��Ŀ����
8�� ����֬�ް�סԼ0.2g�������Ʒ�ĩ������ʯ�����ϣ�����֬���ϵ�ˮ���۲쵽��֬����ȼ��������
����֬�ް�סԼ0.2g�������Ʒ�ĩ������ʯ�����ϣ�����֬���ϵ�ˮ���۲쵽��֬����ȼ����������1��������ʵ���������ó����йع������Ƹ�ˮ��Ӧ�Ľ����ǣ���һ�����������ɣ��ڶ�����Ӧ�ų�������Na2O2��ˮ��Ӧ�Ļ�ѧ����ʽ2Na2O2+2H2O�T4NaOH+O2����
��2��ij�о���ѧϰС��������ͼ��ʾװ�ã����������ã�����ʵ�飬��֤���������ۣ�������֤��һ�����۵�ʵ������ǣ���Һ©������ʹˮ�����Թܣ��ٽ������ǵ�ľ���������ܿ�P����ľ����ȼ��֤����O2���ɣ�
��3����֪Na2O2������ˮ��ַ�Ӧ���ɵ���Һ����H2O2���ڣ�ʵ�飨2�����Թܣ���ͼ���м���ˮ��������ȫ�ܽ��Ҳ������������ɺ�ȡ���Թܣ����Թ��е����̪��Һ�������ǣ���Һ�ȱ�����ɫ��
��4����ȡ3.9g Na2O2���壬����50gˮ�г�ַ�Ӧ��������Һ����������Ϊ7.5%��
��5��H2O2���ȶ���ֽ⣬CuSO4��Һ�Թ������ֽ��д����ã���ͬѧ����������õ���Cu2+�������������ʵ����������ʵ��֤����ͬѧ�IJ�����ȷ��
����ѡ����������Լ����ձ����Թܡ�����������Ͳ������ƿ���ιܡ��ƾ��ơ�ҩ�ס�ľ������̪��Һ��2mol/L ���ᡢ2mol/L������Һ��2mol/L������Һ��5%��H2O2��Һ��CuSO4��Һ������ˮ��
| ���� | ʵ����� | Ԥ������ | ���� |
| ������õ���CuSO4��Һ�е�SO42- | ��ʢ��2mL5%��H2O2��Һ���Թ��еμ���������2mol/LH2SO4��Һ�����Ѵ����ǵ�ľ�������Թܣ� | ���������� | ������õIJ���SO42- |
| ������õ���CuSO4��Һ�е�Cu2+ | ��ʢ��2mL5%��H2O2��Һ���Թ��еμ���������CuSO4��Һ�����Ѵ����ǵ�ľ�������Թܣ� | �Թ����д������ݲ����������ǵ�ľ����ȼ | ������õ���Cu2+ |
���� ��1������֬���ϵ�ˮ���۲쵽��֬����ȼ��������˵������������ˮ��Ӧ�ų���������������غ㶨�ɿ�֪����NaOH��������
��2����Aͼ��ʾװ�ý���ʵ�飬��������������ȼ�ԣ�
��3��Na2O2��ˮ��Ӧ������H2O2��H2O2����ǿ�����Ժ�Ư���ԣ�
��4�������������ʵ���=$\frac{3.9g}{78g/mol}$=0.05mol��2Na2O2+2H2O�T4NaOH+O2������Ӧ����ˮ0.05mol������Ϊ0.05mol��18g/mol=0.9g����������0.025mol����Ϊ0.025mol��32g/mol=0.8g������������������Ϊ0.1mol������Ϊ0.1mol��40g/mol=4g�����������غ���㷴Ӧ����Һ������������������=$\frac{��������}{��Һ����}$��100%��
��5��H2O2���ȶ���ֽ⣬CuSO4��Һ�Թ������ֽ��д����ã�������ͭ����������ã�Ҳ��������������������ã����ʵ����֤�����������������Һ�м������������ͭ�������Ƿ�����������ƣ�
��� �⣺��1������֬���ϵ�ˮ���۲쵽��֬����ȼ����������֪��Ӧ�����������ҷ�Ӧ�ų������������ķ�ӦΪ2Na2O2+2H2O�T4NaOH+O2����
�ʴ�Ϊ����Ӧ�ų�������2Na2O2+2H2O�T4NaOH+O2����
��2����Aͼ��ʾװ�ý���ʵ�飬��֤��һ�����۵�ʵ�鷽���Ǵ�Һ©������ʹˮ�����Թܣ��ٽ������ǵ�ľ���������ܿ�P����ľ����ȼ��֤����O2���ɣ�
�ʴ�Ϊ����Һ©������ʹˮ�����Թܣ��ٽ������ǵ�ľ���������ܿ�P����ľ����ȼ��֤����O2���ɣ�
��3��Na2O2������ˮ��ַ�Ӧ�������������ƺ�����������������Һ�Լ��ԣ����ɵ���Һ����H2O2���ڣ��������������������ʹָʾ����ɫ��ȥ����Ӧ�����Թ��е����̪��Һ����������Һ�ȱ�����ɫ��
�ʴ�Ϊ����Һ�ȱ�����ɫ��
��4�������������ʵ���=$\frac{3.9g}{78g/mol}$=0.05mol��2Na2O2+2H2O�T4NaOH+O2������Ӧ����ˮ0.05mol������Ϊ0.05mol��18g/mol=0.9g����������0.025mol����Ϊ0.025mol��32g/mol=0.8g������������������Ϊ0.1mol������Ϊ0.1mol��40g/mol=4g����Һ����=3.9g+50g-0.8g=53.1g��������������=$\frac{4g}{53.1g}$��100%=7.5%���ʴ�Ϊ��7.5%��
��5������������õ���CuSO4��Һ�е�SO42-����ʢ��2mL 5%��H2O2��Һ���Թ��еμ���������2mol/L H2SO4��Һ�����Ѵ����ǵ�ľ�������Թܣ�����������������õIJ���SO42-������������õ���CuSO4��Һ�е�Cu2+����ʢ��2mL 5%��H2O2��Һ���Թ��еμ��������� CuSO4��Һ�����Ѵ����ǵ�ľ�������Թܣ��Թ����д������ݲ����������ǵ�ľ����ȼ��������õ���Cu2+��
�ʴ�Ϊ��
| ʵ����� | Ԥ������ | ���� |
| ��ʢ��2mL 5%��H2O2��Һ���Թ��еμ���������2mol/L H2SO4��Һ�����Ѵ����ǵ�ľ�������Թܣ� | ���������� | ������õIJ���SO42- |
| ��ʢ��2mL 5%��H2O2��Һ���Թ��еμ��������� CuSO4��Һ�����Ѵ����ǵ�ľ�������Թܣ� | �Թ����д������ݲ����������ǵ�ľ����ȼ | ������õ���Cu2+ |
���� ���⿼��̽���������Ƶ����ʡ���������ֽ�Ĵ������ã���ȷʵ��ԭ���ǽⱾ��ؼ���ͬʱ����ѧ��ʵ������������۲�����������ܽ���������ȣ���������ʵ��������ʵ�飬ע�����֪ʶ�Ļ��ۺ����ã��Ѷ��еȣ�

| A�� | ���顢CO�ϳ�����������2CH4+2CO$\stackrel{����}{��}$CH3COOCH2CH3 | |
| B�� | ϩ����ˮú���������ʻ��ϳɷ�Ӧ��RCH�TCH2+CO+H2$\stackrel{����}{��}$RCH2CH2CHO | |
| C�� | �������ھƻ�ø������ת��Ϊ�ƾ� | |
| D�� | ����ϩ�ϳɾ�����ϩ |
| A�� | �ܷ���������Ӧ | B�� | ��ʹ��ˮ��ɫ | ||
| C�� | ��һ�������¿��������� | D�� | �������ӳ�ֻ�����ɱ�ȩ |
| A�� | O2-�Ľṹʾ��ͼ��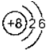 | |
| B�� | ���Ȼ�̼�ĵ���ʽ��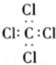 | |
| C�� | HClO�ĽṹʽΪ��H-O-Cl | |
| D�� | �õ���ʽ��ʾ�Ȼ�����ӵ��γɹ��̣� |
| A�� | ��ԭ����������������֮��Ϊ 19 | |
| B�� | ���ǻ����һ����Ԫ�� | |
| C�� | ${\;}_{26}^{45}$Fe��${\;}_{26}^{56}$Fe�Ļ�ѧ���ʼ�����ȫ��ͬ | |
| D�� | ������ԭ��˥��� 2 �����Ӻ���${\;}_{26}^{43}$Fe |
| A�� | ���л����壬����KI ��Һ������ CCl4��ȡ���� | |
| B�� | �����л�����ϩ��ͨ��������һ�������·�Ӧ��ʹ��ϩת��Ϊ���� | |
| C�� | �������л���Ũ HNO3 ��Ũ H2SO4�����䵹�뵽 NaOH ��Һ�У����ã���Һ | |
| D�� | ��ϩ�л��� SO2������ͨ������ KMnO4 ��Һ��ϴ�� |

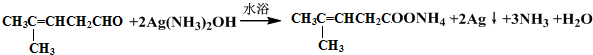
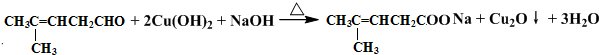 ��Ȼ������Ӧ�����Һ�м���ϡ��������Һ�����ԣ��ٵμ���������ˮ�����������̼̼˫�������ɵ��л�����Ľṹ��ʽ��
��Ȼ������Ӧ�����Һ�м���ϡ��������Һ�����ԣ��ٵμ���������ˮ�����������̼̼˫�������ɵ��л�����Ľṹ��ʽ��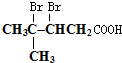 ��
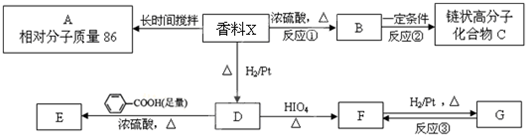
��

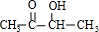 ��C
��C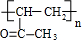 ��
��

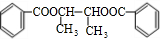 +2H2O��
+2H2O��