��Ŀ����
8����1����ϵͳ��������������
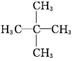 2��2-�������飬д��������������һȡ����Ӧ�ķ���ʽ
2��2-�������飬д��������������һȡ����Ӧ�ķ���ʽ +Cl2$\stackrel{����}{��}$
+Cl2$\stackrel{����}{��}$ +HCl
+HCl��
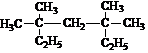 3��3��5��5-�ļ����飻����һ�ȴ�����в�ͬ�е�IJ�����4��
3��3��5��5-�ļ����飻����һ�ȴ�����в�ͬ�е�IJ�����4����2��д�����и��л���Ľṹ��ʽ��
��2��3-����-4-�һ����飺CH3CH��CH3��CH��CH3��CH��C2H5����
��֧��ֻ��һ���һ�����Է���������С��������
 ��
����3���ǻ��ĵ���ʽ
 ��
��
���� ��1������������ʱ��Ҫѡ���̼��Ϊ����������֧������һ�˸�������̼ԭ�ӱ�ţ��ݴ˷��������ڴ��л���ṹ��ֻ��һ��Hԭ�ӣ��ʿ���������һ��ȡ���IJ�����һ�֣�
����������ʱ��Ҫѡ���̼��Ϊ����������֧������һ�˸�������̼ԭ�ӱ�ţ��ݴ˷��������л������м���Hԭ�ӣ�����һ�ȴ����м��ֲ��
��2���ٸ�������������������֪��2��3-����-4-�һ��������������6��̼ԭ�ӣ���2�ź�3��̼ԭ���ϸ���һ��������4��̼ԭ������һ���һ����ݴ�д���ṹ��ʽ��
�������г����һ������������ٺ���5��C���ݴ�д�����л���Ľṹ��ʽ��
��3���ǻ�����ԭ������ԭ���γ�һ�����ۼ�������ԭ��O���⺬��7�����ӣ�
��� �⣺��1������������ʱ��Ҫѡ���̼��Ϊ����������������3��̼ԭ�ӣ�����֧������һ�˸�������̼ԭ�ӱ�ţ�����2��̼ԭ��������������������Ϊ2��2-�������飻���ڴ��л���ṹ��ֻ��һ��Hԭ�ӣ��ʿ���������һ��ȡ���IJ�����һ�֣���ѧ����ʽΪ +Cl2$\stackrel{����}{��}$
+Cl2$\stackrel{����}{��}$ +HCl��
+HCl��
�ʴ�Ϊ��2��2-�������飻 +Cl2$\stackrel{����}{��}$
+Cl2$\stackrel{����}{��}$ +HCl��
+HCl��
����������ʱ��Ҫѡ���̼��Ϊ����������������7��̼ԭ�ӣ�����֧������һ�˸�������̼ԭ�ӱ�ţ�����3�ź�5��̼ԭ���ϸ�����������������Ϊ��3��3��5��5-�ļ����飻���л�������4��Hԭ�ӣ�����һ�ȴ�����4�֣��ʴ�Ϊ��3��3��5��5-�ļ����飻4��
��2���ٸ�������������������֪��2��3-����-4-�һ��������������6��̼ԭ�ӣ���2�ź�3��̼ԭ���ϸ���һ��������4��̼ԭ������һ���һ����ʽṹ��ʽΪCH3CH��CH3��CH��CH3��CH��C2H5�����ʴ�Ϊ��CH3CH��CH3��CH��CH3��CH��C2H5����
�������к����һ������һ�������3��λ������ֻ��һ���һ���ʽ����С�������Ľṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3���ǻ��д���һ��̼�����õ��Ӷԣ��ǻ��ĵ���ʽΪ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼����������������������д���ṹ��ʽ���ѶȲ���Ӧע�����-OH���������ĵ���ʽ������

| A�� | ���³�ѹ�£�3.2 g O3����������Ϊ1.2 NA | |
| B�� | ��״���£�2.24 L CCl4�к��е�C-Cl������ĿΪ0.4 NA | |
| C�� | ��״���£�11.2 L NO��11.2 L O2�������ԭ����Ϊ2 NA | |
| D�� | ��0.1 mol�Ȼ�������1 Lˮ�У�������Һ����0.1 NA Fe3+ |
 ��ͼ�Dz�����Һ�õ������ǣ�����ʽΪC6H12O6��ע��Һ�ı�ǩ�ϵIJ������ݣ��ݱ�ǩ���ṩ����Ϣ������˵��������ǣ�������
��ͼ�Dz�����Һ�õ������ǣ�����ʽΪC6H12O6��ע��Һ�ı�ǩ�ϵIJ������ݣ��ݱ�ǩ���ṩ����Ϣ������˵��������ǣ�������| A�� | ��ע��Һ�������ǵ�����������5% | |
| B�� | �����ǵ�Ħ������Ϊ180g•mol-1 | |
| C�� | ��ע��Һ���ܶ�ԼΪ1g•mL-1 | |
| D�� | ��ע��Һ�������ǵ����ʵ���Ũ��ԼΪ0.014 mol•L-1 |
| A�� | �ڱ�״���£�22.4Lˮ����������ĿΪNA | |
| B�� | 1mol•L-1 K2SO4��Һ����K+��ĿΪ2NA | |
| C�� | ��״���£�11.2L H2�к��еĵ�����Ϊ1NA | |
| D�� | O2��Ħ�����ԼΪ22.4 L•mol-1 |
| A�� | 12gʯī��C60�Ļ�Ϲ����У���NA��̼ԭ�� | |
| B�� | ��״���£�2.24Lˮ�к���0.1NA��ˮ���� | |
| C�� | 7.8g�������ƺ��еĹ��õ��Ӷ���Ϊ0.2NA | |
| D�� | 5.6g��������������ȼ�գ�����ת����ĿΪ0.2NA |
��֪������ݣ�
| ��ѧʽ | ����ƽ�ⳣ����25�棩 |
| HCN | K=4.9��10-10 |
| CH3COOH | K=1.8��10-5 |
| H2CO3 | K1=4.3��10-7 K2=5.6��10-11 |
��1��ȡ10.00mL����Һ���ü�ʽ�ζ�����ȡע����ƿ�У������������ƣ�
��2����0.2000mol•L-1��������Һ�ζ������ռ���Һ���÷�̪��Һ��ָʾ�����ζ�ʱ������ת��ʽ�ζ��ܵIJ������������ֲ�ͣ��ҡ����ƿ������ע����ƿ��ɫ�仯��ֱ���ζ��յ㣮�ﵽ�ζ��յ�ı�־����Һ��ɫ�ɺ�ɫ��Ϊ��ɫ���Ұ���ӱ��ֲ���
��3������ʵ������У�������ⶨ���ƫ�͵ģ�����ĸ��AC
A���ζ�ǰ��ʽ�ζ����¶������ݣ��ζ�����ʽ�ζ����¶˳������ݣ�
B����ʽ�ζ���������ˮϴ�Ӻ�ֱ��װ���
C���ζ�ʱ����Ӧ����ҡ��̫���ң�������Һ�彦����
D���ζ����յ�ʱ���ζ��ܼ�������Һ�Σ�
��4���������вⶨ���ݣ������õ��������ݣ���������ռ���Һ��Ũ�ȣ�0.4000mol•L-1��
| �ζ����� | ����Һ���/mL | �����������mL�� | |
| �ζ�ǰ������mL�� | �ζ��������mL�� | ||
| ��һ�� | 10.00 | 0.50 | 20.40 |
| �ڶ��� | 10.00 | 4.00 | 24.10 |
| ������ | 10.00 | 4.20 | 26.70 |
| A�� | �κ������£���Ӧ2CO2��g���T2CO��g��+O2��g���������Է����� | |
| B�� | ���ݷ�ӦI2+2S2O32-�T2I-+S4O62-����Ҫ�ⶨƽ����ϵI2+I-?I3-��ƽ��ʱ�� c��I2����������֪���ʵ���Ũ�ȵ�Na2S2O3��Һ���еζ�ʵ�� | |
| C�� | ���б��ӵı���Һ�е�������Ũ��ˮδ����ɫ����������Ϊ���ɵ����屽�������ڱ��� | |
| D�� | ����ij�����Ƿ���Fe2O3�IJ��������ǣ���Ʒ�������ˮ�ܽ�����ˣ�����Һ�еμ�KSCN��Һ |