뻹ԭ��������ʵ���֮��Ϊ2��3���ʴ�Ϊ��2��3��
��3��0.1mol?L-1A��Һ��0.1mol?L-1B��Һ�������ϣ���Ӧ����NH4Cl��Ϊǿ�������Σ�ˮ������ԣ�����ʽΪNH4++H2O?NH3?H2O+H+��
�ʴ�Ϊ���NH4++H2O?NH3?H2O+H+��
��4��DΪNH4Cl��NH4+�ļ������������巨����ȡһ�Թܼ���������D���壬Ȼ���������������Һ�����ȣ����Թܿڷ���ʪ��ĺ�ɫʯ����ֽ������ֽ������˵��D�к���NH4+���ʴ�Ϊ��ȡһ�Թܼ���������D���壬Ȼ���������������Һ�����ȣ����Թܿڷ���ʪ��ĺ�ɫʯ����ֽ������ֽ������˵��D�к���NH4+��
��5��pH��ͬ��HCl��NH4Cl��HNO3������HCl��HNO3����ˮ�ĵ��룬NH4Cl��ˮ����ٽ�ˮ�ĵ��룬��pH��ͬʱ��ˮ�������c��OH-���Ĵ�С��ϵ��D��A=H��
�ʴ�Ϊ��D��A=H��
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1HCl����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣�˵����Һ����ΪFeCl2����ClԪ���غ��֪n��FeCl2��=0.05mol��ͬʱת�Ƶ���Ϊ2��
=0.03mol��
��ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1HNO3��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣�������0.05molFe��NO3��2��ͬʱ����NO
=0.01mol����NԪ���غ��֪��Ҫ��HNO3���ʵ���Ϊ0.05mol��2+0.01mol=0.11mol������������Ϊ
=0.11L=110mL��
�ʴ�Ϊ��110��
��1����Ӧ��Ϊʵ�����Ʊ������ķ�Ӧ������ʽΪ2NH4Cl+Ca��OH��2
| ||
| ||
��2��BΪNH3��EΪNO���������Ϣ��֪��Ӧ�ķ���ʽΪ4NH3+6NO=5N2+6H2O���ɷ���ʽ��֪���������뻹ԭ��������ʵ���֮��Ϊ2��3���ʴ�Ϊ��2��3��
��3��0.1mol?L-1A��Һ��0.1mol?L-1B��Һ�������ϣ���Ӧ����NH4Cl��Ϊǿ�������Σ�ˮ������ԣ�����ʽΪNH4++H2O?NH3?H2O+H+��
�ʴ�Ϊ���NH4++H2O?NH3?H2O+H+��
��4��DΪNH4Cl��NH4+�ļ������������巨����ȡһ�Թܼ���������D���壬Ȼ���������������Һ�����ȣ����Թܿڷ���ʪ��ĺ�ɫʯ����ֽ������ֽ������˵��D�к���NH4+���ʴ�Ϊ��ȡһ�Թܼ���������D���壬Ȼ���������������Һ�����ȣ����Թܿڷ���ʪ��ĺ�ɫʯ����ֽ������ֽ������˵��D�к���NH4+��
��5��pH��ͬ��HCl��NH4Cl��HNO3������HCl��HNO3����ˮ�ĵ��룬NH4Cl��ˮ����ٽ�ˮ�ĵ��룬��pH��ͬʱ��ˮ�������c��OH-���Ĵ�С��ϵ��D��A=H��
�ʴ�Ϊ��D��A=H��
��6����һ������Fe��FeO��Fe3O4�Ļ�����У�����1mol?L-1HCl����Һ100mL��ǡ��ʹ�����ȫ���ܽ⣬�ҷų�336mL����״���£������壬��������Һ�м���KSCN��Һ����Һ��ɫ���֣�˵����Һ����ΪFeCl2����ClԪ���غ��֪n��FeCl2��=0.05mol��ͬʱת�Ƶ���Ϊ2��
| 0.336L |
| 22.4L/mol |
��ȡͬ������Fe��FeO��Fe3O4��������1mol?L-1HNO3��Һ��Ҳǡ��ʹ�����ȫ���ܽ⣬����������Һ�м���KSCN��Һ����ҺҲ��ɫ���֣�������0.05molFe��NO3��2��ͬʱ����NO
| 0.03mol |
| 5-2 |
| 0.11mol |
| 1mol/L |
�ʴ�Ϊ��110��
���������⿼��������ƶϣ�������ѧ���ķ��������ͼ��������Ŀ��飬Ϊ�߿��������ͣ�ע�������Ŀ�ƶϵ�ͻ�ƿڣ���C�ͷ�Ӧ��ת����ϵ�������Ƶķ��������ƶϣ���Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ�ǣ�������
| A���õ⻯�ص�����Һ��������������NO2-�Ĵ��ڣ�NO2-+I-+2H+=NO��+I2+H2O |
| B����������ˮ��Cl2+H2O=C1O-+C1-+2H+ |
| C����������������ʴ�ĸ�����Ӧ��Fe-3e-=Fe3+ |
| D����״����11.2L CO2ͨ��500mL1mol/L��NaOH��Һ��CO2+OH-=HCO3- |
 �Т١�����ֶ�����Ԫ�أ���ԭ�����������������Тڡ��ߡ�������Ԫ�������ڱ��е����λ�����£���Ԫ�آ����������λ��Ϊ������������м䣮
�Т١�����ֶ�����Ԫ�أ���ԭ�����������������Тڡ��ߡ�������Ԫ�������ڱ��е����λ�����£���Ԫ�آ����������λ��Ϊ������������м䣮
 ��������PTCԪ������Ҫ�ɷ�Ϊ���ѿ��侧��ṹ��ͼ��ʾ���ýṹ�Ǿ��д����Ե���С�ظ���λ���þ��徭X���߷����������ظ���λΪ�����壬�߳�Ϊ403.1pm������λ��ΪTi4+��ռ������λ��ΪBa2+��ռ����������λ��ΪO2-��ռ��
��������PTCԪ������Ҫ�ɷ�Ϊ���ѿ��侧��ṹ��ͼ��ʾ���ýṹ�Ǿ��д����Ե���С�ظ���λ���þ��徭X���߷����������ظ���λΪ�����壬�߳�Ϊ403.1pm������λ��ΪTi4+��ռ������λ��ΪBa2+��ռ����������λ��ΪO2-��ռ��
