��Ŀ����
2��������һ�ּ��߷�չDZ���������Դ����̫����Ϊ��Դ���Ȼ�ѧ���ѭ���ֽ�ˮ��һ�ָ�Ч������Ⱦ�����ⷽ�����䷴Ӧ������ͼ1��ʾ��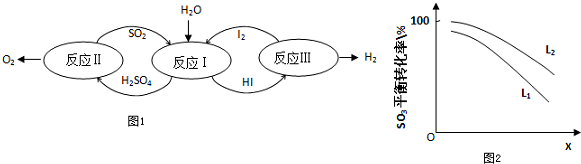
��1����Ӧ��Ļ�ѧ����ʽ��SO2+2H2O+I2=H2SO4+2HI��
��2����Ӧ��õ��IJ�����I2���з��룮�ò������Һ�ڹ���I2�Ĵ����»�ֳ�����--����Ũ��I2��H2SO4���Ũ�ȵ�I2��HI�㣮
�ٸ���������ʵ������˵����ȷ����ac��ѡ����ţ���
a��������Һ���ܶȴ��ڲ���
b����I2ǰ��H2SO4��Һ��HI��Һ������
c��I2��HI��Һ�б���H2SO4��Һ������
�ڱ��������Һ�ķ����ǣ��������ữ���Ȼ�����Һ�����ϲ���Һ�е�SO42-�����ش��á������������ġ������ӡ����ɣ�
�۾���⣬H2SO4����c��H+����c��SO42-��=2.06��1�����ֵ����2��ԭ���ǣ�������к�������I2��I2+H2O=HI+HIO����HI����������ӣ���3����Ӧ��2H2SO4��l���T2SO2��g��+O2��g��+2H2O��g����H=+550kJ/mol
����������Ӧ��ɣ�i��H2SO4��l���TSO3��g��+H2O��g����H=+177kJ/mol
ii��SO3��g���ֽ⣮
L��L1��L2����X�ɷֱ����ѹǿ���¶ȣ�ͼ2��ʾLһ��ʱ��ii��SO3��g����ƽ��ת������X�ı仯��ϵ��
��X��������������ѹǿ��
���ж�L1��L2�Ĵ�С��ϵ�����������ɣ�L1��L2���ֽⷴӦΪ���ȷ�Ӧ���¶ȸߣ�ת���ʴ�
���� ��1����ͼ��֪����ӦIΪ����������ⷢ��������ԭ��Ӧ���������HI��
��2���ٷֳ����㣬���ܽ��ԡ��ܶ��йأ�
�ڸ����������ữ���Ȼ�����Һ�����ϲ���Һ�е�SO42-���������Һ��
��H2SO4��c��H+����c��SO42-��=2.06��1����HI����������ӣ�
��3������ͼ��֪��XԽ��ת����Խ�ͣ�
�ڷֽⷴӦΪ���ȷ�Ӧ���¶ȸߣ�ת���ʴ�
��� �⣺��1����ͼ��֪����ӦIΪ����������ⷢ��������ԭ��Ӧ���������HI���÷�ӦΪSO2+2H2O+I2=H2SO4+2HI���ʴ�Ϊ��SO2+2H2O+I2=H2SO4+2HI��
��2����a��������Һ���ܶȴ��ڲ�ų������²㣬��a��ȷ��
b����I2ǰ��H2SO4��Һ��HI��Һ���ܣ���ֲ��أ���b����
c��I2��HI��Һ�б���H2SO4��Һ�����ܣ�����ڲ�ͬ�ܼ����ܽ��Բ�ͬ��������ȡ����ֲ��йأ���c��ȷ��
�ʴ�Ϊ��ac��
����Ϊ��Һ�ֳɺ���Ũ��I2��H2SO4���Ũ�ȵ�I2��HI�㣬����������Һ�ķ������������ữ���Ȼ�����Һ�����ϲ���Һ�е�SO42-���ʴ�Ϊ���������ữ���Ȼ�����Һ�����ϲ���Һ�е�SO42-��
��H2SO4����c��H+����c��SO42-��=2.06��1�����ֵ����2��ԭ����������к�������I2��I2+H2O=HI+HIO����HI����������ӣ��ʴ�Ϊ��������к�������I2��I2+H2O=HI+HIO����HI����������ӣ�
��3������ͼ��֪��XԽ��ת����Խ�ͣ������¶�ת����������X��ʾѹǿ���ʴ�Ϊ��ѹǿ��
����SO3��g��=SO2��g��+O2��g����H��0���¶ȸߣ�ת���ʴ�ͼ�е�ѹǿʱL2��Ӧ��ת���ʴ���L1��L2���ʴ�Ϊ��L1��L2���ֽⷴӦΪ���ȷ�Ӧ���¶ȸߣ�ת���ʴ�
���� ���⿼����������ᴿ����ѧƽ��ȣ�Ϊ��Ƶ���㣬���շ����ķ�Ӧ��ƽ��Ӱ������Ϊ���Ĺؼ������ط�����Ӧ���������ۺϿ��飬��Ŀ�Ѷ��еȣ�

| A�� | NaHSO4��Һ | B�� | KOH��Һ | C�� | Na2SO4 | D�� | NH4Cl��Һ |
| A�� | ������ѹǿ����ʱ��仯 | B�� | ��ѧ��Ӧ���ʹ�ϵ�ǣ�2������X��=������Y�� | ||
| C�� | �����������ܶȲ���ʱ��仯 | D�� | ������X��Y��Z��Ũ��֮��Ϊl��2��2 |
| A�� | �ԣ�A��=0.5mol/��L•min�� | B�� | �ԣ�B��=1.2mol/��L•s�� | C�� | �ԣ�C��=0.1mol/��L•s�� | D�� | �ԣ�D��=0.4mol/��L•min�� |
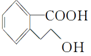 �������й�M��˵����ȷ���ǣ�������
�������й�M��˵����ȷ���ǣ�������| A�� | �÷����д���һ������̼ԭ�� | |
| B�� | M�ķ���ʽΪC9H10O3 | |
| C�� | 1 mol M��������Na��Ӧ����0.5 mol H2 | |
| D�� | 1 mol M��ȫȼ������10 mol O2 |
| t�� | 700 | 800 | 830 | 1000 | 1200 |
| K | 2.6 | 1.7 | 1.0 | 0.9 | 0.6 |
��1���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK=$\frac{{c}^{2}��S{O}_{3}��}{c��{O}_{2}��•{c}^{2}��S{O}_{2}��}$��
��2��ij�¶��£�ƽ��Ũ�ȷ�����ʽ��c2��SO3��=c��O2��•c2��SO2�������жϴ�ʱ���¶�Ϊ830�森
��3������һ��������ܱ������г���0.30mol O2��g����0.60mol SO2��g������Ӧ��һ�������´ﵽƽ��ʱ��SO3�����ʵ���������SO3�����ʵ����뷴Ӧ��ϵ���ܵ����ʵ���֮�ȣ�Ϊ$\frac{4}{7}$����������O2��ƽ��ת����Ϊ$\frac{2}{3}$���������·�Ӧ2SO3��g��?O2��g��+2SO2��g����ƽ�ⳣ��K=40��
��4����O2��SO2��������Բ�ͬ�������ʱ�������ƽ����Է��������ֱ�Ϊ57.6��44.8��ȡ������ͬ�����Ļ�������5L����һ�������³�ַ�Ӧ�����������Ϊ4L���ֱ����O2��������������е��������20%��60%��
��1����֪��C��s��+O2��g���TCO2��g����H=-393.5KJ•mol-1
H2��g��+$\frac{1}{2}$O2��g���TH2O��g����H=-241.8KJ•mol-1
CO��s��+$\frac{1}{2}$O2��g���TCO2��g����H=-283.0KJ•mol-1
��ú������Ҫ��ӦC��s��+H2O ��g���TCO��g��+H2��g���ġ�H=+131.3 kJ/mol
��2����֪CO��g��+O2��g���TCO2��g��+O��g��������Ӧ����Ϊv��=k��•c��CO��•c��O2�����淴Ӧ����Ϊv��=k��•c��CO2��•c��O����kΪ���ʳ�����2500Kʱ��k��=1.21��105 L/mol��k��=3.02��105L/mol������¶��µķ�Ӧƽ�ⳣ��K=0.40
��3���״��Ƽ��ѵ��йط�ӦΪ��2CH3OH ��g���TCH3OCH 3��g��+H2O ��g����һ���¶��£��������ݻ���Ϊ1.0L�ĺ����ܱ������з����÷�Ӧ��
| ������� | �¶ȣ��棩 | ��ʼ���ʵ�����mol�� | ƽ�����ʵ�����mol�� | |
| CH3OH��g�� | CH3OCH3��g�� | H2O��g�� | ||
| �� | 387 | 0.20 | x | |
| �� | 387 | 0.40 | y | |
| �� | 207 | 0.20 | 0.090 | 0.090 |
����֪387��ʱ�÷�Ӧ�Ļ�ѧƽ�ⳣ��K=4�����¶��£�����ʼʱ���������г���0.10molCH3OH ��g����0.15mol CH3OCH3��g����0.10mol H2O��g������Ӧ����������������桱����Ӧ������У�
���������з�Ӧ�ﵽƽ�����Ҫ��һ������ѵIJ��ʣ����Բ�ȡ�Ĵ�ʩΪB��������ţ�
A�������¶�
B�������¶�
C�������������䣬����CH3OH �����ʵ���
D�����������������䣬ͨ������
��4���Լ״�Ϊ��Ҫԭ�ϣ��绯ѧ�ϳ�̼��������Ĺ���ԭ������ͼ��ʾ��
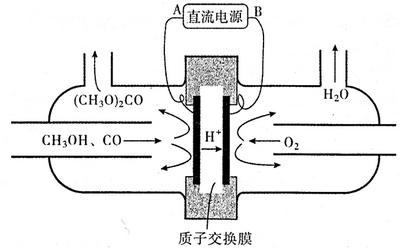
���Դ�ĸ���ΪB���A����B������д�������ĵ缫��Ӧʽ��2CH3OH+CO-2e-=��CH3O��2CO+2H+
��5������ʯȼ�ϵ�ȼ�ղ���CO2ͨ��NaOH��Һ�п��Ƶ�̼�����ƣ�ȡ0.2mol/L̼������Һ��0.2mol/L�Ȼ�����Һ�������ϲ������ǣ�����һ�����ݣ�ͨ������˵���������ǵ�ԭ��[��֪��25��ʱ��0.1mol/L̼��������Һ��̼������ӵ�Ũ��Ϊ0.0011mol/L��Ksp��BaCO3��=5.1��10-9]��
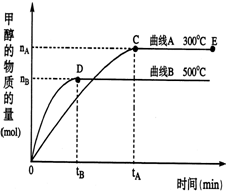 һ�������£������Ϊ2L���ܱ������У�һ����̼�������ڴ������÷�Ӧ���ɼ״���CO��g��+2H2��g��?CH3OH��g������������������и��⣺
һ�������£������Ϊ2L���ܱ������У�һ����̼�������ڴ������÷�Ӧ���ɼ״���CO��g��+2H2��g��?CH3OH��g������������������и��⣺