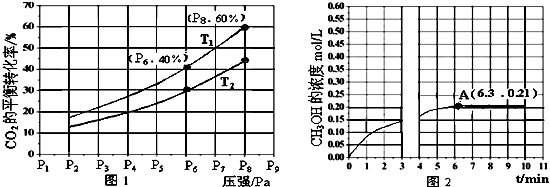
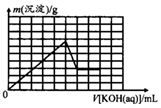
��Ŀ����
15��ij�о���ѧϰС�齫����װ����ͼ���ӣ�C��D��E��F��X��Y���Ƕ��Ե缫������Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ���Իش��������⣺
��1����ԴB���������Ǹ�����
��2������ռ���װ�����������������壬������������ʵ���֮����1��1��
��3����װ���е�ⷴӦ���ܻ�ѧ����ʽ��2CuSO4+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2Cu+O2��+H2SO4��
��4�����ñ�װ�ø�����п��GӦ����п���������п���������Һ����Ҫ�ɷ���ZnCl2���ѧʽ����
��5��װ�ö��е�������Y���������ɫ���
���� ����Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ��˵��F������OH-��FΪ���������֪AΪ������BΪ������C��E��G��XΪ������D��F��H��YΪ������
��1����ⱥ��ʳ��ˮʱ����̪���ļ��������������͵�Դ����������
��2����ⱥ��ʳ��ˮ���ֱ�����������������
��3���������ͭ��Һ�������ᡢͭ��������
��4��������п������ӦΪп������Ϊ�����ú�п���ӵ�����Һ�����Һ��
��5��������������������ɣ��������ƶ���
��� �⣺����Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ��˵��F������OH-��FΪ���������֪AΪ������BΪ������C��E��G��XΪ������D��F��H��YΪ������
��1�������Ϸ�����֪A�ǵ�Դ��������B��ԭ��صĸ������ʴ�Ϊ��������
��2����ⱥ��ʳ��ˮ�ĵ��ԭ���ǣ�2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2�������������������������ʵʩ1��1���ʴ�Ϊ��1��1��
��3���������ͭ��Һ�������ᡢͭ����������ⷽ��ʽΪ2CuSO4+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2Cu+O2��+H2SO4���ʴ�Ϊ��2CuSO4+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2Cu+O2��+H2SO4��
��4��������п������ӦΪп������Ϊ�����ú�п���ӵ�����Һ�����Һ����GΪп�������Һ����Ҫ�ɷ���ZnCl2���ʴ�Ϊ��п��ZnCl2��
��5���������Ե��������ԭ�����������������к��еĴ�����ɵ����ӻ���������Y���ƶ�������Y���������ɫ����ʴ�Ϊ��Y���������ɫ���
���� ���⿼����ԭ�����漰���͵�ƣ���ȷ�ж����������������ǽⱾ��ؼ���֪�������缫�Ϸ����ķ�Ӧ������ȷ��д�缫��Ӧʽ����Ŀ�ѶȲ���

 ������Ӧ���ϵ�д�
������Ӧ���ϵ�д� ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�| A�� | �����������ȥ�����е�Ʈ�������ý������ӵĴ����Զ����Գ�ȥ | |
| B�� | ��Һ�ͽ����Ϊ�ʵ����Եķ�ɢϵ | |
| C�� | FeCl3��Һ��Fe��OH��3����ı��������Ǻ��߿��Բ������������ | |
| D�� | ���ڽ���֮����ų����ã��������ۼ��ɴ�Ŀ��������Խ���Ƚ��ȶ� |
| A�� | 2CH4��g��+4O2��g����2CO2��g��+4H2O��1������H=+890kJ•mol-1 | |
| B�� | CH4��g��+2O2��g����CO2��g��+2H2O��1������H=+445kJ•mol-1 | |
| C�� | CH4��g��+2O2��g����CO2��g��+2H2O��1������H=+890kJ•mol-1 | |
| D�� | $\frac{1}{2}$CH4��g��+O2��g��=$\frac{1}{2}$CO2��g��+H2O��1������H=-445kJ•mol-1 |
| A�� | �۱�ϩ�Ľṹ��ʽΪ��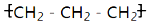 | B�� | �������Ľṹ��ʽ��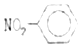 | ||
| C�� | �����ǵ�ʵ��ʽ��CH2O | D�� | ������ӵı���ģ�ͣ�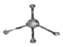 |
| A�� | NH3•H2O�ĵ���� | B�� | c��H+�� | C�� | ����ƽ�ⳣ��K�� | D�� | c��NH4+�� |