��Ŀ����
ʵ������һδ֪Ũ�ȵ�NaOH��Һ��ijѧ��Ϊ�ⶨNaOH��Һ��Ũ����ʵ�����н�������ʵ�飮�������������գ�
����һ������250mL 0.10mol/L ϡ�������Һ��
�������ȡ20.00mL����ϡ���������ƿ�У����μ�2��3�η�̪��Һ��ָʾ�������Լ����Ƶı�NaOH��Һ���еζ����ظ������ζ�����4�Σ���¼�������£�
��1�����������ȡ20.00mL��ϡ�����õ��������� ���ζ��ﵽ�յ�������� ��
��2���ζ�������Ӧ ֱ���ζ����յ㣮
��3�������������ݣ��ɼ������NaOH��Һ��Ũ��Ϊ
��4��������ʵ���У����в���������������ȷ������ɲⶨ���������ҺŨ��ֵ��ƫ�ߵ��� ����ѡ�۷֣���
A��ʢ��Һ�ĵζ���������ˮ��ϴ��ֱ�Ӽ����Һ���еζ�
B����ƿˮϴ��ֱ��װ����Һ
C���ζ������յ�ʱ�����Ӷ����ζ��ܶ�����
D����ʽ�ζ��ܵζ�ǰ���첿�������ݣ��ζ�����ʧ
E���ζ������յ����ʱ���ֵζ��ܴ�����һ����Һ��
����һ������250mL 0.10mol/L ϡ�������Һ��
�������ȡ20.00mL����ϡ���������ƿ�У����μ�2��3�η�̪��Һ��ָʾ�������Լ����Ƶı�NaOH��Һ���еζ����ظ������ζ�����4�Σ���¼�������£�
| ʵ���� | NaOH��Һ��Ũ�� ��mol/L�� | �ζ����ʱ������NaOH��Һ�������mL�� | ����������Һ�������mL�� |
| 1 | 0.10 | 20.02 | 20.00 |
| 2 | 0.10 | 20.00 | 20.00 |
| 3 | 0.10 | 19.00 | 20.00 |
| 4 | 0.10 | 19.98 | 20.00 |
��2���ζ�������Ӧ
��3�������������ݣ��ɼ������NaOH��Һ��Ũ��Ϊ
��4��������ʵ���У����в���������������ȷ������ɲⶨ���������ҺŨ��ֵ��ƫ�ߵ���
A��ʢ��Һ�ĵζ���������ˮ��ϴ��ֱ�Ӽ����Һ���еζ�
B����ƿˮϴ��ֱ��װ����Һ
C���ζ������յ�ʱ�����Ӷ����ζ��ܶ�����
D����ʽ�ζ��ܵζ�ǰ���첿�������ݣ��ζ�����ʧ
E���ζ������յ����ʱ���ֵζ��ܴ�����һ����Һ��
���㣺�к͵ζ�
ר�⣺����ƽ������Һ��pHר��
��������1����ȡ20.00mL��ϡ����������ʽ�ζ��ܣ����ݴ���Һ�м����̪����ҺΪ��ɫ����Ӧ��ȫ���������ƹ�������Һ��ɺ�ɫ�жϴﵽ�յ�����
��2�������к͵ζ��У��۾�Ӧע�ӵ�����ƿ����Һ��ɫ�仯���ζ��յ��жϣ�
��3�����жϵζ����ݵ���Ч�ԣ�Ȼ�����HCl+NaOH=NaCl+H2O����֪n��HCl��=n��NaOH������������NaOH��Һ�����ʵ���Ũ�ȣ�
��4������c�����⣩=
��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��2�������к͵ζ��У��۾�Ӧע�ӵ�����ƿ����Һ��ɫ�仯���ζ��յ��жϣ�
��3�����жϵζ����ݵ���Ч�ԣ�Ȼ�����HCl+NaOH=NaCl+H2O����֪n��HCl��=n��NaOH������������NaOH��Һ�����ʵ���Ũ�ȣ�
��4������c�����⣩=
| V(��)��c(��) |
| V(����) |
���
�⣺��1����ȡ20.00mL��ϡ����������ʽ�ζ��ܣ��÷�̪��Һ��ָʾ�������һ��NaOH��Һ����ʱ����Һ��ɫǡ������ɫ��Ϊdz��ɫ���Ұ�����ڲ���ɫ��Ϊ�ζ��յ㣻
�ʴ�Ϊ����ʽ�ζ��ܣ����һ��NaOH��Һ����ʱ����Һ��ɫǡ������ɫ��Ϊdz��ɫ���Ұ�����ڲ���ɫ��
��2���ζ�ʱ���۾�Ҫע������ƿ����Һ��ɫ�ı仯���ʴ�Ϊ����ƿ����Һ��ɫ�ı仯��
��3��4�εζ����ı�Һ������ֱ�Ϊ��19.92mL��19.90mL��18.90mL��19.88mL����2��������ȥ������3�����ĵı�Һ��ƽ�����Ϊ19.90mL��Ȼ�����HCl+NaOH=NaCl+H2O����֪n��HCl��=n��NaOH����
��0.10mol/L��20.00mL=c��NaOH����19.90mL����ã�c��NaOH��=0.1005mol/L��
�ʴ�Ϊ��0.1005mol/L��
��4��A��ʢ��Һ�ĵζ���������ˮ��ϴ��ֱ�Ӽ����Һ���еζ�����ҺŨ�Ƚ��ͣ����V���꣩ƫ����c�����⣩=
��������֪c�����⣩ƫ��A����
B����ƿˮϴ��ֱ��װ����Һ������Һ�����ʵ������䣬��V���꣩��Ӱ�죬����c�����⣩=
��������֪c�����⣩���䣬��B����
C���ζ������յ�ʱ�����Ӷ����ζ��ܶ��������V���꣩ƫ����c�����⣩=
��������֪c�����⣩ƫ��C��ȷ��
D����ʽ�ζ��ܵζ�ǰ���첿�������ݣ��ζ�����ʧ�����V���꣩ƫ����c�����⣩=
��������֪c�����⣩ƫ��D��ȷ��
E���ζ������յ����ʱ���ֵζ��ܴ�����һ����Һ�����V���꣩ƫ����c�����⣩=
��������֪c�����⣩ƫ��E��ȷ��
��ѡCDE��
�ʴ�Ϊ����ʽ�ζ��ܣ����һ��NaOH��Һ����ʱ����Һ��ɫǡ������ɫ��Ϊdz��ɫ���Ұ�����ڲ���ɫ��
��2���ζ�ʱ���۾�Ҫע������ƿ����Һ��ɫ�ı仯���ʴ�Ϊ����ƿ����Һ��ɫ�ı仯��
��3��4�εζ����ı�Һ������ֱ�Ϊ��19.92mL��19.90mL��18.90mL��19.88mL����2��������ȥ������3�����ĵı�Һ��ƽ�����Ϊ19.90mL��Ȼ�����HCl+NaOH=NaCl+H2O����֪n��HCl��=n��NaOH����
��0.10mol/L��20.00mL=c��NaOH����19.90mL����ã�c��NaOH��=0.1005mol/L��
�ʴ�Ϊ��0.1005mol/L��
��4��A��ʢ��Һ�ĵζ���������ˮ��ϴ��ֱ�Ӽ����Һ���еζ�����ҺŨ�Ƚ��ͣ����V���꣩ƫ����c�����⣩=
| V(��)��c(��) |
| V(����) |
B����ƿˮϴ��ֱ��װ����Һ������Һ�����ʵ������䣬��V���꣩��Ӱ�죬����c�����⣩=
| V(��)��c(��) |
| V(����) |
C���ζ������յ�ʱ�����Ӷ����ζ��ܶ��������V���꣩ƫ����c�����⣩=
| V(��)��c(��) |
| V(����) |
D����ʽ�ζ��ܵζ�ǰ���첿�������ݣ��ζ�����ʧ�����V���꣩ƫ����c�����⣩=
| V(��)��c(��) |
| V(����) |
E���ζ������յ����ʱ���ֵζ��ܴ�����һ����Һ�����V���꣩ƫ����c�����⣩=
| V(��)��c(��) |
| V(����) |
��ѡCDE��
������������Ҫ����������к͵ζ��IJ������衢���������Ѷ��еȣ������к͵ζ���ԭ���ǽ���Ĺؼ���

��ϰ��ϵ�д�
 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д� �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�
�����Ŀ
����ʽΪC4H10O�Ĵ��������������ɵ�ȩ�У�������
| A��2�� | B��3�� | C��4�� | D��5�� |
���й���NaCl�ķ����ȷ���ǣ�������
| A�������� | B�������� |
| C���Ȼ��� | D������ |
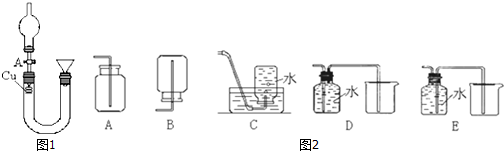
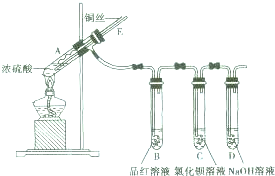 ijʵ��С��ͬѧΪ��̽��ͭ��Ũ����ķ�Ӧ������������ʵ�飬ʵ��װ����ͼ��ʾ��ʵ�鲽�裺
ijʵ��С��ͬѧΪ��̽��ͭ��Ũ����ķ�Ӧ������������ʵ�飬ʵ��װ����ͼ��ʾ��ʵ�鲽�裺