��Ŀ����
4��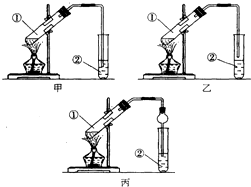 �����Ǽס��ҡ�����λͬѧ��ȡ���������Ĺ��̣�������벢Э������������ʵ������
�����Ǽס��ҡ�����λͬѧ��ȡ���������Ĺ��̣�������벢Э������������ʵ��������ʵ��Ŀ�ġ���ȡ��������
��װ����ơ��ס��ҡ�����λͬѧ�ֱ��������������ʵ��װ�ã�
��Ӽס�����λͬѧ��Ƶ�װ����ѡ��һ����Ϊʵ������ȡ����������װ�ã���ѡ���װ�����ң�ѡ��ס����ҡ�������ͬѧ����װ���еIJ����ܸij������θ���ܳ������������⣬������һ��Ҫ�����Ƿ�������
���������ۡ�
a�������Թܢ��й۲쵽������֪�������������������У���ɫ��״Һ�塢������ˮ������ζ��
b���Թܢ��б��� Na2CO3 ��Һ��������ABC�����ţ���
A���ܽ��Ҵ� B�����������������ܽ�� C���к�����
c�����Թܢ��з��������������ʵ������Ƿ�Һ��
d���������������Ļ�ѧ��Ӧ����ʽCH3COOH+C2H5OH$?_{��}^{Ũ����}$CH3COOC2H5+H2O��
e���Ҵ���ͭ����������ʱ�����Ա�����Ϊһ���д̼�����ζ�����ʣ�д���÷�Ӧ�Ļ�ѧ��Ӧ����ʽ2CH3CH2OH+O2$��_{����}^{ͭ����}$2CH3CHO+2H2O��
���� ��װ����ơ����������л����Ҵ������ᣬ����������ˮ������������
���������ۡ�������Ҵ���Ũ���������·���������Ӧ����������������ˮ����������������ˮ������ζ������̼������Һ�������Ҵ�����ȥ���ᣬ���������������ܽ�ȣ�ʹ��ֲ㣬���÷�Һ�ķ������룻�Ҵ���ͭ����������ʱ������ȩ��ˮ��
��� �⣺��װ����ơ����������л����Ҵ������ᣬ����������ˮ������������������װ����ȡ����ͬѧ����װ���еIJ����ܸij������θ���ܳ������������⣬�����ݻ��ϴ�Ҳ����ֹ���������ã�
�ʴ�Ϊ���ң���������
���������ۡ�a��������������ɫ��״Һ�壬������ˮ������ζ��
�ʴ�Ϊ��������ˮ������ζ��
b��ʵ�������ñ���̼������Һ��ȴ����������ԭ��һ������̼������Һ�е�ˮ�ܽ��Ҵ����Ҵ���ˮ����ܽ�ȴ�������������������̼�����ܸ����ᷴӦ������������ŵ�������������ζ�����������������ڱ���̼������Һ�������Ϳ��Ի�ý�Ϊ��������������Һ�壬���Ա���̼�������ã������Ҵ�����ȥ���ᣬ���������������ܽ�ȣ�ʹ��ֲ�������
�ʴ�Ϊ��ABC��
c���ñ���̼������Һ���������������õ�����Һ�廥�����ܣ������÷�Һ�����룬�ʴ�Ϊ����Һ��
d��������Ҵ���Ũ���������·���������Ӧ����������������ˮ����ѧ����ʽΪ��CH3COOH+C2H5OH$?_{��}^{Ũ����}$CH3COOC2H5+H2O��
�ʴ�Ϊ��CH3COOH+C2H5OH$?_{��}^{Ũ����}$CH3COOC2H5+H2O��
e���Ҵ���ͭ����������ʱ������ȩ��ˮ����Ӧ����ʽΪ��2CH3CH2OH+O2$��_{����}^{ͭ����}$2CH3CHO+2H2O��
�ʴ�Ϊ��2CH3CH2OH+O2$��_{����}^{ͭ����}$2CH3CHO+2H2O��
���� ���⿼�������������Ʊ�����Ŀ�ѶȲ�����ע����������������Ʊ�ԭ����ʵ�鷽����ѧϰ��ע��ʵ������������������

| A�� | 2H2��g��+O2��g��=2H2O��g����H=-483.6kJ/mol | B�� | 2H2��g��+O2��g��=2H2O��l����H=-483.6kJ/mol | ||
| C�� | 2H2��g��+O2��g��=2H2O��g����H=-241.8kJ/mol | D�� | 2H2��g��+O2��g��=2H2O��l����H=-241.8kJ/mol |
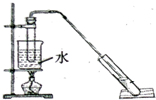 �����dzµ��㡱������Ϊ���ڴ������������������ζ������������ij��ȤС������ˮ�Ҵ��ͱ������Ʊ�����������������ʵ��������£�
�����dzµ��㡱������Ϊ���ڴ������������������ζ������������ij��ȤС������ˮ�Ҵ��ͱ������Ʊ�����������������ʵ��������£�| ���� | �Ҵ� | ���� | �������� |
| ������ | 46 | 60 | 88 |
| �е㣨�棩 | 78.5 | 117.9 | 77.1 |
| �ܶȣ�g/cm3�� | 0.789 | 1.05 | 0.90 |
�����Թ������3mL�Ҵ���Ȼ��һ��ҡ����һ������
����2mLŨ�����2mL�����ᣮ�ټ���������ʯ��
�ڰ���ͼʾ����װ�ã�ˮԡ���ȣ�ʹ����������ͨ������̼������Һ��Һ���ϣ�
�۴�С�Թ����ռ�Լ3mL����ʱֹͣ���ȣ�����С�Թܲ�������Ȼ���ô���ֲ㣮
�ܷ������������������2.3mL��
��1��ʵ�����Ʊ����������Ļ�ѧ����ʽΪ��CH3COOH+C2H5OH$?_{��}^{Ũ����}$CH3COOC2H5+H2O
�÷�Ӧ������Ϊȡ����Ӧ��Ũ�����ڴ˷�Ӧ�е������Ǵ�������ˮ����
��2��������м����ʯ��Ŀ���Ƿ�ֹ���У�����ڲ���ˮԡ���ŵ������Ⱦ��ȣ������ܵ�����������������
��3��������б���̼���Ƶ�������BD
A���к�������Ҵ�
B���к����Ტ���ղ����Ҵ�
C�������������ɣ�������IJ���
D�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�������
��4�����������Ҫ�õ��Ĺؼ������Ƿ�Һ©������������Ӧ�ôӸ��������ϲ�������ѡ��
A���²����� B���ϲ����� C��������
��5������ɵ����������IJ���Ϊ67.2%������=ʵ�ʲ���/���۲�����������λ��Ч���֣�
��6�����ſƼ�ˮƽ����ߣ���ҵ��������������ʱ���ʿ��ܻ����ߣ����ʲ����Ƿ��п��ܴﵽ100%���ܣ���п��ܡ������ܡ����������Ǹ÷�Ӧ�ǿ��淴Ӧ����Ӧ�ﲻ������ȫת���������
��4NH3 ��g��+5O2 ��g��?4NO ��g��+6H2O ��g������H=-907.2kJ•mol-1 K=1.1��1026
��4NH3 ��g��+4O2 ��g��?2N2O ��g��+6H2O ��g������H=-1104.9kJ•mol-1 K=4.4��1028
��4NH3 ��g��+3O2 ��g��?2N2 ��g��+6H2O ��g������H=-1269.2kJ•mol-1K=7.1��1034
����˵����ȷ���ǣ�������
| A�� | ����ѹǿ����Ӧ�Ģ�K���䣬��Ӧ�ٺ͢۵�K��С | |
| B�� | 500��ʱ��2NH3��g��+2O2 ��g��?N2O ��g��+3H2O ��g�� K=2.2��1028 | |
| C�� | 500��ʱ��N2 ��g��+O2 ��g��=2NO ��g����H=+181 kJ•mol-1 | |
| D�� | 500��ʱ��2 mol NH3 ��2.5 mol O2��Ϸ�����Ӧ�٣��ɷų�����453.6 kJ |
| A�� | ���ʵ��� | B�� | ���ʵ���Ũ�� | C�� | �������� | D�� | Ħ������ |
��2KClO3+4MnO2�T2KCl+2Mn2O7���� �� ��
��2Mn2O7�T4MnO2+3O2������������ ������
�����й�˵������ȷ���ǣ�������
| A�� | 1 mol KClO3���������������1 mol KCl����������� | |
| B�� | KClO3�ֽ����ʿ�����Ҫȡ���ڷ�Ӧ�� | |
| C�� | 1 g KClO3��1 g MnO2��0.1 g Mn2O7��ϼ��ȣ���ַ�Ӧ��MnO2����Ϊ1 g | |
| D�� | ����������������飬�ɼӿ�KClO3�ķֽ����� |