��Ŀ����
2�������ʵ���Ϊ���ĵļ����ǻ�ѧ����Ļ��������������ʵ�����صļ�����ȷ��Ϊ��������| A�� | ����CO CO2 O3�������壬���Ƿֱ���1Ħ��O����������������ʵ���֮��3��2��1 | |
| B�� | 5.6gCO��22.4L CO2�к���̼ԭ����һ����� | |
| C�� | ����£�11.2��X�����������Ϊ16g����X�����Ħ������Ϊ32 | |
| D�� | ngCl2����m��Clԭ�ӣ���٤������NA����ֵ���Ա�ʾΪ 35.5m/n |
���� A��CO��CO2��O3�������壬���Ǻ��е���ԭ�Ӹ���֮��Ϊ1��2��3������ԭ�ӵ����ʵ�����ͬ���趼Ϊ1mol����n��CO��=1mol��n��CO2��=$\frac{1}{2}$mol��n��O3��=$\frac{1}{3}$mol���ݴ˼���������������ʵ���֮�ȣ�
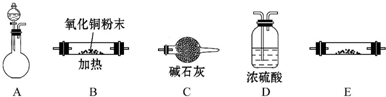
B��״����֪���������
C��Ħ�������ĵ�λ��g•mol-1��
D��ng Cl2�����ʵ���Ϊ$\frac{n}{71}$mol��������ԭ����Ϊ��$\frac{n}{71}$��2NA������$\frac{n}{71}$mol��2NA=m���ݴ˼��㰢���ӵ�������
��� �⣺A��CO��CO2��O3�������壬���Ǻ��е���ԭ�Ӹ���֮��Ϊ1��2��3������ԭ�ӵ����ʵ�����ͬ���趼Ϊ1mol����n��CO��=1mol��n��CO2��=$\frac{1}{2}$mol��n��O3��=$\frac{1}{3}$mol������������������ʵ���֮��Ϊ1��$\frac{1}{2}$��$\frac{1}{3}$=6��3��2����A����
B�������״����֪���������̼�����ʵ�������B����
C��Ħ�������ĵ�λ��g•mol-1������M�����Ħ��������32g•mol-1����C����
D��ng Cl2�����ʵ���Ϊ$\frac{n}{71}$mol��������ԭ����Ϊ��$\frac{n}{71}$��2NA������$\frac{n}{71}$mol��2NA=m�����ӵ�����NA����ֵ�ɱ�ʾΪ$\frac{35.5m}{n}$����D��ȷ��
��ѡD��
���� ���⿼�����ʵ������йؼ��㣬��Ŀ�ѶȲ���Ҫע�����ʵ����������������������Ŀ��Ũ�ȵļ��㹫ʽ�����ã�

 ��У����ϵ�д�
��У����ϵ�д���2������һ����������Ӳ�ȵ�ˮ��������Ҫ����MgCl2��CaCl2�����������Լ��е�һ���ֻ�ȫ�������������ˮ��ʵ�鷽������ѡ�Լ�����Ca��OH��2��Һ����NaOH��Һ���۱���Na2CO3��Һ���ܷ���ˮ��ʵ�鷽��������д�±���
| ʵ�鲽�� |
| ��Ӳˮ�м���٣�����������ֱ�����ٲ�������Ϊֹ�� |
| ��������Һ�м���ۣ�����������ֱ�����ٲ�������Ϊֹ�� |
| �������������ˣ�������� |
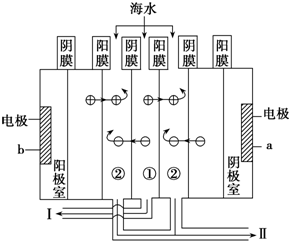
a�ӵ�Դ�ĸ���������ų����ǵ�ˮ�����ˮ����ˮ������
��4���ڣ�3����ij���ų���Ũ��ˮ����Ԫ�ص���������Ϊa%���������������е�����������Ϊ�嵥�ʣ�����1 000t��Ũ��ˮ���״���µ����������Ϊ1400am3��
| A�� | ����������������Һ | B�� | ���� | ||
| C�� | ��Ӿ | D�� | ���� |
| A�� | ���ܱ������м���3mol H2��1molN2����ַ�Ӧ��ɵõ�NH3������Ϊ2NA | |
| B�� | 0.1mol/L Na2SO4��Һ�к���Na+Ϊ0.2NA | |
| C�� | һ�������£�23 g Na��O2��Ӧ����Na2O2ʱʧȥ�ĵ�����ΪNA | |
| D�� | ��״���£�22.4L SO3�к��е���ԭ����Ϊ3NA |
| A�� | ��NaOH��Ӧ������һ��Ϊ0.3 mol | |
| B�� | n��Na+����n��Cl-�� ����Ϊ7��3 | |
| C�� | ����Ӧ��ת�Ƶĵ���Ϊn mol����0.3��n��0.5 | |
| D�� | n��NaCl����n��NaClO����n��NaClO3������Ϊ11��1��2 |
| A�� | 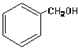 ����-OH ����-OH | B�� | 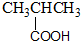 ����-CHO ����-CHO | ||
| C�� | 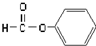 ȩ��-CHO ȩ��-CHO | D�� | CH3-O-CH3 ���� 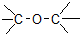 |

 ��
��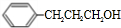
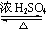
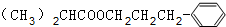 +H2O��
+H2O�� ��
��