��Ŀ����
������Һ�д��ڵ���ƽ�⣺CH3COOH?H++CH3COO-������������ȷ���ǣ�������
| A��0.10mol/L��CH3COOH��Һ�м�ˮϡ�ͣ���Һ��c��OH-����С |
| B��CH3COOH��Һ�м�������CH3COONa���壬ƽ�������ƶ� |
| C��������Һ������Ũ�ȵĹ�ϵ���㣺c��H+��=c��OH-��+c��CH3COO-�� |
| D��������pH=2��CH3COOH��Һ��pH=12��NaOH��Һ�������Ϻ���Һ��pH��7 |
���㣺���������ˮ��Һ�еĵ���ƽ��
ר�⣺����ƽ������Һ��pHר��
���������������ᣬ��Һ�д��ڵ���ƽ�⣬������Һ��������Ũ��С�ڴ���Ũ�ȣ����뺬�д�������ӵ�������������룬ϡ�ʹ����ܴٽ�������룬��ϵ���غ���
���
�⣺A����ˮϡ�ʹ��ᣬ������Ӻ�������Ũ�Ⱦ���С���¶Ȳ��䣬ˮ�����ӻ��������䣬��������������Ũ������A����
B��������Һ�м�������CH3COONa���壬���������Ũ������ƽ�������ƶ�����B����
C��������������ʣ�ֻ�в��ֵ��룬CH3COOH?H++CH3COO-�����ݵ���غ�ã�c��H+��=c��OH-��+c��CH3COO-������C��ȷ��
D�������£�pH=2��CH3COOH��Һ�д���Ũ�ȴ���0.01mol/L��pH=12��NaOH��Һ����������Ũ�ȵ���0.01mol/L���������ֵ������Ϻ���ʣ�࣬��Һ�����Զ�ʹ��Һ��pH��7����D��ȷ��
��ѡCD��
B��������Һ�м�������CH3COONa���壬���������Ũ������ƽ�������ƶ�����B����
C��������������ʣ�ֻ�в��ֵ��룬CH3COOH?H++CH3COO-�����ݵ���غ�ã�c��H+��=c��OH-��+c��CH3COO-������C��ȷ��
D�������£�pH=2��CH3COOH��Һ�д���Ũ�ȴ���0.01mol/L��pH=12��NaOH��Һ����������Ũ�ȵ���0.01mol/L���������ֵ������Ϻ���ʣ�࣬��Һ�����Զ�ʹ��Һ��pH��7����D��ȷ��
��ѡCD��
���������⿼��������ʵĵ��룬��ȷ������ʵ����ص��ǽⱾ��ؼ���ע�����ϡ��������Һ�и�����Ũ�ȱ仯��Ϊ�״��㣮

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
����ʵ�鲻�ܴﵽĿ���ǣ�������
| A��������KMnO4��Һ��ͨ��SO2��֤SO2�Ļ�ԭ�� |
| B�������Ȼ�����������ƹ���������ȡ���� |
| C���ö������̺�ϡ���ᷴӦ��ȡ���� |
| D������ˮ����ˮ�м������Ȼ�̼����ȡ���е��塢�� |
��һ�������£������ܱ������н��еĿ��淴ӦA��g��+3B��g��?2C��g��������˵���У���˵����һ��Ӧ�Ѿ��ﵽ��ѧƽ��״̬���ǣ�������
| A������B��������C�ֽ��������� |
| B��A��B��C��Ũ����� |
| C����λʱ������ n mol A��ͬʱ���� 2n mol C |
| D��A��B��C�ķ�����֮��Ϊ 1��3��2 |
���и��������У���Ϊͬ���칹����ǣ�������
| A�����ͺ��� |
| B��23Na��22Na |
| C�������Ǻ����� |
| D��CH3CH��CH3��CH3��CH3CH2CH2CH3 |
��NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A��25��ʱ��pH=13��1.0L Ba��OH��2��Һ�к��е�OH-��ĿΪ0.2NA |
| B����CO2��O2��ɵĻ�����й���NA�����ӣ����е���ԭ����Ϊ2NA |
| C����״���£�22.4L�����й��ۼ���ĿΪ6NA |
| D��1L 0.2mol?L-1 CH3COONa��Һ��1L 0.1mol?L-1 CH3COONa��Һ��ȣ�CH3COO-��Ŀ֮��Ϊ2��1 |
��2mol����A��3mol����B��һ�������·�����Ӧ��A��g��+3B��g��?2C��g����H��0���ﵽƽ��ʱ��B��ת����Ϊ50%����A��ת����Ϊ��������
| A��17% | B��20% |
| C��25% | D��50% |
���з�Ӧ�У���ԭ���Ͽ�����Ƴ�ԭ��ص��ǣ�������
| A��Ba��OH��2?8H2O��NH4Cl�ķ�Ӧ |
| B������������Ũ����ķ�Ӧ |
| C�������������ķ�Ӧ |
| D��ʯ��ʯ�ķֽⷴӦ |
���ⶨij��Һ������ֻ��Na+��CH3COO-��H+��OH-���֣�������Ũ�ȴ�С������˳��Ϊ��c��CH3COO-����c��Na+����c��H+����c��OH-��������ܵ������ ��������
| A������Һ������0.1mol/L��CH3COOH��Һ��0.1mol/L��NaOH��Һ�������϶��� |
| B������Һ������pH=3��CH3COOH��Һ��pH=11��NaOH��Һ�������϶��� |
| C������Һ������Ũ�ȵĹ�ϵ����c��CH3COO-��+c��OH-����c��Na+��+c��H+�� |
| D����������Һ�м�������NaOH������Ũ�ȴ�С�ɸı�Ϊ��c��CH3COO-����c��Na+����c��OH-����c��H+�� |
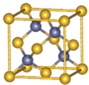 �黯��Ϊ�������뵼����ϣ������ṹ��ͼ��ʾ��
�黯��Ϊ�������뵼����ϣ������ṹ��ͼ��ʾ��