��Ŀ����
11����ͼ������Fe����ʯī������1L����NaCl��Һ�У�����˵����ȷ���ǣ�������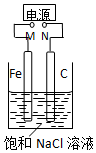
| A�� | M�ӵ�Դ������N�ӵ�Դ��������C�缫����Cu�缫���������Һ����CuSO4��Һ�����ʵ�������϶�ͭ | |
| B�� | M�Ӹ�����N������������Һ�е��˷�̪��Һ��C�缫��Χ��Һ��� | |
| C�� | M�Ӹ�����N�������������ձ�����Һ����1 L CuSO4��Һ����Ӧһ��ʱ����ձ��в�����ɫ���� | |
| D�� | M�Ӹ�����N��������������������������Ϊ22.4 L����״����ʱ������1molNaOH |
���� M�Ӹ�����N����������������2Cl--2e-=Cl2������������2H2O+2e-=2OH-+H2����M�ӵ�Դ������N�ӵ�Դ��������C�缫����Cu�缫���������Һ����CuSO4��Һ����������Fe-2e-=Fe2+����������Cu2++2e-=Cu���Դ˽����⣮
��� �⣺A��M�ӵ�Դ������N�ӵ�Դ��������C�缫����Cu�缫����������Fe-2e-=Fe2+����������Cu2++2e-=Cu������ʵ�������϶�ͭ����A����
B��M�Ӹ�����N��������C�缫����2Cl--2e-=Cl2���������̪����죬��B����
C��M�Ӹ�����N�������������ձ�����Һ����1L CuSO4��Һ����������2Cl--2e-=Cl2������������Cu2++2e-=Cu��û��������ͭ�������ɣ���C����
D��M�Ӹ�����N����������������2Cl--2e-=Cl2������������2H2O+2e-=2OH-+H2����������������������Ϊ22.4L����״����ʱ����������ʵ���Ϊ1mol������������0.5molH2��ͬʱ����1molNaOH����D��ȷ��
��ѡD��
���� ���⿼����صĹ���ԭ����ע��缫��Ӧ���жϣ��������ӷŵ�˳��Ϊ������Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
2���������з�Ӧ�����Ƶ������д������ʵ��ǣ�������
| A�� | ��ϩ��Cl2�ӳɣ�1��2-�������飩 | |
| B�� | ��ϩ��ˮ�ӳɣ��Ҵ��� | |
| C�� | �����뱽�������ȵ������·����ķ�Ӧ�������飩 | |
| D�� | ��������鰴�ձ���2��1�ڹ��������·�Ӧ�����ȼ��飩 |
16�����������ܴ���������ǣ�������
| A�� | K+��OH-��Na+��CO32- | B�� | Cl-��Na+��Ag+��NO3- | ||
| C�� | Fe3+��Cl-��Na+��OH- | D�� | NH4+��Cl-��Na+��OH- |
3������ұ��������ԭ���У������Ȼ�ԭ�����ǣ�������
| A�� | 2Ag2O$\frac{\underline{\;\;��\;\;}}{\;}$4Ag+O2�� | B�� | Fe2O3+3CO$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2�� | ||
| C�� | Fe+CuSO4�TFeSO4+Cu | D�� | 2NaC12$\frac{\underline{\;���\;}}{\;}$2Na+C12�� |
1�����ж�ijδ֪��Һ�����ӵļ��鷽���������һ����ȷ���ǣ�������
| A�� | �����Ȼ�����Һ������ɫ������һ������SO42- | |
| B�� | ȡ��Һ������ɫ��Ӧ�ǻ�ɫ��һ������Na+ | |
| C�� | ����̼������Һ������ɫ������һ������Ca2+ | |
| D�� | ����ϡ���������ɫ���壬һ������CO32- |
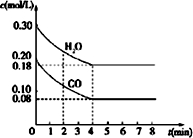 ��1����֪��һ���¶��£�����Ӧ��ƽ�ⳣ�����£�
��1����֪��һ���¶��£�����Ӧ��ƽ�ⳣ�����£�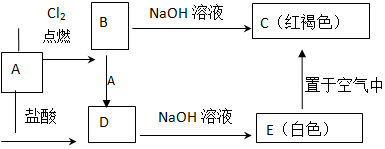
 ij�о���ԱӦ����ͼ����ʾ�ķ����о����ʵ����ʣ���������A����Ҫ�ɷ��������������ǿ�����ˮ������������������⣺
ij�о���ԱӦ����ͼ����ʾ�ķ����о����ʵ����ʣ���������A����Ҫ�ɷ��������������ǿ�����ˮ������������������⣺