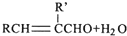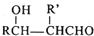��Ŀ����
�л���Aֻ����C��H��O����Ԫ�أ��������л��ϳɵ��м��壮16.8g���л�����ȫȼ�����ɵ�������ͨ�������ij���ʯ��ˮ�У��õ�100g����������ȼ�պ������ͨ��������Na2O2���壬������������29.6g������ͼ��������Է�������Ϊ84���������������A�����к���O-H����λ�ڷ��Ӷ˵�C��C�����˴Ź����������������壬�����֮��Ϊ6��1��1��
��1��A�Ľṹ��ʽ�� ���ǻ���̼̼������������ͬһ��̼�ϣ���
��2��д��A��������ˮ������Ӧ�Ļ�ѧ����ʽ��
��3���л���B��A��ͬ���칹�壬1mol B������1mol Br2�ӳɣ����л���������̼ԭ����ͬһ��ƽ�棬û��˳���칹������B�Ľṹ��ʽ�� ��
��1��A�Ľṹ��ʽ��
��2��д��A��������ˮ������Ӧ�Ļ�ѧ����ʽ��
��3���л���B��A��ͬ���칹�壬1mol B������1mol Br2�ӳɣ����л���������̼ԭ����ͬһ��ƽ�棬û��˳���칹������B�Ľṹ��ʽ��
���㣺�й��л������ʽȷ���ļ���
ר�⣺
��������1����2���л���Aֻ����C��H��O����Ԫ�أ�����ͼ��������Է�������Ϊ84��16.8g�л���A�����ʵ���=
=0.2mol����ȫ��Ϊ����������̼���Եõ�100g̼��Ƴ�����̼��Ƶ����ʵ���Ϊ
=1mol�������A������N��C��=
=5����ȼ�պ������ͨ��������Na2O2���壬������������29.6g���ɻ�ѧʽ�仯��֪����������ΪCO����Ԫ��������CO������Ϊ1mol��28g/mol=28g������Ԫ��������Ϊ29.6g-28g=1.6g�������ʵ���Ϊ
=1.6mol����A������N��H��=
=8����N��O��=
=1����A�ķ���ʽΪC5H8O���������������A�����к���O-H����λ�ڷ��Ӷ˵�C��C�����˴Ź����������������壬�����֮��Ϊ6��1��1���ʷ����к���2��CH3��1��-OH������ͬһCԭ���ϣ���A�Ľṹ��ʽΪ ��
��
��3���л���B��A��ͬ���칹�壬1molB����1molBr2�ӳɣ��ʷ����к���1��C=C˫�������л�������̼ԭ����ͬһ��ƽ�棬�����̼ԭ������C=C˫����û��˳���칹��������˫����������һ��Cԭ������������ͬ�Ļ��ż������A�Ľṹ��ʽ��д��
| 16.8g |
| 84g/mol |
| 100g |
| 100g/mol |
| 1mol��1 |
| 0.2mol |
| 1.6g |
| 1g/mol |
| 1.6mol |
| 0.2mol |
| 84-12��5-8 |
| 16 |
 ��
����3���л���B��A��ͬ���칹�壬1molB����1molBr2�ӳɣ��ʷ����к���1��C=C˫�������л�������̼ԭ����ͬһ��ƽ�棬�����̼ԭ������C=C˫����û��˳���칹��������˫����������һ��Cԭ������������ͬ�Ļ��ż������A�Ľṹ��ʽ��д��
���
�⣺��1���л���Aֻ����C��H��O����Ԫ�أ�����ͼ��������Է�������Ϊ84��16.8g�л���A�����ʵ���=
=0.2mol����ȫ��Ϊ����������̼���Եõ�100g̼��Ƴ�����̼��Ƶ����ʵ���Ϊ
=1mol�������A������N��C��=
=5����ȼ�պ������ͨ��������Na2O2���壬������������29.6g���ɻ�ѧʽ�仯��֪����������ΪCO����Ԫ��������CO������Ϊ1mol��28g/mol=28g������Ԫ��������Ϊ29.6g-28g=1.6g�������ʵ���Ϊ
=1.6mol����A������N��H��=
=8����N��O��=
=1����A�ķ���ʽΪC5H8O���������������A�����к���O-H����λ�ڷ��Ӷ˵�C��C�����˴Ź����������������壬�����֮��Ϊ6��1��1���ʷ����к���2��CH3��1��-OH������ͬһCԭ���ϣ���A�Ľṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2��A��������ˮ������Ӧ�Ļ�ѧ����ʽΪ�� +2Br2��
+2Br2�� ��
��
�ʴ�Ϊ�� +2Br2��
+2Br2�� ��
��
��3���л���B��A��ͬ���칹�壬1molB����1molBr2�ӳɣ��ʷ����к���1��C=C˫�������л�������̼ԭ����ͬһ��ƽ�棬�����̼ԭ������C=C˫����û��˳���칹��������˫����������һ��Cԭ������������ͬ�Ļ��ż������A�Ľṹ��ʽ��֪��B�л�����-CHO����BΪ��CH3��2C=CHCHO���ʴ�Ϊ����CH3��2C=CHCHO��
| 16.8g |
| 84g/mol |
| 100g |
| 100g/mol |
| 1mol��1 |
| 0.2mol |
| 1.6g |
| 1g/mol |
| 1.6mol |
| 0.2mol |
| 84-12��5-8 |
| 16 |
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����2��A��������ˮ������Ӧ�Ļ�ѧ����ʽΪ��
 +2Br2��
+2Br2�� ��
���ʴ�Ϊ��
 +2Br2��
+2Br2�� ��
����3���л���B��A��ͬ���칹�壬1molB����1molBr2�ӳɣ��ʷ����к���1��C=C˫�������л�������̼ԭ����ͬһ��ƽ�棬�����̼ԭ������C=C˫����û��˳���칹��������˫����������һ��Cԭ������������ͬ�Ļ��ż������A�Ľṹ��ʽ��֪��B�л�����-CHO����BΪ��CH3��2C=CHCHO���ʴ�Ϊ����CH3��2C=CHCHO��
���������⿼���л����ƶϡ����������ʡ�ͬ���칹����д�ȣ��Ѷ��еȣ��Ƕ�ѧ���ۺ������Ŀ��飬ȷ������ʽ�ǽ���Ĺؼ���

��ϰ��ϵ�д�
 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д� �����Ծ���Ԫ���Ծ�ϵ�д�
�����Ծ���Ԫ���Ծ�ϵ�д�
�����Ŀ
���й�����Ȼ��ˮ���������ַ��Ӽ��Ե�������ȷ���ǣ�������
| A�����ֶ��Ǽ��Է��� |
| B�����ֶ��ǷǼ��Է��� |
| C��CH4�Ǽ��Է��ӣ�H2O�ǷǼ��Է��� |
| D��H2O�Ǽ��Է��ӣ�CH4�ǷǼ��Է��� |
����˵������ȷ���ǣ�������
| A��1mol�κ����ʶ�����6.02��1023��ԭ�� |
| B�������ӵ���������1mol�������ļ��壬0.012kg12C�к���Լ6.02��1023��12C |
| C��Ħ����һ�������������� |
| D��1molˮ�к���2mol���1mol�� |
����˵����ȷ���ǣ�������
�ٷ����ж����ڹ��ۼ���
����������ȣ�������Ҳ��ȵ������������Dz�ͬ�����ӣ�
��CO2��SiO2�Ļ�ѧ���;������;���ͬ��
�ܻ�ѧ�����з����Ժͱ����ԣ�
�����ӻ�������ܺ��й��ۼ������ۻ����ﲻ�����Ӽ���
��̬�����磬����״̬���ܵ���ľ������Ϊ���Ӿ��壮
�ٷ����ж����ڹ��ۼ���
����������ȣ�������Ҳ��ȵ������������Dz�ͬ�����ӣ�
��CO2��SiO2�Ļ�ѧ���;������;���ͬ��
�ܻ�ѧ�����з����Ժͱ����ԣ�
�����ӻ�������ܺ��й��ۼ������ۻ����ﲻ�����Ӽ���
��̬�����磬����״̬���ܵ���ľ������Ϊ���Ӿ��壮
| A���٢ܢ� | B���ڢۢ� |
| C���٢ڢܢ� | D���ڢݢ� |
������һ��ʱ���������κα�����ϣ���ȫȼ������CO2��H2O����Ϊ��ֵ���ǣ�������
| A��CH2O��C2H4O2 |
| B��C2H4��C3H8 |
C�� ��C2H4 ��C2H4 |
| D��C3H8O��C2H6O |
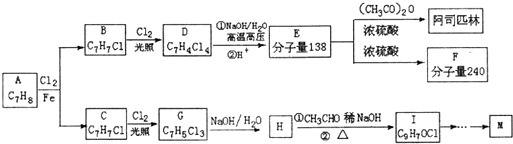
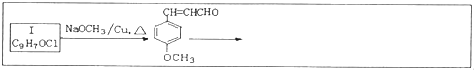
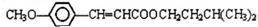 ���ϳ�·�����£�
���ϳ�·�����£�